
Evaluating Caries Risk
Clinicians must assess each patient’s risk for developing decay in order to effectively implement a caries prevention program.
This course was published in the February 2014 issue and expires February 28, 2017. The authors have no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Identify the stages of caries development.
- Discuss the factors that influence caries risk in children.
- List the caries risk assessment tools that are available and explain their purpose.
- Detail appropriate intervention for children at high caries risk.
Dental caries is not only expensive to treat, but it can also negatively impact children’s quality of life. Recent studies have found a correlation between children’s dental health, school performance, and psychosocial well-being.4–6 Children who experience caries are at increased risk for malocclusion of the permanent dentition, oral pain, failure to thrive, and, in rare cases, severe dental infection resulting in death.7
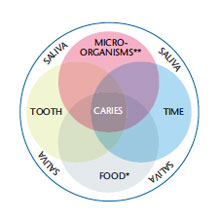
Caries is a multifactorial disease process involving the characteristics of the host, microflora, environment, and time (Figure 1). A child’s potential to develop caries is mediated by a complex interaction of these factors. When pathologic factors are left unchecked, caries will result. In children younger than 6, any caries lesion (noncavitated or cavitated) or missing (due to caries) is considered early childhood caries (ECC).8 Any sign of smooth-surface caries in children younger than 3 is an indication of severe ECC.8
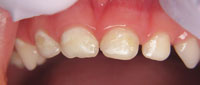
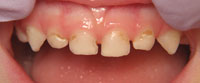
In the earliest stages of disease, ECC presents as a noncavitated white spot lesion (Figure 2). At this point, preventive measures can stop the disease progression. Without intervention, the lesion will become cavitated (Figure 3). At this stage of the process, restorative treatment is generally required to prevent progression to acute dental infection.
In order to reduce the burden of dental caries, clinicians must consider each individual’s risk for developing disease. This principle is currently known as caries risk assessment (CRA), and it focuses on the measures that most dramatically impact an individual’s risk for disease.
INDIVIDUAL RISK FACTORS
Tooth development begins as early as 11 weeks in utero, with the permanent incisors and first molars entering the maturation stage at birth. Therefore, teeth are susceptible to damaging factors that occur during fetal development and early childhood. Malformation of tooth enamel is one of the most common and visually apparent signs that a tooth has experienced pre-eruptive damage, and it inherently increases caries risk. Poorly formed enamel may be thin, inadequately mineralized (weak), and difficult to clean.
Children with special health care needs (CSHCN) have physical, developmental, mental, sensory, behavioral, cognitive, or emotional impairments that require medical management.9 As a group, CSHCN may experience more dental defects and an increased risk for caries. For example, systemic diseases, such as congenital biliary atresia, biliary hypoplasia, and chronic kidney disease, can have a profound effect on the primary and permanent dentition.10,11 In these diseases, the enamel is poorly formed, resulting in malformation and discoloration. The outcome is teeth that are difficult to keep clean and esthetically displeasing. Children born prematurely experience increased rates of infection and receive more early-life medical treatments. Such factors may contribute to generalized enamel defects, and an increased risk for caries.12 Localized mineralization defects, as occur in severely hypomineralized first permanent molars, also increase caries risk. For CSHCN, challenges in maintaining optimal oral health may also be related to difficulty performing quality toothbrushing, dietary restrictions, and decreased ability to access dental care.13
Medical conditions and medications may also impact the quality and quantity of saliva produced. Any condition that reduces salivary flow increases caries risk. For example, children who have received cranial radiation treatment for cancer (with radiation in the field of major salivary glands), as well as those with juvenile Sjögren’s syndrome and moderate to severe asthma, all experience reduced salivary flow rates.14 Among children with chronic medical problems, each condition and medication should be evaluated for such effects.
Dental plaque is composed of aggregated bacterial colonies that together form a biofilm. This biofilm provides a zone of safety where diverse species of bacteria can survive and function as a community at the tooth surface. Though more than 700 types of bacteria live in the mouth, only certain species contribute to caries formation.15 Mutans streptococci (MS) are the most prevalent organisms associated with ECC development. Equipped to survive and thrive in the oral environment, which is frequently acidic and filled with competing microflora and antibodies, MS produce lactic acid as a byproduct of carbohydrate consumption. Subsequently, demineralization of the tooth surface results when acid remains in contact with the tooth. The presence of high MS levels in the plaque and saliva of preschool children is associated with an increased risk of caries.16 These bacteria may be transmitted from mother to child (vertical transmission) or later in life from other sources (horizontal transmission). Children whose parents or siblings have caries are at increased risk of ECC because these virulent bacteria may be transferred from person to person.
ENVIRONMENTAL FACTORS
Diet may be one of the most important factors associated with caries risk, as MS need an energy source for survival—preferably sucrose. This is hazardous because lactic acid is produced as a byproduct of sucrose consumption. Subsequently, the pH in the oral cavity is lowered. This promotes demineralization, the leaching of minerals from the tooth surface, and, ultimately, loss of tooth structure. In many cases, caries risk is affected not only by the foods that children eat, but also the drinks they consume. The sugars added to sweetened beverages and processed foods may actually be more cariogenic than natural sugar, further increasing caries risk.17
The burden of dental caries is disproportionally felt by individuals who are economically disadvantaged. Approximately 80% of ECC in the US is experienced by a quarter of the population—mainly those of low socioeconomic status.18 The relationship between a child’s social status, risk factors, and dental caries is multidimensional. For example, economically disadvantaged families have reduced access to dental care and utilize other health care services less frequently. This suggests that these families may have lower perceived need for health care.20 A mother’s educational status, cultural beliefs, and stress levels may also lead to practices that are detrimental to oral health, including poor food choices, negative feeding habits, and inadequate oral hygiene.19
FREQUENCY AND DURATION
The pH in the oral cavity drops after eating, at which time the teeth become susceptible to demineralization. Over time, the mouth returns to a neutral pH and remineralization of the tooth surface occurs. When children eat frequently, the pH of the oral cavity stays acidic and the demineralization process exceeds the remineralization process.20 This explains why frequent snacking is associated with an increased caries rate. Infant bottle-feeding habits, sipping frequently from a bottle during the day, and going to bed with cariogenic liquids in a bottle are highly correlated with ECC.
Visual plaque on children’s teeth is associated with MS colonization and high caries risk.21,22 Effective brushing disrupts the bacterial activity by removing the biofilm and decreases the duration of time the tooth is exposed to acidic byproducts. Orthodontic brackets and bands create areas that are difficult to clean, making effective oral hygiene challenging. The resulting increase in the time the tooth is exposed to the biofilm may contribute to demineralization and cavitation during orthodontic treatment.23
PROTECTIVE FACTORS
When considering risk assessment, protective factors are just as important as the factors that increase caries risk. Caries protective factors include: establishment of a dental home, optimal oral hygiene, good dietary habits, adequate salivary flow, and fluoride use.24 A dental home is where comprehensive, continuously accessible, family centered, coordinated, compassionate, and culturally effective care is provided.25 Data show that when children have a dental home, they are more likely to receive appropriate early interventions for caries, such as professionally applied fluoride varnish, protective sealants, and anticipatory guidance.
Good dietary habits reduce the food sources that are available for the biofilm and the duration of time the teeth are exposed to acidic pH. Children often have different diets than adults. They typically eat three meals per day with one to three additional snacks.
Effective toothbrushing regularly disrupts biofilm. Children who brush their teeth two times daily with parental assistance will achieve optimal biofilm removal. An inherited but important factor in protection is high-quality saliva. Saliva buffers a low oral pH; is a reservoir for minerals (including phosphate and calcium, which contribute to remineralization) and antibodies (which neutralize bacteria and viruses); and rinses debris from the oral cavity.26 Fluoride provides caries protection by reducing the acid solubility of enamel, attracting minerals for remineralization, and decreasing the rate of transport of minerals out of the enamel surface.27
TOOLS TO ASSESS RISK
Determining which pediatric patients are at risk for ECC can be difficult. Fortunately, several excellent age-based tools are available to identify children at greatest risk. This paper will focus on caries-risk assessment tools (CATs) from the American Academy of Pediatric Dentistry (AAPD), California Dental Association (CDA), and the American Dental Association (ADA).
Most CATs attempt to assess the balance of risk factors and protective factors. When risk factors outweigh protective factors, the child is at increased risk for developing dental disease. Risk factors that are highly correlated with ECC include the presence of MS bacteria and cavitated or noncavitated lesions and a history of dental treatment. These risk factors must be weighed strongly when identified by a CAT. While CATS are effective at estimating caries risk, the practitioner’s clinical judgment is also important. CATs also help clinicians engage the patient/family in a discussion of prevention strategies.
Though CATs are incredibly useful, they are not without drawbacks. One underlying problem is CATs are generally sensitive, but not highly specific. This means they will correctly identify individuals at high risk for the disease, but may not accurately identify individuals who are at low risk for the disease.28 The result is that low-risk individuals may receive more preventive treatments than necessary. Research validating CATs to predict future caries is limited; further study needs to be completed in this area.29
The AAPD has CATs for three patient age groups: 0 to 3; 0 to 5; and 6 and older.30 Each CAT contains a table that is divided into three categories: biological factors, protective factors, and clinical findings. The biological section includes environmental risk factors, such as active caries present in mother, socioeconomic status, diet, special health care needs, and immigrant status. The protective section includes sources of fluoride exposure (drinking fluoridated water or using fluoride supplements, receiving topical fluoride from health professionals, and daily toothbrushing with fluoride toothpaste), and if the child has a dental home. The clinical findings section includes active lesions and malformed teeth, restored/missing teeth, salivary flow rate, visual plaque build-up on teeth, and MS levels. The patient is then ranked as low, moderate, or high risk for each section. This approach supports the assessment of both protective and risk factors. The forms are relatively simple in design, easy to use, and can be found at aapd.org/policies.
The CDA has developed CATs for both dental and medical professionals. They were created by researchers and clinicians who have embraced the concept of caries management by risk assessment (CAMBRA). There are currently two age-specific forms: CAMBRA age 0 to 5, and CAMBRA age 6 through adult.31,32 The CAMBRA tools are designed to follow the flow of an office visit, and they cover a wide range of risk and protective factors. The first part of the forms contains topics that clinicians can bring up for discussion during the parent interview, such as caries risk indicators (history of dental disease of parent/patient, patient’s restoration history, special health care needs, presence of a dental home where the patient is routinely seen); biological caries risk factors (diet, medical diagnosis/medications that affect salivary flow); and biological and nonbiological protective factors (mother is caries-free for 3 years, patient has dental home and is routinely seen, fluoride exposure, patient’s oral hygiene routine). The last section covers risk factors found during the clinical examination (cavitated and noncavitated lesions, restorations placed in the past 2 years, visual plaque on teeth, orthodontic appliances, and salivary flow). CAMBRA also helps clinicians determine when it is time to consider testing the bacterial microflora of the parent or child. These forms include an area to identify goals for improvement and the recommended follow-up interval. They can be accessed by visiting: cda.org/Portals/0/journal/journal_102007.pdf.
The ADA developed CATs in conjunction with the AAPD and the CAMBRA coalition. The forms are simple, easy to use, and represent a blending of the AAPD and CAMBRA styles. Two forms are available: caries risk assessment age 0 to 6, and caries risk assessment for those older than 6.33 The ADA forms can be downloaded directly from ada.org, completed online, and printed for the patient to take home. They also contain an open section at the bottom to identify the patient’s self-selected prevention goals. The forms are divided into three sections: contributing conditions (fluoride exposure, diet, family caries experience, and dental home with routine visits), general health conditions (special health care needs, receiving chemo/radiation therapy, and medications that reduce salivary flow), and clinical conditions (cavitated/noncavitated lesions or restorations, teeth missing due to caries, visible plaque, malformed enamel, defective restorations, orthodontic appliances, and salivary flow). For each question, there is a color-coded risk category, which makes it easy for clinicians and providers to instantly visualize the factors that increase caries risk.
PATIENTS AT HIGH RISK
Traditional methods of delivering prevention education to patients have not been overwhelmingly successful. In general, these methods increase the patient’s knowledge about the condition, but do not produce a significant change in oral health behavior.34 Over the past 10 years, the motivational interviewing technique has been proven to promote behavior change. Motivational interviewing is a brief therapeutic counseling session with the patient that involves five key principles: respect the patient’s autonomy and choices within the context of a trustful treatment relationship; don’t immediately attempt to deliver advice during a clinical encounter; ask open-ended questions that engage the patient; empower the patient to personally choose which areas he or she feels can be improved; and follow up with a brief telephone call or in-person interaction to discuss progress.35 The key is to not just deliver advice; the patient must be part of the decision-making process. A CAT can be used to initiate a discussion about creating new habits that will decrease caries risk. It is important not to overwhelm a patient. If the patient can identify just one area he or she is willing to work on, this is progress. Follow up can be conducted via a telephone call or a discussion at the next scheduled visit.
Regular dental appointments for the child and the rest of the family should be encouraged, including completion of any restorative needs. In many cases, patients may have Medicaid insurance or a limited ability to pay. If the office does not routinely offer care for these patient populations, a list of providers who accept Medicaid insurance or provide services at reduced rates should be maintained so that referrals can be provided. For children at high caries risk, an increased frequency of recare appointments is appropriate. Disease can progress rapidly in the primary dentition, thus, the conventional 6-month follow-up period may be too long. In these situations, a 3-month to 4-month interval is appropriate.36
Providers should recommend age-appropriate fluoride use, including systemic and topical application. The drinking of tap water should be encouraged if the family lives in an area with fluoridated water. One of the best ways to deliver fluoride is through regular toothbrushing with fluoride toothpaste. Therefore, parents should be encouraged to assist the child with quality brushing until the child can effectively brush on his or her own. The parent’s participation will increase the likelihood that plaque is removed from all tooth surfaces and that the appropriate amount of toothpaste is used. Professionally applied fluoride varnish can also be used to reduce caries risk. Fluoride varnish has been associated with about a 40% reduction of caries in both the primary and permanent dentition.36 Fluoride can be applied at preventive visits up to four times a year in high-risk patients.
A child’s diet may be one of the most difficult items to assess and modify, but it greatly impacts caries risk. For infants/toddlers, the most important, yet difficult, change is to decrease bottle-feeding. Parents should be counseled to begin weaning at 12 months. Furthermore, allowing children to sleep with a bottle should be discouraged. Sippy cups function in nearly the same fashion as a bottle, thus parents should be advised to only place water in sippy cups. When juice is consumed from a sippy cup, it increases exposure to sucrose. Parents should be encouraged to limit juice intake or only serve it at mealtimes. Similarly, soda should be consumed only on special occasions. The intake of sports drinks should be limited to during sports practice or games. Because of the high sugar content, parents also should be counseled to decrease the availability of candy or other types of junk food. Instead, focus can be directed toward increasing the availability of snacks that are less likely to contribute to decay, such as meats, cheese, fresh fruits and vegetables, and air-popped popcorn. As an additional benefit, dietary changes that benefit oral health will benefit the whole body.
Because pathologic bacteria are central to the development of dental caries, consideration should be given to promoting a shift toward a less pathologic microflora. Chlorhexidine (CHX) and xylitol-containing products may reduce pathologic bacteria levels. There is some evidence that CHX varnish may be effective but it is not available in the US.37 At this time, there is insufficient evidence to support the widespread use of chlorhexidine mouthrinse to prevent caries. Adjunct mouthrinses that contain xylitol and fluoride may be beneficial, and they do not cause the extrinsic staining associated with CHX mouthrinse.38 There is limited evidence to support the use of chewing gum containing xylitol as a prevention strategy.39 Parents/caregivers with active caries may consider chewing xylitol gum after meals to help prevent the transfer of the bacteria to their young children.
CONCLUSION
Caries risk assessment is a contemporary practice that improves prevention of ECC in the dental office. A number of CATs are currently available and can be used to highlight individual risk and protective factors. These provide the clinician with a method of targeting risky behaviors, amplifying positive preventive practices, and engaging the patient in discussion. Effective change takes place in the context of trusting relationships established with care providers. Dental hygienists are well positioned to build strong relationships with families and counsel patients to reduce their caries risk.
REFERENCES
- National Institute of Dental and Craniofacial Research. Oral Health in America: A Report of the Surgeon General. Available at: nidcr.nih.gov/ datastatistics/surgeongeneral/report/executivesummary.htm. Accessed January 19, 2014.
- Dye BA, Thornton-Evans G. Trends in oral health by poverty status as measured by Healthy People 2010 objectives. Public Health Rep. 2010;125:817–830.
- Rashewsky S, Parameswaran A, Sloane C, Ferguson F, Epstein R. Time and cost analysis: pediatric dental rehabilitation with general anesthesia in the office and the hospital settings. Anesth Prog. 2012;59:147–153.
- Guarnizo-Herreno CC, Wehby GL. Children’s dental health, school performance, and psychosocial well-being. J Pediatr. 2012;161:1153–1159.
- Jackson SL, Vann WF Jr, Kotch JB, Pahel BT, Lee JY. Impact of poor oral health on children’s school attendance and performance. Am J Public Health. 2011;101:1900–1906.
- Seirawan H, Faust S, Mulligan R. The impact of oral health on the academic performance of disadvantaged children. Am J Public Health. 2012;102:1729–1734.
- Finucane D. Rationale for restoration of carious primary teeth: a review. Eur Arch Paediatr Dent. 2012;13:281–292.
- American Academy of Pediatric Dentistry, American Academy of Pediatrics. Policy on early childhood caries (ECC): classifications, consequences, and preventive strategies. Pediatr Dent. 2008;30(Suppl 7):40–43.
- American Academy of Pediatric Dentistry. Definition of special health care needs. Available at: aapd.org/media/Policies_Guidelines/ D_SHCN.pdf. Accessed January 19, 2014.
- Amaral TH, Guerra Cde S, Bombonato-Prado KF, Garcia de Paula E Silva FW, de Queiroz AM. Tooth pigmentation caused by bilirubin: a case report and histological evaluation. Spec Care Dentist. 2008;28:254–257.
- Koch MJ, Buhrer R, Pioch T, Scharer K. Enamel hypoplasia of primary teeth in chronic renal failure. Pediatr Nephrol. 1999;13:68–72.
- Nelson S, Albert JM, Lombardi G, et al. Dental caries and enamel defects in very low birth weight adolescents. Caries Res. 2010;44:509–518.
- Lewis CW. Dental care and children with special health care needs: a population-based perspective. Acad Pediatr. 2009;9:420–426.
- Paganini M, Dezan CC, Bichaco TR, de Andrade FB, Neto AC, Fernandes KB. Dental caries status and salivary properties of asthmatic children and adolescents. Int J Paediatr Dent. 2011;21:185–191.
- Aas JA, Paster BJ, Stokes LN, Olsen I, Dewhirst FE. Defining the normal bacterial flora of the oral cavity. J Clin Microbiol. 2005;43:5721–5732.
- Thenisch NL, Bachmann LM, Imfeld T, Leisebach Minder T, Steurer J. Are mutans streptococci detected in preschool children a reliable predictive factor for dental caries risk? A systematic review. Caries Res. 2006;40:366–374.
- Edgar WM. Extrinsic and intrinsic sugars: a review of recent UK recommendations on diet and caries. Caries Res. 1993;27(Suppl 1):64–67.
- Kaste LM, Selwitz RH, Oldakowski RJ, Brunelle JA, Winn DM, Brown LJ. Coronal caries in the primary and permanent dentition of children and adolescents 1-17 years of age: United States, 1988-1991. J Dent Res. 1996;75:631–641.
- Kim Seow W. Environmental, maternal, and child factors which contribute to early childhood caries: a unifying conceptual model. Int J Paediatr Dent. 2012;22:157–168.
- Chankanka O, Marshall TA, Levy SM, et al. Mixed dentition cavitated caries incidence and dietary intake frequencies. Pediatr Dent. 2011;33:233–240.
- . Weber-Gasparoni K, Goebel BM, Drake DR, et al. Factors associated with mutans streptococci among young WIC-enrolled children. J Public Health Dent. 2012;72:269–278.
- Meurman PK, Pienihakkinen K. Factors associated with caries increment: a longitudinal study from 18 months to 5 years of age. Caries Res. 2010;44:519–524.
- Gorelick L, Geiger AM, Gwinnett AJ. Incidence of white spot formation after bonding and banding. Am J Orthod. 1982;81:93–98.
- Featherstone JD. Caries prevention and reversal based on the caries balance. Pediatr Dent. 2006;28:128–132.
- Medical Home Initiatives for Children With Special Needs Project Advisory Committee. American Academy of Pediatrics. The medical home. Pediatrics. 2002;110:184–186.
- Featherstone JD. The continuum of dental caries—evidence for a dynamic disease process. J Dent Res. 2004;83:C39–42.
- Institute of Medicine (US) Standing Committee on the Scientific Evaluation of Dietary References Intakes. Dietary Reference Intakes for Calcium, Phosphorous, Magnesium, Vitamin D, and Fluoride. Washington, DC: National Academy Press; 1997:288–313.
- Gao X, Di Wu I, Lo EC, Chu CH, Hsu CY, Wong MC. Validity of caries risk assessment programmes in preschool children. J Dent. 2013;41:787–795.
- Carson SJ. Limited evidence for existing caries assessment systems. Evid Based Dent. 2013;14:10–11.
- American Academy on Pediatric Dentistry Council on Clinical Affairs. Policy on use of a caries-risk assessment tool (CAT) for infants, children, and adolescents. Pediatr Dent. 2008;30(Suppl 7):29–33.
- Ramos-Gomez FJ, Crall J, Gansky SA, Slayton RL, Featherstone JD. Caries risk assessment appropriate for the age 1 visit (infants and toddlers). J Calif Dent Assoc. 2007;35:687–702.
- Featherstone JD, Domejean-Orliaguet S, Jenson L, Wolff M, Young DA. Caries risk assessment in practice for age 6 through adult. J Calif Dent Assoc. 2007;35:703-707, 710–713.
- American Dental Association. Caries Risk Assessment Form (Ages 0-6). Available at: ada.org/sections/professionalResources/pdfs/topics_caries_under6.pdf. Accessed January 19, 2014.
- Kay E, Locker D. A systematic review of the effectiveness of health promotion aimed at improving oral health. Community Dent Health. 1998;15:132–144.
- Weinstein P. Motivational interviewing concepts and the relationship to risk management and patient counseling. J Calif Dent Assoc. 2011;39:742–745.
- Marinho VC, Worthington HV, Walsh T, Clarkson JE. Fluoride varnishes for preventing dental caries in children and adolescents. Cochrane Database Syst Rev. 2013;7:CD002279.
- Autio-Gold J. The role of chlorhexidine in caries prevention. Oper Dent. 2008;33:710-716.
- Subramaniam P, Nandan N. Effect of xylitol, sodium fluoride and triclosan containing mouth rinse on Streptococcus mutans. Contemp Clin Dent. 2011;2:287–290.
- Fontana M, Gonzalez-Cabezas C. Are we ready for definitive clinical guidelines on xylitol/polyol use? Adv Dent Res. 2012;24:123–128.
From Dimensions of Dental Hygiene. February 2014;12(2):53–56,59.



