
Tricho-Dento-Osseous Syndrome
A form of ectodermal dysplasia, this genetic disease asserts a variety of effects on the oral cavity.
This course was published in the February 2014 issue and expires February 28, 2017. The authors have no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Define tricho-dento-osseous syndrome (TDO).
- Discuss TDO’s impact on oral health.
- Identify the role of the dental hygienist in treating patients with TDO.
Many developmental and/or hereditary conditions exhibit oral signs or actively impact dental health. Ectodermal dysplasia (ED)—a group of hereditary conditions with 150 to 170 variations, many of which are rare—is an example. Patients with ED may experience abnormal ectodermal development of the skin, hair, nails, salivary glands, and/or teeth. The skin may be dry and pale, the hair fine and sparse (lanugo), and the nails dystrophic and brittle. The dentition exhibits varying degrees of hypodontia and abnormal development.
The prevalence of ED is estimated at seven per 10,000 births. Hypohidrotic ED is the most common type, but there is great variability in signs and symptoms.1–7 Individuals affected by this condition often require extensive dental reconstructive procedures to acquire a functional, esthetic dentition.
TRICHO-DENTO-OSSEOUS SYNDROME
Tricho-dento-osseous syndrome (TDO) is an uncommon form of ED.8–13 The literature consists primarily of case reports and genetic studies, with little prevalence data due to its rarity. In the United States, three large families in Virginia, North Carolina, and Tennessee are affected by TDO. Much of the research concerning the dental manifestations of TDO has been conducted at the University of North Carolina (UNC) School of Dentistry in Chapel Hill. UNC?researchers continue to study and observe cohorts from the affected families.
TDO is inherited as an autosomal dominant trait and is caused by mutations in the distal-less-3 homeobox (DLX3) gene, with the most common mutation being a four-base-pair deletion.14–17 The DLX3 gene has an important role in the development of the epidermis, branchial arches (embryonic tissues giving rise to specialized structures in the head and neck), and dental tissues.9,14 Autosomal dominance indicates that descendants of an individual with the gene have a 50% chance of acquiring the disorder. Genetic analyses have revealed a mutation in the DLX3 gene, which results in altered function of the gene product that produces manifestations of the syndrome.9–11 Early epidemiologic investigations suggested three subtypes—TDO I, TDO II, and TDO III, based primarily on variation in bone defects. Recent genetic mapping, however, has discounted these subtypes, as it was discovered that these groups share identical genetic mutation.15 There is simply great phenotypic variation among those affected.
Though highly variable, TDO syndrome is characterized by abnormalities in ectodermal structures (eg, hair, nails, and teeth) and mesenchymal structures, or bone.8,11,15,18–22 Bone changes are part of the syndrome in approximately 50% of cases, and are especially prevalent among adults and older individuals (but do not affect height or weight).9,18 Though craniofacial bones tend to be significantly affected, vision and intellectual capabilities remain untouched.9 Other reported abnormalities associated with TDO include clinodactyly (curvature of the fifth finger) and skin lesions.8,23 The bone defects are not necessarily pathologic and are typically of little significance; however, consequences of the resultant dental defects can be severe.
IMPACT ON DENTAL HEALTH
Dental defects are present in both the primary and permanent dentitions. The most common dental manifestations are enamel hypoplasia (abnormal enamel development) and taurodontism (elongated pulp chambers).13,19,20 Some patients display a prognathic mandible, impacted and congenitally missing teeth, and small tooth size. Tooth eruption can be an issue, and small tooth size gives the appearance of increased spacing in the dental arch.12
Enamel hypoplasia is defective enamel development that occurs as a result of environmental or hereditary factors. The etiology of enamel hypoplasia is elusive, necessitating a thorough medical/dental history. Among the many environmental factors responsible for ameloblastic disruption during amelogenesis are bacterial and viral infections, nutritional deficiencies, and trauma. Inflammation from extensive caries in primary teeth or trauma to the developing tooth bud can affect succedaneous teeth, resulting in hypoplasia of the developing enamel. This is sometimes referred to as Turner’s tooth. Chemical injury to teeth can be the culprit. Excessive ingested fluoride and certain antibiotics can affect amelogenesis—both resulting in hypoplastic enamel.1–3 Amelogenesis can even be disrupted by the birth process, producing a microscopic area of mild hypoplasia, called the neonatal line.24 The degree of enamel hypoplasia depends on the causative factor, stage of enamel development (matrix formation, mineralization, or maturation) at the time of the injury, and duration of the injury.1–3 Consequentially, hypoplasia can be localized, affecting one or several teeth, or generalized to the entire dentition. Clinical appearance is highly variable, ranging from barely noticeable white opacities to blatant opaque or brown spots and striations to pitting and mottling (Figure 1).1–3
Although hereditary disturbances of enamel are exhibited in many conditions and syndromes, the most common is amelogenesis imperfecta (AI). It consists of a group of inherited disorders of enamel, with a range in prevalence of one in 700 to one in 15,000.20 There are four types of AI: hypoplastic, hypomaturation, hypocalcified, and hypomaturation-hypoplastic, with each containing several subtypes. Each type correlates with the stage of enamel development in which the defect occurs.3 Clinical appearance varies according to type, ranging from normal to severely affected. The enamel may appear pitted, grooved, and furrowed—similar to generalized hypoplasia. Affected teeth are also prone to chipping from the defective union of enamel and dentin, and the exposed dentin absorbs stains—resulting in discolored teeth (Figure 2).1–4,20
Enamel hypoplasia is detected in virtually all patients with TDO. Among this patient population, enamel is 1?8th to 1?4th of its normal thickness, with randomly distributed pits and depressions.8–13,20,21 Both hypocalcification and hypomaturation of enamel structure have been reported. Several studies have used histologic examination with light microscopy and electron microscopy, which revealed irregular enamel development with local calcification defects.13,17,25 Teeth are characterized by yellow-brown discoloration with pitting and attrition, increasing this population’s risk of dental caries.12 Clinical appearance of enamel in those affected by TDO can be identical to the type-IV hypomaturation-hypoplastic variant of AI. There was much discussion in the literature about whether AI hypomaturation-hypoplastic type with taurodontism and TDO were the same condition. Subsequent research, however, determined that they are both genetically distinct conditions.11,15,22
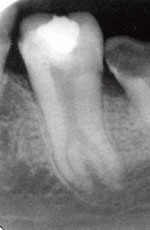
The other common dental manifestation among patients with TDO is taurodontism. Taurodontism is characterized by an enlarged pulp chamber and body in molar teeth, due to displacement of the furcation area of the tooth.12,19,20 Radiographically, molar teeth exhibit an elongated rectangular shape with short roots (Figure 3). This defect is common in both primary and permanent dentitions among patients with TDO. It is often seen in mandibular first permanent molars, but can also present in multiple teeth.12 While taurodontism is seen in patients with TDO, it is also common among populations affected by other common hereditary conditions, such as Down’s syndrome and Kleinfelter’s syndrome.1 These syndromes, however, typically do not exhibit multiple taurodontic teeth. The enlarged pulp chambers in close proximity to thin hypoplastic enamel of patients with TDO contribute to dentoalveolar abscess formation, a commonly encountered dental complication.12,19,20 It is believed to result from attrition/pulpal exposure and increased susceptibility to and progression of dental caries. A dentoalveolar abscess can develop quickly under these conditions. Endodontic treatment of taurodontic teeth is especially challenging, due to the proximity and apical displacement of the roots.26
Patients with TDO may experience hypersensitivity of the teeth, malocclusion due to attrition and loss of tooth structure, and esthetic problems. Premature tooth loss, caused by dental abscesses and various other complications, may occur.25 Dental management requires a team approach, including the general and/or pediatric dentist, dental hygienist, orthodontist, endodontist, oral surgeon, prosthodontist, and oral radiologist.12
The osseous component of TDO varies but is reported to occur in half of those affected.13,23 Bones are very strong, due to an increase in density and thickness—with no change in frequency of bone fractures.18 Haldeman et al16 studied a cohort of individuals with TDO and found increased bone mineral density in the lumbar spine, the femoral neck of the hip, and the radius and ulna. As the individuals aged, various areas of the body continued to experience increased bone density and thickness—especially in the cranial bones. This resulted in dolichocephaly, the premature closure of the sutures of the skull, which prevents lateral growth of the head.8,12,15,18,19,21 It is characterized by a long head/facial appearance that does not become fully noticeable until maturity. Other manifestations include shortened mandibular rami, sclerotic mastoids, and obliteration of maxillary sinuses over time.16,23 No medical intervention is typically prescribed for the bone manifestations of TDO.
Hair and fingernails are additional ectodermal structures that can be affected by TDO. All newborns with TDO are born with curly hair, which may straighten with age.8,12 This characteristic distinguishes TDO from other syndromes because curly/coarse hair is present in the majority of people with TDO. Recent studies using electron microscopy have demonstrated an actual defect in the structure of hair shafts in those with TDO, displaying abnormal shaft morphology and reduced diameters.11 Thin, flat, and brittle nails are also characteristic of TDO. As the nails increase in length, they also become prone to superficial splitting.12,15,18 Toenails may also be affected.12
A CASE REVIEW
This case study is of a 23-year-old woman (Patient A) with TDO. The patient’s maternal great-grandfather had TDO, and he passed it on to three of his five children. Two of the three affected children passed the gene mutation to their children.
The characteristic kinky hair present at birth left no doubt that Patient A was affected by the same inherited genetic mutation as multiple members of her maternal grandfather’s family. Those suspicions were shared by pediatric professionals who recommended genetic testing. Patient A was diagnosed with TDO at age 8. She participated in a DNA analysis at the UNC School of Dentistry years later. The analysis confirmed Patient A has a four-base pair deletion on the DLX3 gene, resulting in a definitive diagnosis of TDO. This exact deletion is seen in other Southern populations, believed to have descended from a Scottish immigrant.
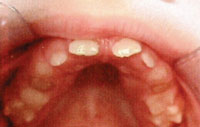
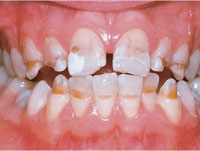
FIGURE 4. At age 8, Patient A exhibited small, hypoplastic teeth, indicative of tricho-dento-osseous syndrome.
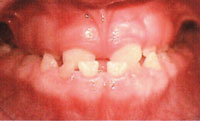
FIGURE 5. Patient A also exhibited a prognathic mandible at age 8, another sign of tricho-dento-osseous syndrome.
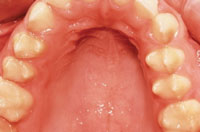
Patient A began the extensive process to ensure a functional, esthetic dentition at age 8. Early intraoral photographs demonstrate enamel hypoplasia with associated discoloration, and small, widely spaced teeth with localized severe attrition (Figure 4). She also presented with a prognathic mandible (Figure 5), a characteristic of TDO.14 At age 10, Patient A also displayed taurodontism in both primary and permanent teeth and impacted mandibular canines (Figure 6). At this point, she had already received endodontic therapy on tooth #8—the result of a dentoalveolar abscess, which is commonly encountered among those with TDO.
From age 13 to age 23, Patient A received numerous oral procedures at several oral specialty offices. Full-mouth reconstruction was accomplished, with most teeth ultimately restored with porcelain-fused-to-metal gold crowns (Figure 7). Additional abscesses developed on tooth #19 and tooth #31 (a consequence of hypoplasia and taurodontism), necessitating endodontic therapy. Impacted mandibular canines were extracted and replaced with dental implants. As a result of normal masticatory function, the crown of tooth #12 fractured completely at the cervico-enamel junction, requiring extraction of the remaining root structure and replacement with a dental implant. Additional procedures included bone grafting and crown lengthening procedures to improve esthetics.
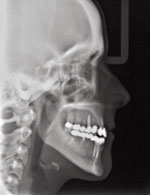
Cephalometric radiographs exposed at various points during Patient A’s life have revealed increased bone density and subsequent diagnosis of dolichocephaly (Figure 8). In addition to increased bone density, Patient A exhibits an enlarged, mineralized area of cranium with obliteration of diploe space (area between inner and outer cortex).
Patient A’s hair has straightened since birth.8,12 She has mild fingernail involvement, with a tendency for splitting upon increased length. Though changes in bone density are observed in cephalometric X-rays, the increased density has not caused any problems or pathologic symptoms.
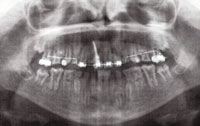
FIGURE 6. This panoramic radiograph shows the presence of taurodontism in Patient A at age 10.
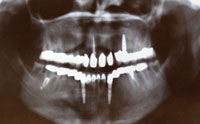
FIGURE 7. Now 23, Patient A has had a full mouth reconstruction, as seen in this panoramic radiograph.
ROLE OF THE DENTAL HYGIENIST
Patients with TDO often require long-term restorative treatment, and are frequently seen in the dental office to address oral complications associated with the syndrome. Recare appointments must include regular evaluation of oral hygiene procedures and anticipatory guidance for the next step in treatment. Clinicians will likely need to recommend desensitizing agents for hypersensitivity and home fluoride therapy for patients who are at increased caries risk. Among this patient population, a power toothbrush is preferred for its consistent vibratory action and to reduce the pressure applied to compromised dentition. Dietary counseling should also be considered, including suggestions for caries prevention and sensitivity reduction. Ensuring the soft tissues are in optimal health is key, as this is critical when proceeding with restorative care—especially bonding approaches that are typically used as transitional therapy in children and adolescents.
Significant in periodontal debridement, the rectangular shape of taurodontic teeth alter the morphology of the furcation area. Dental hygienists may find better resolution of inflammation in these areas as the morphology makes it less susceptible to periodontal diseases. When pocketing or gingival recession occurs, significant periodontal destruction must take place before furcation involvement transpires.12
As oral health professionals become more involved in overall health care, knowledge of a multitude of diseases and conditions will be required. The variability in manifestations of TDO syndrome has been noted throughout the literature;15 however, knowledge of TDO and its various characteristics is useful not only for dental providers as they deliver care to affected patients, but also to assist with diagnosis in otherwise unaware individuals. Generalized enamel hypoplasia can be the sign of various environmental disturbances, or a hereditary disorder necessitating interdisciplinary collaboration and communication with the various dental specialties to ensure continuity of care and monitoring of complications. Dental hygienists should be knowledgeable about the various types and etiologies of enamel hypoplasia and taurodontism, documenting such findings in the dental record.
Sensitivity to the psychological impact of such extensive dental procedures is warranted when providing care for those with TDO, especially in children who are beginning the arduous process of dental restorative treatment and management. The financial burden associated with management of TDO can be significant, and may serve as a barrier to accessing dental care. Office staff may need to help patients and their families investigate payment options and navigate the medical/dental care system. The entire dental team should work collaboratively in providing optimal care for patients with TDO syndrome, so as to ensure the best possible outcomes.
ACKNOWLEDGEMENTS
”Patient A” is Mackenzie Bacot, co-author of this article, who graciously shared her health history to educate oral health professionals about TDO. The authors would also like to thank J. Timothy Wright, DDS, MS, James W. Bawden Distinguished Professor and chair of the Department of Pediatric Dentistry at UNC School of Dentistry, for his guidance in preparing this manuscript.
REFERENCES
- Neville BW, Damm DD, Allen CM, et al. Epithelial pathology. In: Dolan J, ed. Oral and Maxillofacial Pathology. 3rd ed. St. Louis: Saunders Elsevier; 2009:741–743.
- Regezi JA, Sciubba JJ, Jordan RC. In: Dolan J, ed. Oral Pathology: Clinical Pathologic Correlations. 5th ed. St. Louis: Saunders Elsevier; 2012:378–379.
- Sapp JP, Eversole LR, Wysocki GP. Contemporary Oral and Maxillofacial Pathology. 2nd ed. St. Louis: Mosby Elsevier;2003:3, 17.
- Guckes AD, Roberts MW, McCarthy GR. Pattern of permanent teeth present in individuals with ectodermal dysplasia and severe hypodontia suggests treatment with dental implants. Pediatr Dent. 1998;20:278–280.
- Talo T, Acun Kaya F. The effects of ectodermal dysplasia on periodontal tissues. Journal of International Dental and Medical Research. 2009;2(2):53–57.
- Bani M, Tezkirecioglu AM, Akal N, Tuzuner T. Ectodermal dysplasia with anodontia: a report of two cases. Eur J Dent. 2010;4:215–222.
- Freiman A, Borsuk D, Barankin B, Sperber GH, Krafchik B. Dental manifestations of dermatologic conditions. J Am Acad Dermatol. 2009;60:289–298.
- Lichtenstein J, Warson R, Jorgenson R, Dorst JP, McKusick VA. The tricho-dento-osseous (TDO) syndrome. Am J Hum Genet. 1972;24:569–582.
- Wright TJ, Kula K, Hall K, Simons JH, Hart TC. Analysis of the tricho-dento-osseous syndrome genotype and phenotype. Am J Med Genet. 1997;72:197–204.
- Price JA, Wright JT, Kula K, Bowden DW, Hart TC. A common DLX3 gene mutation is responsible for tricho-dento-osseous syndrome in Virginia and North Carolina families. J Med Genet. 1998;35:825–828.
- Wright JT, Hong SP, Simmons D, Daly B, Uebelhart D, Luder HU. DLX3 c.561_562delCT mutation causes attenuated phenotype of tricho-dento-osseous syndrome. Am J Med Genet A. 2008;146:343–349.
- Al-Batayneh OB. Tricho-dento-osseous syndrome: diagnosis and dental management. Int J Dent. 2012;2012:514692.
- Wright JT, Roberts MW, Wilson AR, Kudhail R. Tricho-dento-osseous syndrome. Features of the hair and teeth. Oral Surg Oral Med Oral Pathol. 1994;77:487–493.
- Duverger O, Isaac J, Zah A, Hwang J, Berdal A, Lian J, Morasso M. In vivo impact of DLX3 conditional inactivation in neural crest-derived craniofacial bones. J Cell Physiol. 2014;228:654–664.
- Islam M, Lurie AG, Reichenberger E. Clinical features of tricho-dento-osseous syndrome and presentation of three new cases: an addition to clinical heterogeneity. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2005;100:736–742.
- Haldeman RJ, Cooper LF, Hart TC, et al. Increased bone density associated with DLX3 mutation in the tricho-dento-osseous syndrome. Bone. 2004;35:988–997.
- Sharpiro SD, Quattromani FL, Jorgenson RJ, Young RS. Tricho-dento-osseous syndrome: heterogeneity or clinical variability. Am J Med Genet. 1983;16:225–236.
- Jorgenson RJ, Warson RW. Dental abnormalities in the tricho-dento-osseous syndrome. Oral Surg Oral Med Oral Pathol. 1973;36:693–700.
- Price JA, Bowden DW, Wright JT, Pettenati MJ, Hart TC. Identification of a mutation in DLX3 associated with tricho-dento-osseous (TDO) syndrome. Hum Mol Genet. 1998;7:563–569.
- UNC School of Dentistry. Developmental defects of the teeth: tricho-dento-osseous syndrome, amelogenesis imperfecta, Available at: dentistry.unc.edu/research/defects/pages/tdo.htm. Accessed January 14, 2014.
- National Organization for Rare Disorders. Rare disease information. Available at: rarediseases.org/rare-disease-information/rare-diseases/byID/386/viewFullReport. Accessed January 14, 2014.
- Price JA, Wright JT, Walker SJ, Crawford PJ, Aldred MJ, Hart TC. Tricho-dento-osseous syndrome and amelogenesis imperfecta with taurodontism are genetically distinct conditions. Clin Genet. 1999;56:35–40.
- Seow WK. Trichodentoosseous (TDO) syndrome: case report and literature review. Pediatr Dent. 1993;15:355–361.
- Eli I, Sarnat H, Talmi E. Effect of the birth process on the neonatal line in primary tooth enamel. Pediatr Dent. 1989;11:220–223.
- Atar M, Korperich EJ. Systemic disorders and their influence on the development of dental hard tissues: a literature review. J Dent. 2010;38:296–306.
- Bharti R, Chandra A, Tikku AP, Wadhwani KK. “Taurodontism” an endodontic challenge: a case report. J Oral Sci. 2009;51:471–474.
From Dimensions of Dental Hygiene. February 2014;12(2):60–64.



