Assessing Chronic Periodontitis
Periodontal probing and dental radiography are important components of evaluating the location and severity of this common oral disease.
This course was published in the January 2016 issue and expires January 31,2019. The authors have no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Discuss the subclassifications of chronic periodontitis.
- Identify the tools used in periodontal assessment and disease diagnosis.
- Explain the role of traditional radiography in periodontal assessment.
Chronic periodontitis is an inflammatory destruction of the supporting apparatus of the teeth. It results in inflammation and destruction of alveolar bone and the periodontal fibers that insert into cementum, thereby exposing cementum to dental plaque within a periodontal pocket. If left unchecked, the mechanical stability of a tooth may become compromised, or the localized chronic infection may become acute—both resulting in tooth loss. Early detection of the signs of chronic periodontitis is essential to halting disease progression and improving outcomes.1 As such, clinicians should be familiar with the subclassifications of chronic periodontitis, the tools used to assess and interpret its location and severity, and how to document anatomic changes over time.
The subclassifications of chronic periodontitis are based on whether the anatomic changes are localized (attachment loss is present in less than 30% of tooth surfaces) or generalized (attachment loss is present in more than 30% of tooth surfaces). These classifications are then organized into groups based on the amount of attachment loss present: mild (1 mm to 2 mm of attachment loss), moderate (3 mm to 4 mm of attachment loss), and severe (greater than 5 mm of attachment loss). Chronic periodontitis is distinguished from less common forms of periodontal diseases by the presence of certain risk factors, such as tobacco use and accumulations of dental plaque, as well as the presence of calculus. Diabetes, human immunodeficiency virus infection, and chronic stress may also contribute. As chronic periodontitis progresses, changes in epithelial tissues occur, including the junctional epithelium receding apically from the cementoenamel junction (CEJ) and thickening of the sulcular epithelium with increased extension of epithelial ridges into subjacent connective tissue. Changes to connective tissue and bone include increased number of inflammatory cells, destruction of periodontal ligament (PDL) fibers, and exposure of root cementum to the sulcular or oral environment or both. The gingival consistency may be edematous or fibrotic, or both. Accompanying attachment loss is characterized by increased periodontal pocket depths, gingival recession, or both.
ASSESSMENT PROCESS
Probing and charting of periodontal tissues to assess clinical attachment losses and the use of radiography to evaluate the underlying osseous support and possible presence of furcation involvement are essential components of accurate periodontal assessment. Pocket probing depths (or sulcus depths) are measured using gentle pressure from the gingival margin to the most coronal aspects of the junctional epithelium while paralleling the long axis of the tooth. Because inflamed epithelium is easily penetrated by periodontal probing, true periodontal pocket depths can only be determined histologically. Therefore, the term “probing depth” is preferred over “pocket depth” for clinical assessments.2 The most commonly used probe is a manual circular probe with a tip diameter varying between 0.4 mm and 0.5 mm.3 While manual probes are reasonably easy to use and inexpensive, their measurements can vary greatly due to differences in probing pressures. Electronic probes, on the other hand, have high reproducibility because of their tightly controlled probing pressure. This results in more consistent and reliable readings. These devices, however, are expensive and require additional training for proper use.4,5 A specialized manual probe, such as the Naber’s probe, can also be used to detect and assess the extent of furcation invasion caused by periodontitis.
In addition to the need for reproducible techniques for measuring probing depths, measurement landmarks may change. For example, the gingival margin may change its position relative to other anatomy as a result of edema or recession. For this reason, some experts recommend measuring probing depths from the CEJ, but this approach introduces other variables and is rarely performed. A tooth-fitted stent may be used to facilitate a reproducible probing depth technique over time.6,7 To make a stent, an impression is taken, a dental model is poured, and a tooth-fitted acrylic stent is fabricated. The stent is then trimmed apically and coronally to the height of contour of the facial and lingual tooth surfaces. Grooves are cut in the stent to guide the periodontal probe into place at the sites to be measured. The stent is then polished and final adjustments are made to ensure its complete seating. Vertical landmarks are marked on the stent from which pocket depths are then measured over time. Stents can provide relatively stable vertical landmarks while also guiding the probe into the pocket using the same angle. Thus, errors resulting from differences in inclination or angulation over time can be reduced.8 Clinicians need to be aware, however, that stents typically fail to fully seat onto the teeth over time, thereby introducing the potential for overestimating attachment loss.
Intra- and extraoral radiographs are used to assess the apical-coronal extent of alveolar bone present, and they may serve as a baseline record for future comparisons aimed at detecting longitudinal changes in bone dimensions and densities. Bitewing and periapical radiographs are most commonly used. They are generally easy to acquire, inexpensive, and provide reasonable resolutions.9 Optimal bitewing radiographs are achieved with horizontal angulation perpendicular to the tooth surfaces and vertical angulation above or below perpendicular.10 Vertical bitewings using #2-size films are recommended in cases where bone loss > 4 mm in order to properly capture bone heights.5 A decision to use vertical bitewings is often made after a complete record of probing or visible attachment loss has been conducted. Vertical bitewings usually fail to show the apices of most teeth but are useful for assessing the presence and extent of bone losses typical of mild to severe periodontitis.11 A full-mouth series of radiographs is appropriate for a patient who may have chronic periodontitis to help detect any periapical pathoses that could produce localized drainage tracts, which can mimic chronic periodontitis. However, a full-mouth series is time consuming and periapical radiographs tend to be more geometrically distorted than bitewings.12
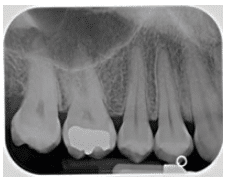
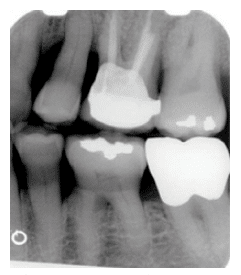
FIGURE 2B. Moderate horizontal bone loss can be seen in this bitewing of the right molar-premolar region.
Digital intraoral radiography may enhance the imaging of intraoral structures, while also reducing radiation exposure.13 Two-dimensional (2D) intraoral radiography, however, is difficult to standardize geometrically.14,15 There may be superimposition and distortion of relevant anatomical structures and underestimation of the size and presence of bony defects.13,16,17 Lingually located and funnel-shaped defects are difficult to detect in 2D radiographs.18 It is also challenging to differentiate the destruction of facial and/or lingual cortical bone plates, which can lead to the underdiagnosis of bone defect severity.19 An extraoral panoramic radiograph is another 2D image that can be used for viewing large areas. Unfortunately, this technique’s inherent distortions limit its diagnostic value for assessing subtle characteristics, such as alveolar bone levels.2
Cone-beam computed tomography (CBCT) provides a three-dimensional (3D) assessment of bony changes. It permits cross-sectional and 3D analyses of craniofacial structures. Research comparing the use of 3D volumetric images and 2D images in artificial bone defects has shown that CBCT has a sensitivity of 80% to 100% in the detection and classification of bone defects, while intraoral radiographs present a sensitivity of 63% to 67%.15,20 Vandenberghe et al20 also observed that the cross-sectional slices of CBCT data allowed for better assessment of periodontal bone levels.
The quality of images obtained by CBCT depends on acquisition parameters.21,22 To measure alveolar bone height, CBCT requires identification of the alveolar bone margin and the CEJ. The accuracy of identifying these intersections is limited by the size of each voxel in the image. Therefore, locating the CEJ on the CBCT image within the margin of error of a single voxel size should be possible. In contrast to the CEJ, the alveolar bone margin is the junction between cementum and bone. Here, the accuracy is limited by the physical spatial resolution of the image.23 To establish the bony changes characteristic of chronic periodontitis, images with better definition obtained by smaller voxel size are desirable, but this increases radiation exposure.24 Technical parameters should be balanced between the need for high image resolution and reducing radiation exposure. The presence of metallic artifacts in patients with metal fillings or other restorations also affects CBCT image quality.
Although CBCT may be theoretically superior to conventional intraoral radiography, it lacks widespread clinical applicability, at least in part because it remains unclear how CBCT use might favorably affect periodontal treatment decisions and outcomes.25,26
INTERPRETING RADIOGRAPHS
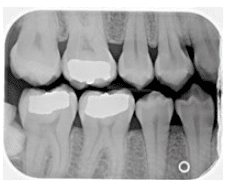
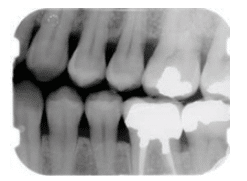
In radiographs, healthy alveolar bone presents as a thin line of what appears to be densely radiopaque bone that is continuous with the lamina dura of adjacent teeth. The lamina dura can be seen starting approximately 0.5 mm to 2.0 mm apical to tooth roots (Figure 1A and Figure 1B). Clinically, bone loss can be classified into horizontal and angular (vertical) defects. It runs parallel to an imaginary line joining CEJs of adjacent teeth, and can be further classified as mild, moderate, or severe (Figure 2A, Figure 2B, and Figure 2C).
Vertical or angular bone loss is characterized by nonuniform resorption of either facial or lingual cortical plate, and does not appear parallel to an imaginary line joining the CEJ of adjacent teeth. Initial signs of vertical defects may appear as abnormal widening of the PDL space at the alveolar crest, although this phenomena can be observed in mobile teeth around which no attachment loss has occurred.17,27 Bone destruction may lead to the formation of a radiographic image in which the apparent base of a defect is located well-apical to an alveolar crest.28,29 There are many classifications describing the morphology of vertical bone defects and the remaining osseous walls but the appearance of such defects on radiographs may have little to do with the actual connective tissue attachment levels. This is especially true when it comes to teeth that are mobile. Furcation involvements of multirooted teeth, recession, and other conditions, such as fenestrations and dehiscences, are included in the category of vertical bone defects.30 Table 1 and Table 2 describe the morphology of vertical and furcal bony defects.28–31
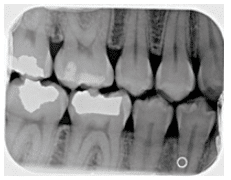
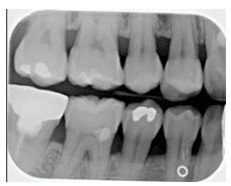
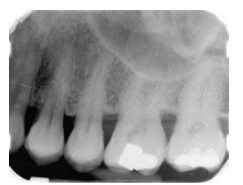
Vertical bony defects, especially three-wall and four-wall (circumferential) defects, may be difficult to detect on conventional radiographs because facial or lingual cortical plates remain superimposed over the defect. Clinical and surgical evaluation of these defects is the best mode of determining the number of remaining walls, as well as defect type. Two-wall defects, however, can sometimes be detected as what appears to be inconsistent bone margins (Figure 3A and Figure 3B). One-wall defects often present with a ramp-like appearance because the interdental septal bone slopes consistently between the adjacent tooth. (Figure 4). Grade 3 and grade 4 furcation involvements of multirooted teeth can sometimes be seen as radiolucencies between roots on conventional radiographs. Typically when such radiolucencies are detected, bone loss is significant. Conversely, lack of a radiolucency may mask furcal involvement because of superimposition of roots. This is because 2D radiographs are limited in their ability to assess anatomy that occurs in three dimensions.
Radiography is a key component of periodontal assessment and radiographs that are geometrically well-standardized over time can be highly useful when it comes to evaluating changes in alveolar bone. While new imaging techniques, such as CBCT, may eventually show benefits, 2D radiography remains an important modality in periodontal disease diagnosis and treatment planning.
References
- Langen HJ, Fuhrmann R, Diedrich P, Gunther RW. Diagnosis of infra-alveolar bony lesions in the dentate alveolar process with high-resolution computed tomography: experimental results. Invest Radiol. 1995;30:421–426.
- Akesson L, Hakansson J, Rohlin M. Comparison of panoramic and intraoral radiography and pocket probing for the measurement of the marginal bone level. J Clin Periodontol. 1992;19:326-32.
- Ramfjord SP. Indices for prevalence and incidence of periodontal disease. J Periodontol. 1959;30:51–59.
- Reddy MS, Palcanis KG, Geurs NC. A comparison of manual and controlled-force attachment-level measurements. J Clin Periodontol. 1997;24:920–926.
- Reddy MS. Radiographic methods in the evaluation of periodontal therapy. J Periodontol. 1992;63:1078–1084.
- Carlos JP, Brunelle JA, Wolfe MD. Attachment loss vs. pocket depth as indicators of periodontal disease: a methodologic note. J Periodontol Res. 1987;22:524–525.
- Reddy MS. The use of periodontal probes and radiographs in clinical trials of diagnostic tests. Ann Periodontol. 1997;2:113-22.
- Deas DE, Moritz AJ, McDonnell HT, Powell CA, Mealey BL. Osseous surgery for crown lengthening: a 6-month clinical study. J Periodontol. 2004;75:1288–294.
- de Faria Vasconcelos K, Evangelista KM, Rodrigues CD, Estrela C, de Sousa TO, Silva MA. Detection of periodontal bone loss using cone beam CT and intraoral radiography. Dentomaxillofac Radiol. 2012;41:64–69.
- van der Stelt PF, Ruttiman UE, Webber RL, Heemstra P. In vitro study into the influence of X-ray beam angulation on the detection of artificial caries defects on bitewing radiographs. Caries Res. 1989;23:334–341.
- de Toledo BE, Barroso EM, Martins AT, Zuza EP. Prevalence of periodontal bone loss in brazilian adolescents through interproximal radiography. Int J Dent. 2015;2012:357056.
- Tugnait A, Carmichael F. Use of radiographs in the diagnosis of periodontal disease. Dent Update. 2005;32:536–538, 541–542.
- Mol A, Balasundaram A. In vitro cone beam computed tomography imaging of periodontal bone. Dentomaxillofac Radiol. 2008;37:319–324.
- Jeffcoat MK. Current concepts in periodontal disease testing. J Am Dent Assoc. 1994;125:1071–1078.
- Misch KA, Yi ES, Sarment DP. Accuracy of cone beam computed tomography for periodontal defect measurements. J Periodontol. 2006;77:1261–1266.
- Eickholz P, Hausmann E. Accuracy of radiographic assessment of interproximal bone loss in intrabony defects using linear measurements. Eur J Oral Sci. 2000;108:70–73.
- Rethman M, Ruttiman U, O’Neal R, et al. Diagnosis of bone lesions by subtraction radiography. J Periodontol. 1985;56:324–329.
- Ramadan AB, Mitchell DF. A roentgenographic study of experimental bone destruction. Oral Surg Oral Med Oral Pathol. 1962;15:934–943.
- Rees TD, Biggs NL, Collings CK. Radiographic interpretation of periodontal osseous lesions. Oral Surg Oral Med Oral Pathol. 1971;32:141–153.
- Vandenberghe B, Jacobs R, Yang J. Diagnostic validity (or acuity) of 2D CCD versus 3D CBCT-images for assessing periodontal breakdown. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2007;104:395–401.
- Ballrick JW, Palomo JM, Ruch E, Amberman BD, Hans MG. Image distortion and spatial resolution of a commercially available cone-beam computed tomography machine. Am J Orthod Dentofacial Orthop. 2008;134:573–582.
- Ludlow JB, Davies-Ludlow LE, Brooks SL, Howerton WB. Dosimetry of 3 CBCT devices for oral and maxillofacial radiology: CB Mercuray, NewTom 3G and i-CAT. Dentomaxillofac Radiol. 2006;35:219–226.
- Leung CC, Palomo L, Griffith R, Hans MG. Accuracy and reliability of cone-beam computed tomography for measuring alveolar bone height and detecting bony dehiscences and fenestrations. Am J Orthod Dentofacial Orthop. 2010;137:S109–S119.
- Kamburo?lu K, Ere? G, Akgün C, Yeta EN, Gülen O, Karacao?lu F. Effect of voxel size on accuracy of cone beam computed tomography–aided assessment of periodontal furcation involvement. Oral Surg Oral Med Oral Pathol Oral Radiol. 2015;120:644–650.
- Songa VM, Jampani ND, Babu V, Buggapati L, Mittapally S. Accuracy of cone-beam computed tomography in diagnosis and treatment planning of periodontal bone defects: a case report. J Clin Diagn Res. 2014;8:ZD23–ZD25.
- Bagis N, Kolsuz ME, Kursun S, Orhan K. Comparison of intraoral radiography and cone-beam computed tomography for the detection of periodontal defects: an in vitro study. BMC Oral Health. 2015;15:64.
- Pilhstrom B, Anderson K, Aeppli D, Schaffer E. Association between signs of trauma from occlusion and periodontitis. J Periodontol. 1986;57:1–7.
- Goldman HM, Cohen DW. The infrabony pocket: classification and treatment. J Periodontol. 1958;29:272–276.
- Prichard JF. The etiology, diagnosis and treatment of the intrabony defect. J Periodontol. 1967;38:455–465.
- Pepelassi EA, Tsiklakis K, Diamanti-Kipioti A. Radiographic detection and assessment of the periodontal endosseous defects. J Clin Periodontol. 2000;27:224-30.
- Karn KW, Shockett HP, Moffitt WC, Gray JL. Topographic classification of deformities of the alveolar process. J Periodontol. 1984;55:336–340.
From Dimensions of Dental Hygiene. January 2016;14(01):42–44,47.