
Promoting Oral Health Among the Visually Impaired
Dental professionals need to be prepared to safely and effectively treat patients with age-related macular degeneration.
This course was published in the January 2016 issue and expires January 31,2019. The authors have no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Identify the two types of age-related macular degeneration (AMD).
- Discuss the risk factors associated with AMD.
- Recognize the barriers to dental treatment that exist for patients who are visually impaired.
- Explain the treatment modifications that can be implemented for patients who are visually impaired.
Age-related macular degeneration (AMD) is a leading cause of low vision and blindness among adults 50 and older. Currently, more than 2 million Americans experience some degree of vision loss due to AMD. It is projected that by 2030 nearly 3.5 million Americans will have advanced AMD.1 While the prevalence of AMD is lower than other eye diseases such as cataracts and glaucoma, as the United States population continues to age, dental professionals will likely encounter patients with AMD in the clinical setting. Visual impairment can impact oral health in many ways. As such, clinicians need to be knowledgeable about this disease so treatment can be modified to meet the needs of this population.
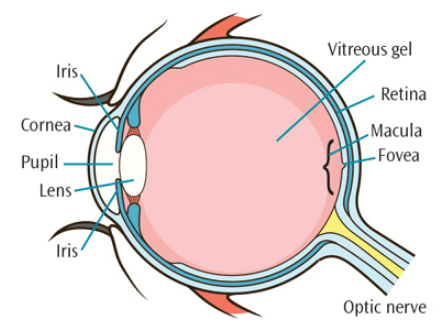
AMD is a painless condition characterized by deterioration of the macula— the central part of the retina responsible for sharp central vision. In the beginning, patients diagnosed with AMD may be completely asymptomatic. As the disease progresses, however, central vision is compromised and patients become more sensitive to glare, lose color vision, and become less able to distinguish contrasting colors.1 The retina, located in the back of the eye, is composed of light-sensitive tissue that records the images seen by the eye and sends them to the brain via the optic nerve (Figure 1). The macula, located in the center of the retina, is the most sensitive part of the retina and is responsible for central vision, allowing the eye to see straight ahead and in fine detail.
When AMD damages the macula, a blurred, distorted, or dark area near the center of vision occurs. As the disease progresses, the blurred area can grow larger and blank spots appear in the central vision.2 Central vision impairment means that activities of daily living, such as reading, driving, and shopping, are significantly impaired.3,4 Daien et al5 observed a strong relationship between moderate and severe visual impairment and limited ability to engage in instrumental activities of daily living. The study also revealed that mild vision problems resulted in an increased risk of activity limitations. These results suggest the need for early detection and intervention by health care professionals before a decline in autonomy occurs.5
CLASSIFICATION
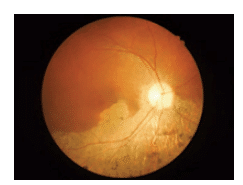
AMD is classified into two types: wet and dry. Dry or atrophic AMD is more common, occurring in approximately 85% to 90% of cases, and typically results in a more gradual loss of vision compared to wet AMD.2 Dry AMD has three stages: early, intermediate, and advanced, which can occur in one or both eyes. Patients with early AMD have small or medium-sized deposits under the retina, called drusen (Figure 2). Considered cellular waste products, retinal drusen appear as yellowish deposits on the surface of the retina. In the early stage, ophthalmologists may detect several small or medium drusen under the retina in one or both eyes. Typically, patients in the early stage of AMD do not experience any vision loss.1
As dry AMD progresses to the intermediate stage, medium or large drusen appear under the retina and patients may begin to notice changes in their central vision. Reading or other tasks that require fine detail may necessitate more light. Advanced dry AMD is classified by further breakdown of the macula and large drusen appearing under the retina. This breakdown causes blurred spots in the central vision that become larger and darker over time (Figure 3). Dry AMD can progress and produce greater vision loss without turning into the wet form. However, dry AMD can quickly transform into wet AMD when new blood vessels grow in the eye.2
The wet or exudative form of AMD is considered more severe than the dry form. It is less common, occurring in only about 10% to 15% of AMD cases.1 The wet form is also referred to as neovascular AMD and occurs when there is new growth of abnormal blood vessels in the macular portion of the retina. These blood vessels are very delicate and can leak blood and fluid near the macula, causing it to shift from its place in the back of the eye. The movement of the macula interferes with the function of the retina and causes blurring of the central vision. Unlike the dry form of AMD, the wet form does not have stages. All wet AMD cases are considered severe. Presently, there is no definite way to predict when or if the dry form will progress to the neovascular form of AMD.
There is no cure for AMD. As such, individuals with AMD must remain under the care of an ophthalmologist or retinal specialist to monitor disease progression.2
RISK FACTORS
Several risk factors are associated with AMD, including age, race, gender, family history, diet, smoking, and the presence of certain systemic diseases. While the exact cause of AMD is unknown, age is a key risk factor. The likelihood of developing AMD increases exponentially with age. It is estimated that nearly 25% of individuals age 65 to 74 will develop AMD, and almost one in three individuals age 75 and older has AMD.2 Macular degeneration is seen more often in whites than individuals of other races. In 2010, according to the National Eye Institute, 2.5% of whites age 50 and older had AMD, whereas only 0.9% of African-Americans, Hispanics, and individuals of other races had the disease.1 Incidence and prevalence of AMD varies across populations, but several large population-based studies have confirmed that non-Hispanic whites have a higher prevalence of both wet and dry AMD than Hispanics and non-Hispanic blacks. Researchers have speculated several reasons for this, including melanin pigmentation acting as a protective mechanism for the retina and distribution of protective and harmful genes among different racial groups.6

Women are more likely to develop AMD than men. According to Zetterberg,7 the decline of estrogen after menopause contributes to this increased risk. Chronic low-grade inflammation is a hallmark of AMD. When estrogen levels drop after menopause, the anti-inflammatory properties that estrogen provides are diminished, putting women at greater risk for AMD.7
Family history of macular degeneration, especially among parents and siblings, is also a risk factor. Those who have a family member with the disease are more likely to develop the condition at a younger age than an individual without an affected parent or sibling.8 It has been suggested that certain genes or genetic variants within families can greatly increase the risk of developing AMD. There is little understanding about the clinical differences connecting patients with and without a family history of AMD, however, and research remains ongoing.
A diet that contains few nutrient-rich fruits and vegetables—such as kale, spinach, and broccoli—is also a risk factor for AMD. The 2001 Age-Related Eye Disease Study (AREDS) conducted by the National Eye Institute found that patients with intermediate or advanced AMD in one eye but not the other lowered their risk of developing the disease in the unaffected eye by approximately 25% when consuming a high-dose mixture of vitamin C, vitamin E, beta-carotene, and zinc. Eating a diet rich in green, leafy vegetables may reduce the risk of AMD.9 Studies suggest that consuming increased levels of dietary carotenoids, omega-3 fatty acids, and vitamin B may help slow or prevent the progression of AMD. However, a consensus has not been reached as to what levels of dietary supplements should be consumed on a consistent basis.10
Smoking is a major risk factor associated with AMD. The Beaver Dam Eye Study—a longitudinal population-based study on the relationship between smoking and incidence of AMD over a 20-year period—revealed that AMD risk was directly associated with the number of packs of cigarettes smoked and the number of years patients used tobacco.11 In addition, results showed that smoking increased the likelihood individuals with AMD would progress to a more severe form. A study released in 2014 by Erke et al,12 concluded that age and smoking were both strong risk factors for AMD in women and men. The study also found that a significant relationship exists between current and former smokers and late-stage AMD.12
Some systemic diseases are also considered risk factors for AMD, including hypertension, coronary heart disease, dementia, dyslipidemia, and kidney disease. A literature review performed by Cheung and Wong13 suggests that AMD is the result of systemic disease and that the disease shares many of the same risk factors as hypertension, cardiovascular disease, dyslipidemia, and cerebrovascular disease, among others. The authors concluded that individuals with systemic disease, such as hypertension, are at increased risk of developing not only AMD, but also cardiovascular diseases, including stroke.13
Recently, researchers have begun exploring the relationship between AMD and thyroid disease, specifically hypothyroidism. The diseases possess some of the same risk factors, including age and gender. Diseases connected to hypothyroidism have also been linked to AMD, including hypertension and cardiovascular disease.13,14 Research on the possible link between AMD and hypothyroidism and associated risk factors is ongoing. Further evaluation of the link between AMD and systemic diseases is needed to understand the underlying relationships and create new opportunities for treatment.
DISABILITY
The connection between systemic diseases, such as diabetes and cardiovascular disease, and oral health has been widely studied. While conditions like AMD do not have a direct impact on oral health, they may predispose patients to poor oral health due to physical, social, and emotional barriers that patients with visual impairments experience. Research suggests patients with any stage of AMD are more likely to experience impairment of basic or instrumental activities of daily living than individuals with adequate vision.15 Patients who lose the ability to drive themselves to appointments, use the telephone, manage money, and maintain medication regimens may experience reduced access to dental care.
Educating patients with AMD and their family members/caregivers about the importance of maintaining optimal oral health is crucial. Regular dental visits are critical to oral health maintenance. A thorough exam and prophylaxis should be performed at regular intervals. Patients who are visually impaired may be able to adequately preserve their oral health if they are appropriately educated and motivated.16 Transportation and cost are major barriers to dental care access, but individuals may be more motivated to overcome these barriers if they are properly educated about their oral health and how it relates to their quality of life.
Older adults with visual impairment are more likely to experience depression, which can impact the provision of oral health care services.17 Patients with AMD who are depressed may be inattentive and unmotivated to follow through with appointments and recommendations.3 Clinicians should regularly inquire about medication usage, changes in overall health, and patients’ current level of support. Assessing patients’ social support systems can help dental hygienists better understand their home environment. For example, if the dental hygienist knows the patient lives with a spouse or child, including that family member in the discussion about recare interval and self-care procedures can help with compliance. Generally, a strong social support network acts as a buffer against the risk of depression that can accompany vision loss. Evidence suggests that low levels of social support correlate to an increased risk of depression among those with vision problems.17
TREATMENT CONSIDERATIONS
AMD can create physical, emotional, and social barriers to maintaining oral health among older adults. Research has shown that individuals with a disability—regardless of type—usually have poor oral health, including periodontal diseases, dental caries, and tooth loss.18,19 In order to provide the most effective care, the dental team should be prepared to manage patients with impaired vision in the office environment and understand the emotional and psychological issues that often impact dental care and oral health.
The American with Disabilities Act requires any public place or commercial facility to meet the standards of accessibility by removing structural, communication, and transportation barriers.20 Therefore, when a patient with impaired vision is scheduled for an office visit, the dental team must consider how to best accommodate the individual within the office setting. Implementing the office procedures designed for treating blind patients, with some minor adjustments, can make a dental visit much more feasible for those with low vision.
Modifications for the dental team to consider when treating visually impaired patients include: speaking clearly while facing the patient; creating medical/dental history forms, brochures, and other printed materials in a large, bold print; and offering to assist the patient when filling out forms. In addition, providing ample lighting in walkways and operatories, escorting the patient from the reception area to the dental treatment chair, and, if necessary, placing the patient’s hand on the back of the chair to help guide him or her into the chair safely are helpful.21 Pathways should be free of loose rugs, hoses, cords, etc, and any change in the elevation of the floor or flooring texture (eg, hardwood to carpet) should be communicated to the patient.
Patients with AMD may be sensitive to bright light, so care should be taken to avoid shining the dental light into the their eyes. Tinted glasses should be offered if patients do not have their own. Depending on patients’ vision levels, using the “tell, show, do” method may be beneficial to make them feel comfortable and safe. Avoid startling patients by warning ahead of time about any applications of suction, air, water, or power instruments.21
Dental hygienists can recommend oral hygiene aids according to the patient’s degree of visual impairment. These may include power toothbrushes, double-headed toothbrushes, denture brushes with suction cups to attach to the sink surface, oral irrigators, and wall-mounted holders for toothbrushes and paste for easy access.16 If the patient has difficulty remembering the suggested self-care routine, the dental hygienist can print directions in large, plain bold print to hang in the bathroom. The dental team needs to be aware that treating patients with impaired vision requires some additional planning and effort but is instrumental in helping these individuals maintain their functional independence.
Dental management of patients with AMD should focus on the fact that optimal oral health plays an integral role in overall health. It also has a direct impact on oral function, appearance, and social relationships.16 Dental hygienists should modify the self-care routine to enable patients with low vision to be as independent as possible and achieve good oral hygiene. The successful self-care routine should include:
- Intervention and care during the early stages of disease
- Provision of education on preventive measures and oral health aids to foster success and independence
- Scheduling of regular recare appointments to review the patient’s needs and minimize the need for further intervention
- Reduction of patient fear, stress, and embarrassment through a caring and supportive environment16
CONCLUSION
AMD is a leading cause of low vision among individuals older than 50. More than 2 million Americans have some degree of vision loss caused by AMD.1 As a result, the dental team will likely encounter this patient population. Dental hygienists need to be knowledgeable about how best to manage patients with AMD within the office setting and beyond. Clinicians must remember that visually impaired patients may be unable to examine their oral cavities for signs of gingivitis, decay, food debris, and biofilm. Many oral conditions, such as periodontal diseases and dental caries, are asymptomatic and relatively painless and, thus, may go unnoticed by patients until the disease has advanced. By understanding the physical and emotional problems that occur in patients with low vision, dental hygienists can help this population maintain their oral health, independence, and quality of life.
References
- National Institutes of Health National Eye Institute. Facts about Age-Related Macular Degeneration. Available at: nei.nih.gov /health/maculardegen/armd_facts. Accessed December 23, 2015.
- American Macular Degeneration Foundation. What Is Macular Degeneration? Available at: macular.org/what-macular-degeneration.Accessed December 23, 2015.
- Wilkins EM. Clinical Practice of the Dental Hygienist. 11th ed. Philadelphia: Lippincott, Williams, & Wilkins; 2013.
- Activities of Daily Living: What are ADLs and IADLs? Available at: caring.com/articles/activities-of-daily-living-what-are-adls-and-iadls. Accessed December 23, 2015.
- Daien V, Peres K, Villain M, Colvez A, Carriere I, Delcourt C. Visual acuity thresholds associated with activity limitations in the elderly. The Pathologies Oculaires Liées à l’Age study. Acta Ophthalmol. 2014;92:e500–506.
- Klein R. Race/ethnicity and age-related macular degeneration. Am J Opthamol. 2011;152:153–154.
- Zetterberg M. Age-related eye disease and gender. Maturitas. 2016;83:19–26.
- Saksens NT, Kersten E, Groenewoud JM, et al. Clinical characteristics of familial and sporadic age-related macular degeneration: differences and similarities. Invest Opthalmol Vis Sci. 2014;55:7085–7092.
- Age-Related Eye Disease Study Research Group. A randomized, placebo-controlled, clinical trial of high-dose supplementation with vitamins C and E, beta carotene, and zinc for age-related macular degeneration and vision loss: AREDS Report No. 8. Arch Ophthalmol. 2001;119:1417–1136.
- Sin HP, Liu DT, Lam DS. Lifestyle modification, nutritional and vitamins supplements for age–related macular degeneration. Acta Ophthalmol. 2013;91:6–11.
- Myers CE, Klein BE, Gangnon R, Sivakumaran TA, Iyengar SK, Klein R. Cigarette smoking and the natural history of age-related macular degeneration—The Beaver Dam Eye Study. Ophthalmology. 2014;121:949–1955.
- Erke MG, Bertelsen G, Peto T, Sjølie AK, Lindekleiv H, Njølstad I.Cardiovascular risk factors associated with age-related macular degeneration: the Tromsø Study. Acta Ophthalmologica. 2014;92:662–669.
- Cheung CMG, Wong,TY. Is age-related macular degeneration a manifestation of systemic disease? New prospects for early intervention and treatment. J Intern Med. 2014;276:140–153.
- Bromfield S, Keenan J, Jolly P, McGwin G Jr. A suggested association between hypothyroidism and age-related macular degeneration. Curr Eye Res. 2012;37:549–552.
- Gopinath B, Liew G, Burlutsky G, Mitchell P. Age-related macular degeneration and 5-year incidence of impaired activities of daily living. Maturitas. 2014;77:263-–66.
- Schembri A, Fiske J. The implications of visual impairment in an elderly population in recognizing oral disease and maintaining oral health. Spec Care Dentist. 2001;21:222–226.
- Papadopoulos K, Papakonstantinou D, Montgomery A, Solomou A. Social support and depression of adults with visual impairments. Res Dev Disabil. 2014;35:1734–1741.
- Azrina AN, Norzuliza G, Saub R. Oral hygiene practices among the visually impaired adolescents. Annal Dent Univ Malaya. 2007;14:1–6.
- Chavez E, Ship J. Sensory and motor deficits in the elderly: impact on oral health. J Public Health Dent. 2000;602:97–303.
- United States Department of Justice. Americans with Disabilities Act. Available at: ada.gov. Accessed December 23, 2015.
- Jaccarino J. Vision impairment: treating the special needs patient with a sensory disability. Dent Assist. 2009;78:8–10,30,32–33.
From Dimensions of Dental Hygiene. January 2016;14(01):38–41.



