 RENDEEP/ISTOCK/GETTY IMAGES PLUS
RENDEEP/ISTOCK/GETTY IMAGES PLUS
Air Polishing as an Adjunctive Therapy
Subgingival air polishing can serve as an adjunctive method for biofilm removal in periodontal maintenance therapy.
This course was published in the March 2019 issue and expires March 2022. The authors have no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Describe appropriate technique for subgingival air polishing.
- Explain the properties, benefits, and risks of the more commonly used air polishing powders.
- Identify the benefits and risks of subgingival air polishing.
Air polishing was originally introduced in 1945 as a cavity preparation method that used highly abrasive particles.1 Since the 1980s, supragingival air polishing has been used to remove plaque and stain. Significant advancements in air polishing powders, device nozzles, and equipment have enabled the creation of subgingival air polishing procedures.2 Subgingival air polishing can serve as an alternative or adjunctive method for biofilm management in periodontal maintenance therapy.2,3
EQUIPMENT
Air polishing devices are commonly used to remove supragingival biofilm and, with advances in device design and powders, they are now able to remove biofilm subgingivally.2 The devices operate by creating and spraying a slurry that consists of powder, air, and water.2 The air polishing device can be a stand-alone unit with a cylindrical handpiece or a larger handpiece with a powder chamber that connects directly to a dental unit.2 The handpieces come with two types of nozzles: one for supragingival biofilm removal and one for subgingival biofilm removal.
The typical nozzle has a round opening made of two concentric circles.2 The outer circle is where water flows from the device and the inner circle is where air and powder exit from the device.2 There are a couple of different nozzles that can be used for periodontal pockets. One nozzle is made from a flexible thermoplastic elastomer with several small holes.3 Water exits the hole in the tip of the nozzle and the air and powder flow from the small holes on the lateral sides of the nozzle.2,4 Compared with a standard system, the periodontal nozzle devices use lower air pressure that is more suitable for subgingival debridement.3,5 Depending on the powder used, specific equipment may be needed.
SUBGINGIVAL TECHNIQUE
There are two different techniques used for subgingival air polishing procedures. The first uses a supragingival adaptation of the standard device nozzle in which it is aimed toward the tissue for subgingival polishing.2 The nozzle is held nearly parallel to the root surface at a distance of 4 mm to 6 mm and activated in a constant sweeping motion.2 A single tooth surface is polished for 5 seconds to 10 seconds, for a total of 20 seconds to 40 seconds per tooth.2 Several studies have evaluated the subgingival polishing technique in different pocket depths, some up to 5 mm.2-4,6
The second technique uses a single-use wedge-shaped periodontal nozzle that can be inserted into periodontal pockets with probing depths between 4 mm and 9 mm.2 The periodontal nozzle is inserted directly into the pocket until it meets the resistance of the junctional epithelium and then is withdrawn slightly before activating.4 The nozzle is moved in a circular motion as each tooth surface (distal, buccal, mesial, and lingual) is treated for approximately 5 seconds.4 With both nozzles, the air-water pressure can be regulated based on the intended use.2
POLISHING POWDERS
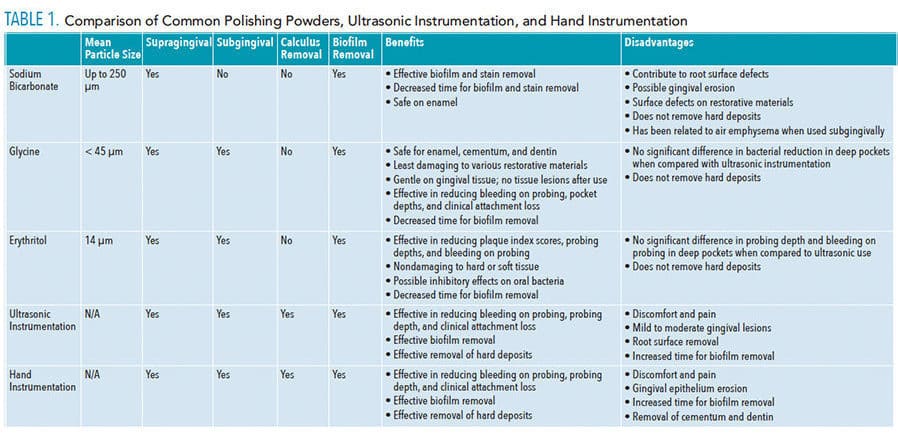
Several polishing powders are available. Sodium bicarbonate, glycine, and erythritol are some of the more commonly used powders. Released in 2018, erythritol is new to the United States, but it has been used in Europe for several years. Other, less commonly used powders include calcium carbonate, aluminum trihydroxide, and calcium sodium phosphosilicate.7 The following section will discuss the properties, benefits, and risks of the more commonly used air polishing powders. See Table 1 for a summary of the properties of a variety of air polishing powders and instrumentation procedures.
Sodium bicarbonate. This nontoxic and water-soluble powder is often used for the removal of supragingival biofilm.2 The average particle size varies up to 250 µm.2 Air polishing with sodium bicarbonate powder is effective in biofilm and stain removal.7,8 Bühler et al8 demonstrated that air polishing with sodium bicarbonate for biofilm removal resulted in less time instrumenting each tooth surface compared to the use of hand curets. Overall, research suggests that sodium bicarbonate powder may cause damage to the hard and soft periodontal tissues when used subgingivally.7,8 The use of sodium bicarbonate for air polishing has been considered safe for enamel but can contribute to root surface defects.7 The results of an in vitro study by Herr et al9 indicated that sodium bicarbonate powder created more damage to the cementum and exposed more dentinal tubules compared to glycine powders. Additionally, there is some indication that sodium bicarbonate powder directed at the gingiva with an air polishing device results in substantial gingival erosion.7 Simon et al10 concluded that air polishing with sodium bicarbonate powder resulted in erosion of gingival epithelium with areas where connective tissue was exposed. Finally, significant surface defects can result from the use of air polishing with sodium bicarbonate on various restorative materials, but it has been considered safe for amalgam restorations.7 Sodium bicarbonate is not suitable for subgingival air polishing. However, its properties are discussed here because it is a common polishing powder used for supragingival biofilm removal and it has been used as a comparison in the research and development of other powders including those used for subgingival air polishing.
Glycine powder. This amino-acid salt has a small particle size (average size is less than 45 µm) in comparison to sodium bicarbonate powder.2,11 The use of glycine powder air polishing is considered safe for enamel, cementum, and dentin.3,7 Also, compared with other powders, glycine powder results in the least amount of damage to restorative materials, such as amalgam, glass ionomer, composite, and porcelain.3,7 According to a systematic review by Bühler et al8 on the effects of air polishing on hard and soft oral tissues, a moderate level of evidence demonstrates that glycine powder causes potentially less damage to periodontal tissues.
When compared to air polishing with sodium bicarbonate, ultrasonic instrumentation, or hand instrumentation, the literature suggests the use of glycine powder air polishing is gentler on the gingival tissue, dentin, and root surfaces.7,8 Wenström et al12 concluded that glycine powder air polishing was comparable to ultrasonic instrumentation in regard to improvement in clinical indices, such as reduced bleeding on probing, probing depths, and improved average clinical attachment levels. Even though subgingival biofilm may be effectively removed with glycine powder air polishing in moderate to deep periodontal pockets, there are no significant differences in bacterial reduction when compared to ultrasonic use.3,12 In a study of 10 participants, subgingival glycine powder air polishing removed biofilm and was gentle on the tissue.10 Researchers performed biopsies of the gingival tissue to evaluate the histopathologic effects of the powders and the ultrasonic. The biopsy revealed glycine powder left no lesions, compared to mild-moderate lesions where ultrasonic scaling was performed.10
An overall beneficial change in the oral bacteria can occur as a result of air polishing subgingival sites and the oral mucosa (labial, palatal, buccal, and tongue).4 Flemmig et al4 found that after 90 days the amount of the periodontal pathogen Porphyromonas gingivalis was reduced in shallow pockets and throughout the mouth with full mouth glycine powder air polishing compared to traditional periodontal maintenance therapy.
A particular powder may be combined with other chemicals to enhance performance.2 One formulation of glycine powder includes tricalcium phosphate.9 In a study by Herr et al,9 the use of glycine powder with tricalcium phosphate resulted in less exposed dentinal tubules, compared to the use of glycine alone. The exposure of dentinal tubules can increase root hypersensitivity to substances such as hot and cold liquids.13 However, these data are based on root surfaces below the gum tissue that have limited exposure to surface stimuli.9
Erythritol powder. This chemically neutral, water-soluble sugar alcohol has the smallest particle size (average 14 µm), but the level of abrasiveness is not significantly lower than glycine.14,15 The use of erythritol powder is considered safe in subgingival areas.3 The results of a randomized controlled trial revealed that significant improvements occurred in plaque index scores and bleeding on probing with the subgingival use of erythritol powder air polishing.15 The authors noted that after 6 months, significant plaque removal was demonstrated by subgingival air polishing with erythritol powder and was comparable to plaque removal by hand instrumentation.15 However, there was no significant difference in air polishing outcomes between the use of erythritol and glycine powder.15 Müller et al14 found no visible damage to hard or soft tissue after using erythritol powder. In comparison to sodium bicarbonate and glycine powders, erythritol produced the smoothest surface when used on dentin.14 Over 12 months, polishing with erythritol powder every 3 months resulted in significant reductions in probing depth and bleeding on probing.14 However, there was not a significant difference in treatment outcomes between erythritol powder and the use of ultrasonic instrumentation.14 Erythritol (in lozenge form) may also reduce dental plaque accumulation, inhibit the growth and formation of Streptococcus mutans, and reduce the number of caries lesions.16
Other powders. Several other air polishing powders have been introduced over the years, but have not been used extensively due to a variety of factors. There is limited research on the use of calcium carbonate, aluminum trihydroxide, and calcium sodium phosphosilicate in regard to their effects on clinical indices such as probing depth, bleeding on probing, and clinical attachment levels. However, multiple studies indicate the use of calcium carbonate, aluminum trihydroxide, and calcium sodium phosphosilicate powders can result in enamel and root defects, as well as significant defects in a variety of restorative materials.7,17–19 In addition, aluminum trioxide and calcium carbonate demonstrate poor water solubility, which limited their subgingival use.2
BENEFITS
Subgingival air polishing is less aggressive than conventional methods of biofilm removal (Table 1). Some benefits of subgingival air polishing are a reduction in damage to hard and soft tissue, short-term reduction of bacterial load, pocket reduction, and patient acceptance.3 Subgingival biofilm can be effectively removed with air polishing devices.6 In shallow to moderately deep pockets, the use of subgingival air polishing may also be more effective in biofilm removal than curets.6,7 A systematic review determined that, compared to conventional treatments, subgingival air polishing with a low abrasive powder (glycine or erythritol) showed minimal root surface removal and intact gingival epithelium.3
Pocket reduction and patient acceptance are also possible benefits of subgingival air polishing. During supportive periodontal therapy, a significant reduction in the average number of pockets greater than 4 mm was found after several subgingival air polishing treatments.14 Compared to ultrasonic scalers, the results of probing depth reduction were similar for both subgingival air polishing and ultrasonic scaling.3,14 However, another study reported deeper probing depths at 1-month, 3-month, and 6-month intervals for areas treated with subgingival glycine powder air polishing when compared to the outcomes of hand and ultrasonic instrumentation.5
Following conventional periodontal maintenance treatment methods—such as plaque and biofilm removal—patients frequently experience root sensitivity or tissue trauma.3,20 However, air polishing reduces iatrogenic damage to hard and soft tissue resulting in improved patient comfort.3,20,21 Patients may prefer subgingival air polishing compared to ultrasonic or hand instrumentation, because it is more comfortable and takes less time to complete.3,12,21,22 Plaque removal can be completed within 5 seconds to 10 seconds per root surface (approximately 10 minutes to 20 minutes for full dentition), where conventional treatment options are more time-consuming.3 The procedure of subgingival air polishing resulted in less pain for patients, no pressure, and was the preferred treatment for subsequent visits.5 Additional benefits of subgingival air polishing may include reducing the need for local anesthetics and decreasing the risk of increased sensitivity as a result of exposure of dentinal tubules.3
RISKS
Aside from previously discussed side effects specific to each powder in subgingival air polishing, there is a slight risk for general side effects such as air emphysema. Air emphysema is a complication of oral trauma that can result from the use of compressed air. Several studies report that there were no serious adverse medical reactions or side effects from subgingival air polishing.5,6,10,12,14,15,22 The level of risk for air emphysema is extremely rare.7 In a small case report by Alonso et al,23 the authors noted three cases of subcutaneous emphysema that occurred during polishing in three different patients over 2 years at a single dental clinic. The reported cases resolved within 3 days to 5 days. The safety of subgingival air polishing depends on the use of the proper equipment and technique.6,7 However, caution is warranted as the reporting of such cases is voluntary and it is likely that the incidence is under-reported.
PATIENT CONSIDERATIONS
Appropriate patient selection is key to assuring safety and efficacy of subgingival air polishing. Subgingival air polishing does not remove calculus, and traditional calculus-removal methods need to be used in patients with hard deposits.3,6,7,11,24 Many of the studies reported here utilized subgingival air polishing when patients were obtaining supportive periodontal therapy and not during initial periodontal treatment.3,4,12,14 Air polishing is considered an adjunctive therapy to traditional debridement methods, but has been shown effective as a monotherapy in patients undergoing supportive periodontal therapy.3
Patient perceptions are an important factor when considering compliance. Compared to ultrasonic or hand instrumentation, minimal discomfort was noted for the use of glycine powder and erythritol powder air polishing.7,8,11 Several studies noted subgingival air polishing produced only minor discomfort, which was significantly less painful than ultrasonic scaling and hand instrumentation.6–8,14,24
CONCLUSION
Subgingival air polishing is a safe and effective adjunctive method of biofilm management when proper technique and powders are used. While there are benefits to using other air polishing powders, the use of glycine and erythritol powders have demonstrated consistent results with less potential damage occurring to the gingiva, oral mucosa, cementum, dentin, and various restorative materials. Compared to conventional biofilm removal methods, the air polishing procedure seems to be a more comfortable treatment option for patients undergoing periodontal maintenance therapy.
The subgingival air polishing procedures for shallow (up to 4 mm) and deep (5 mm to 9 mm) pockets are equivalent or superior to hand and ultrasonic instrumentation in regard to biofilm removal. The procedures are often preferred by patients and can produce clinically favorable results. Additional research with varying device nozzles and powders may provide additional evidence on safety, efficacy, and treatment outcomes and recommendations.
REFERENCES
- Black R. Technic for nonmechanical preparation of cavities and prophylaxis. J Am Dent Assoc. 1945;32:955–965.
- Petersilka GJ. Subgingival air-polishing in the treatment of periodontal biofilm infections. Periodontol 2000. 2011;55:124-142.
- Ng E, Byun R, Spahr A, Divnic-Resnik T. The efficacy of air polishing devices in supportive periodontal therapy: a systematic review and meta-analysis. Quintessence Int. 2018;49:453–467.
- Flemmig TF, Arushanov D, Daubert D, Rothen M, Mueller G, Leroux BG. Randomized controlled trial assessing efficacy and safety of glycine powder air polishing in moderate-to-deep periodontal pockets. J Periodontol. 2012;83:444–452.
- Kargas K, Tsalikis L, Sakellari, D, Menexes G, Konstantinidis A. Pilot study on the clinical and microbiological effect of subgingival glycine powder air polishing using a cannula-like jet. Int J Dent Hyg. 2015;13:161–169.
- Sculean A, Bastendorf KD, Becker C, Bush B, et al. A paradigm shift in mechanical biofilm management? Subgingival air polishing: a new way to improve mechanical biofilm management in the dental practice. Quintessence Int. 2013;44:475–477.
- Cobb CM, Daubert DM, Davis K, et al. Consensus conference findings on supragingival and subgingival air polishing. Compend Contin Educ Dent. 2017;38:e1–e4.
- Bühler J, Amato M, Weiger R, Walter C. A systematic review on the effects of air polishing devices on oral tissues. Int J Dent Hyg. 2016;14:15–28.
- Herr ML, DeLong R, Li Y, Lunos SA, Stoltenberg JL. Use of a continual sweep motion to compare air polishing devices, powders, and exposure time on unexposed root cementum. Odontology. 2017;105:311–319.
- Simon CJ, Munivenkatappa Lakshmaiah Venkatesh P, Chickanna R. Efficacy of glycine powder air polishing in comparison with sodium bicarbonate air polishing and ultrasonic scaling – a double-blind clinico-histopathologic study. Int J Dent Hyg. 2015;13:177–183.
- Aimetti M. Nonsurgical periodontal treatment. Quintessence Int. 2014:9:251–267.
- Wenström JL, Dahlén G, Ramberg P. Subgingival debridement of periodontal pockets by air polishing in comparison with ultrasonic instrumentation during maintenance therapy. J Clin Periodontol. 2011;38:820–827.
- Kim JJ. Dentinal hypersensitivity management. In: Darby ML,Walsh MM, eds. Dental Hygiene Theory and Practice. 4th ed. St. Louis: Elsevier; 2015:696–706.
- Müller N, Moëne R, Cancela JA, Mombelli A. Subgingival air-polishing with erythritol during periodontal maintenance: randomized clinical trial of twelve months. J Clin Periodontol. 2014;41:883–889.
- Hägi TT, Hofmänner P, Eick S, et al. The effects of erythritol air-polishing powder on microbiologic and clinical outcomes during supportive periodontal therapy: six-month results of a randomized controlled clinical trial. Quintessence Int. 2015;46:31-41.
- de Cock P, Mäkinen K, Honkala E, Saag M, Kennepohl E, Eapen A. Erythritol is more effective than xylitol and sorbitol in managing oral health endpoints. Int J Dent. 2016;2016:9868421.
- Barnes CM, Covey DA, Watanabe H, Simetich B, Schulte JR, Chen H. An in vitro comparison of the effects of various air polishing powders on enamel and selected esthetic restorative materials. J Clin Dent. 2014;25:76-87.
- Pelka M, Trautmann S, Petschelt A, Lohbauer U. Influence of air-polishing devices and abrasives on root dentin-an in vitro confocal laser scanning microscope study. Quintessence Int. 2010;41:e141–e148.
- Pelka MA, Altmaier K, Petschelt A, Lohbauer U. The effect of air-polishing abrasives on wear of direct restoration materials and sealants. J Am Dent Assoc. 2010;141:63–70.
- Latheef P, Sirajuddin S, Veenadharini G, Mn K, Apine A. Iatrogenic damage to the periodontium caused by periodontal treatment procedures. Open Dent J. 2015;9(suppl 1:M7):203–207.
- Bühler J, Amato M, Weiger R, Walter C. A systematic review on the patient perception of periodontal treatment using air polishing devices. Int J Dent Hyg. 2016;14:4–14.
- Moëne R, Décaillet F, Andersen E, Mombelli A. Subgingival plaque removal using a new air-polishing device. J Periodontol. 2010;81:79–88.
- Alonso V, Garcia-Caballero L, Couto I, Diniz M, Diz P, Limeres J. Subcutaneous emphysema related to air-powder tooth polishing: a report of three cases. Aust Dent J. 2017;62:510–515.
- Bozbay E, Dominici F, Gokbuget AY, et al. Preservation of root cementum: a comparative evaluation of power driven versus hand instruments. Int J Dent Hygiene. 2018;16:207–209.
From Dimensions of Dental Hygiene. March 2019;17(3):41–44.




Thank you Brandy and Ewa for singing the light on this important method to manage biofilm! Kudos on an article well-researched! I hope every RDH/DDS interested in preservation of oral and restorative surfaces reads this article and begins to make the shift to Guided Biofilm Therapy incorporating Erythritol Powder.