
The Risks of Radiographic Imaging
An open discussion between practitioners and their patients can allay many of the fears associated with dental radiographic imaging.
This course was published in the June 2012 issue and expires June 2015. The author has no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Define background radiation exposure.
- Identify the difference between stochastic and deterministic effects of radiation.
- Discuss the role of the effective dose equation in comparing risks of radiation exposure.
- Detail strategies for effectively educating patients on radiation risks.
The risks associated with exposure to ionizing radiation are often discussed in the media. Just recently, a study that linked the incidence of meningioma, a type of brain tumor, to dental radiography exposure was widely publicized. 1 Some of this information can be misleading, confusing, or incorrect. As a result, patients may have questions and concerns about the safety of dental diagnostic imaging. As dental professionals, we need to be prepared to answer them.
BACKGROUND EXPOSURE

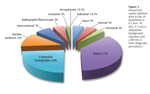
People are constantly exposed to ionizing radiation in the environment in addition to receiving periodic exposure from other sources, such as medical imaging. In the United States, the average person is exposed to 3.1 mSv of “ubiquitous background” radiation from a variety of sources, including cosmic rays from outer space and radioactive elements in some foods (Figure 1).2 The greatest single source of per-capita exposure, however, arises from the radioactive decay of radon gas arising from the soil in most areas of the US.2 Radon gas can concentrate in poorly ventilated areas, such as basements, creating a hazard for individuals living in homes or working in buildings with high concentrations. On average, background exposures make up about half of the annual dose of ionizing radiation for the US population. The other half comes from medical uses of ionizing radiation. There are many sources of medical exposures, with dentistry contributing only 2.8% to the percapita dose, mostly due to intraoral imaging.2 Computed tomography (CT) is responsible for about half of the medical contribution to our total dose, with approximately 24% of per-capita dose originating from diagnostic CT examinations. Considering that CT wasn’t commercialized until the 1970s, this represents a remarkable increase in population dose over a historically short timeframe. Some experts suggest that this increase represents a significant risk, and that from 1.5% to 2% of all US cancers may be attributable to radiation dose from CT examinations.3
STOCHASTIC AND DETERMINISTIC EFFECTS
Effects from ionizing radiation can be classified as stochastic or deterministic. Cancer is an example of a stochastic effect where the chance of the effect’s occurrence increases with more exposure but the exposures do not impact the severity of the effect. Heritable effects from ova and sperm (germ cell) mutations are another stochastic risk. Germ cell effects are insignificant for maxillofacial imaging for two reasons. First, no heritable effects attributable to ionizing radiation have been observed in human populations.3 Second, gonadal areas receive no direct exposure and only minimal amounts of scatter from radiographic examinations of the teeth and jaws. The small amounts of scatter to the gonads from dental radiography are similar to daily ubiquitous background doses.5
Deterministic effects of X-ray exposure occur when the severity of the effect increases with more exposure. There is a dose threshold below which deterministic effects do not occur. Above the threshold, the effect grows in severity with increasing dose. Examples of deterministic effects include birth defects, which have a threshold of 100 mSv to 250 mSv; opacification of the eye lens, which has a threshold of 0.5 Gy to 2 Gy of exposure; and frank cataract formation, which has a threshold of 5 Gy.6 None of these effects are found with the relatively low and localized exposures used in dental and maxillofacial imaging.
EFFECTIVE DOSE EQUATION

The International Commission on Radiological Protection (ICRP) has suggested that effective dose is the preferred method for comparing risks from different radiation exposures.6 Effective dose is a calculation that considers the most radiosensitive tissues and organs of the body and provides a fractional weighting reflecting the degree of sensitivity for each of those organs. Effective dose for diagnostic imaging is reported in milisiverts or microsieverts and is calculated using the equation:E = ? wT x HT, where E is the summation of the products of the tissue weighting factor (wT) and the dose within that tissue (HT).5
Risk assessment is related to effective dose by calculating the risk per sievert from dose-response curves. For example, the numbers of additional cancers occurring over time in an exposed population can be plotted against dose to derive a cancer/sievert risk rate. While this makes mathematical sense, the reality is that X-ray radiation is not a particularly potent carcinogen. Therefore, to see a dose-response relationship, very large populations exposed to low doses of radiation need to be studied, or the dose-response from smaller groups exposed to higher doses needs to be mathematically extrapolated. Epidemiologists and bio-statisticians perform both to arrive at risk estimations for the low doses associated with diagnostic imaging.
Long-term study of the health effects of exposure to the atomic bomb explosions of Hiroshima and Nagasaki is an example of using cancer incidence and mortality data to develop dose-response curves. Evidence for cancer risk from exposure to ionizing radiation has been documented down to 100 mGy for an adult exposure and 10 mG to 20 mG for fetal exposure.7 A linear-no-threshold hypothesis of X-ray risk fits most data for cancer development. But extrapolation of this data must be used to estimate risks from the lower doses that are used for diagnostic imaging. Current best estimates for a dose/risk relationship for cancer are 0.055 additional cancers per sievert of effective dose.
Updating of data from survivors of the atomic bombs led to changes in the calculation of effective dose recommended by the ICRP in 2007.8 Table 1 compares tissues and weighting factors from the 1990 ICRP calculation with the 2007 calculation. The risk estimation for brain tissues and the addition of salivary glands as a weighted tissue are significant for maxillofacial imaging. Also important is the addition of oral mucosa and the extrathoracic airway (nasal passages and oral pharynx) to the remainder tissue group, as well as a large increase in weight given to the remainder tissues. The 10% increase in weight of tissues located in maxillofacial area and 28% increase in weight adjusted for distribution of tissues in maxillofacial area has resulted in 32% to 42% increases in the calculation of effective dose for a variety of common dental and maxillofacial radiographic examinations.9 Another important factor to consider in relation to children is growth. In the developing child, cellular growth and organ development are associated with increased radiosensitivity of tissues. In conjunction with a longer life expectancy in which cancer can develop, children may be two times to 10 times more sensitive to radiation carcinogenesis than mature adults.10,11 The mortality risk due to radiation-induced cancer is three times to five times higher among children than adults.12

RESPONDING TO CONCERNS
When determining whether a patient needs a radiographic examination, both risks and benefits should be considered. In discussing this with patients, dental professionals should start with an assessment of potential benefits. For a patient in pain, accurate diagnosis will lead to rapid initiation of corrective therapy, thereby reducing patient discomfort and reestablishing health as soon as possible. Incomplete or inaccurate diagnosis delays appropriate therapy and may lead to a recommendation for the wrong treatment. In addition, rapid and accurate diagnosis minimizes diagnostic and therapeutic costs, as well as treatment and recovery time.
Patients need to understand that the likelihood of dental X-ray radiation exposure increasing the risk of cancer is so small, it may be meaningless. For example, if people live long enough, they have a one in five risk of dying from cancer, even without radiation exposure.13 The added risk from dental diagnostic imaging is on the order of 1-10s in 1,000,000.

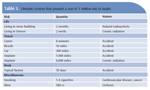
Providing patients with comparisons of alternate exposures and risks offers a context for decision making. Dental professionals can compare the expected dose of a particular radiographic examination with ubiquitous background doses (Table 2). Patients may also respond to information that compares the cancer risk associated with a specific dose to similar risks encountered from occupational, travel, and other life activities (Table 3).14
Describing the features utilized by dental practices to keep patient dose as low as reasonably achievable (ALARA principle) is often reassuring for patients. These might include high-speed film or digital receptors, which can reduce doses from 50% to 88% (Table 4). Rectangular collimation of intraoral images can further reduce the dose by approximately 40%. Appropriate use of thyroid collars can reduce dose to the thyroid among children by 26% to 33%.
The recent report of a statistically significant association between exposure to dental radiographs and meningioma has raised new concerns among patients and health care providers.14 While most meningiomas are benign, their proximity to important structures can significantly impact health. Fortunately, current imaging technologies cited above drastically reduce patient doses from dental imaging. It is important to keep diagnostic imaging risks in perspective. For instance, the pituitary dose (an area of brain close to the oral cavity) from an adult FMX produced with a round cone and D-speed film (237 ?Gy) is about the same as a month of US per-capita background dose or the dose from increased annual exposure due to cosmic rays when moving from sea level to a high elevation, such as Denver (Table 5). One misstep that should be avoided is dismissing patient concerns about radiation exposure. Educating patients by comparing dose with background exposures and risk with comparable life activities will go a long way toward gaining patient confidence and acceptance of treatment recommendations.
The contributions that radiographs make to reaching a diagnosis and directing treatment are invaluable to good oral health. In comparison to the small risks associated with state-of-the-art imaging, benefits far outweigh risks. An important part of our job as health care professionals is to communicate this message to patients in an accurate and understandable fashion.
REFERENCES
- Claus EB, Calvocoressi L, Bondy ML,Schildkraut JM, Wiemels JL, Wrensch M. Dental x-rays and risk of meningioma. Cancer. 2012 Apr 10. Epub ahead of print.
- National Council on Radiation Protection and Measurements. Report No. 160—Ionizing Radiation Exposure of the Population of the United States. Bethesda, Md: 2009.
- Brenner DJ, Hall EJ. Computed tomography—an increasing source of radiation exposure. N Engl J Med. 2007;357:2277–2284.
- Committee on the Biological Effects of Ionizing Radiation, National Research Council. Health Effects of Exposure to Low Levels of Ionizing Radiation: BEIR V. Washington, DC: National Academies Press; 1990.
- Wood RE, Harris AM, van der Merwe EJ,Nortjé CJ. The leaded apron revisited: does it reduce gonadal radiation dose in dental radiology? Oral Surg Oral Med Oral Pathol.1991;71:642–646.
- 1990 Recommendations of the InternationalCommission on Radiological Protection. Ann ICRP. 1991;21:1–201.
- Brenner DJ, Doll R, Goodhead DT, et al.Cancer risks attributable to low doses of ionizing radiation: assessing what we really know. Proc Natl Acad Sci USA. 2003;100:13761–13766.
- The 2007 Recommendations of the International Commission on Radiological Protection. ICRP publication 103. Ann ICRP. 2007;37:1–332.
- Ludlow JB, Davies-Ludlow LE, White SC. Patient risk related to common dental radiographic examinations: the impact of 2007 International Commission on Radiological Protection recommendations regarding dose calculation. J Am Dent Assoc. 2008;139:1237–1243.
- Brenner DJ, Elliston CD, Hall EJ, Berdon WE.Estimated risks of radiation-induced fatal cancer from pediatric CT. ARA Am J Roentgenol. 2001;176:289–296.
- Smith-Bindman R, Lipson J, Marcus R, et al.Radiation dose associated with common computed tomography examinations and the associated lifetime attributable risk of cancer. Arch Intern Med. 2009;169:2078–2086.
- National Research Council, Committee toAssess Health Risks from Exposure to Low Levels of Ionizing Radiation. Health Risks from Exposure to Low Levels of Ionizing Radiation: BEIR VII—Phase 2. Washington, DC: National Academies Press; 2006:424.
- Eheman C, Henley SJ, Ballard-Barbash R, et al.Annual Report to the Nation on the status of cancer, 1975-2008, featuring cancers associated with excess weight and lack of sufficient physical activity. Cancer. 2012;118:2338–2366.
- Cohen BL. Catalogue of risks extended andupdated. Health Phys. 1991;61:317–335.
From Dimensions of Dental Hygiene. June 2012; 10(6): 56, 59-61.



