How the Epstein-Barr Virus Impacts Periodontal Health
From oral lesions to periodontal destruction, this virus is emerging as a critical yet often overlooked factor in dental patient care.
This course was published in the May/June 2026 issue and expires June 2029. The authors have no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
AGD Subject Code: 730
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Identify the signs and symptoms of the Epstein-Barr virus (EBV).
- Discuss the periodontal pathogens that correlate with EBV.
- Note the role that dental hygienists play in managing patients with EBV.
The Epstein-Barr virus (EBV), a member of the human herpesvirus IV family, is a widespread and impactful pathogen with a complex history.1 Recognized in 1968, EBV is the etiological agent of infectious mononucleosis, commonly referred to as mono.. EBV primarily spreads through saliva, which led to its nickname “the kissing disease,” though its modes of transmission are not limited to this pathway. Sharing drinks, eating utensils, or any other contaminated object with infected saliva can facilitate transmission, making it a master of subtle spread.2–10
Dental hygienists are easily exposed in clinical settings through routine procedures that involve saliva, especially those that generate aerosols. Dental hygienists are not only at risk for EBV exposure, they are uniquely positioned to recognize its oral manifestations and guide patients on proper management.
Signs and Symptoms
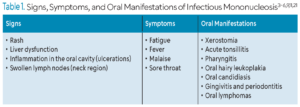
EBV is typically asymptomatic in most individuals; however, it can cause visible signs and symptoms in cases of infectious mononucleosis (Table 1).2–7,9,11 These include fatigue, fever, swollen lymph nodes, rash, oral candidiasis, oral hairy leukoplakia, gingivitis, and periodontitis.12
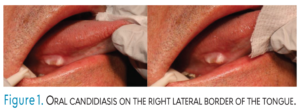
Oral candidiasis, also known as “oral thrush,” arises from an overgrowth of Candida albicans fungi, appearing as soft, creamy white patches on the tongue, buccal mucosa, and the soft and hard palate (Figure 1).8,13 These fungal plaques can be easily scraped away. C. albicans is a commensal species, meaning that it benefits from the oral environment but does not harm it. However, in large amounts, C. albicans can become pathogenic, thus causing denture stomatitis. Oral candidiasis is commonly found in patients with oral prostheses.14,15 Immunocompromised patients, especially those with human immunodeficiency virus, are at risk for infections from C. albicans.15
EBV may also cause oral hairy leukoplakia to develop in the oral cavity, which may be misdiagnosed as oral candidiasis.2-4,16 Hairy leukoplakia presents as persistent white patches or lesions firmly attached to the ventral, dorsolateral, and lateral borders of the tongue, soft palate, floor of the mouth, and oropharynx. This manifestation is extremely resistant to scraping and will continue until the viral source is addressed.8,17 Like oral candidiasis, oral hairy leukoplakia is more often found in immunosuppressed individuals.16 Oral hairy leukoplakia is contagious, and oral candidiasis is typically not.13
Research shows EBV is associated with increased risk of periodontitis, specifically chronic periodontitis.9,18–20 Zeng et al19 infected human gingival fibroblasts with levels of EBV. This resulted in high levels of inflammatory cytokines. Inflammatory cytokines are associated with periodontal inflammation, which explains how EBV may cause gingivitis.
Pathophysiology
A thorough understanding of the pathophysiology of EBV enables clinicians to comprehend how the virus targets the body and how the body responds. EBV first crosses the body’s physical barriers, then activates both the innate and adaptive immune responses. The virus specifically infects B lymphocytes and epithelial cells in the oropharynx, particularly the tonsils. The adaptive immune response is activated, targeting the specific antigens involved. B cells attach to the antigens, processing them and initiating an immune response locally in the tonsils. This activation of B cells results in IgG, IgM, and IgA antibody production. Additionally, B cells and macrophages stimulate cytotoxic T cell (CD8) activity, which identifies and eliminates infected B cells.2,3,5-7,21-23
After the initial infection, EBV establishes latency through memory B cells. The virus produces latency-associated transcripts, which enable the virus to remain undetectable, with the opportunity to reactivate. This can lead to the recurrence of symptoms and contribute to chronic conditions. Specific factors, such as stress, immunosuppressants, and environmental influences, can trigger this viral reactivation, perpetuating its impact on the body.2-4,6,7,9,22
Chronic periodontitis and EBV continue to rise globally, with correlations found between the two. Current evidence suggests a potential link between EBV reactivation and butyric acid (BA) found in oral fluids, likely influenced by BA-producing bacteria such as Porphyromonas gingivalis and Fusobacterium nucleatum.9,24 Although more mechanistic research and larger clinical studies are still needed, progress has been limited by the fact that EBV infects only humans, making traditional animal models unsuitable. To address this challenge, a new humanized-mouse model is being developed, and early findings show EBV-positive B-cell infiltration and osteoclast induction in periodontal tissues, with validation ongoing.9
Co-Infection With Periodontally Involved Bacteria
The periodontal pathogens that correlate with EBV-reactivation are: P. gingivalis, Aggregatibacter actinomycetemcomitans, F. nucleatum, Prevotella intermedia, Helicobacter pylori, Streptococcus pyogenes, Treponema pallidum, and Tannerella forsythia.9,24
- gingivalis, a red-complex bacteria, is highly prevalent in EBV-positive patients with periodontal diseases. In patients with chronic periodontitis, EBV DNA and P. gingivalis were detected together more often in deeper periodontal pockets than when either organism was present alone.20 This EBV-P. gingivalis coexistence occurred in approximately 40% of chronic periodontitis patients with probing depths ≥ 5 mm, and the associated odds ratio of 4.67 was higher than that of either microorganism individually. These findings indicate that the combined presence of EBV and periodontopathic bacteria may significantly elevate the risk of periodontitis.8,9,20
- actinomycetemcomitans is a Gram-negative anaerobic bacterium associated with periodontitis, previously referred to as aggressive periodontitis. A. actinomycetemcomitans triggers reactivation of EBV into its productive viral cycle.24 P. intermedia is an obligate anaerobic, rod-shaped bacterium that is known for causing periodontal diseases. Unlike other periodontal pathogens, this bacterium does not secrete high concentrations of BA. This suggests that it does not create an EBV reactivation through the histone deacetylases pathway, ultimately, facilitating an alternate mechanism. Because P. intermedia produces lipopolysaccharides (LPS) that drive inflammatory responses in periodontal diseases, it also contributes to structural and molecular changes mediated by pro-inflammatory interleukins. Additionally, the LPS in P. intermedia in host immune cells activates dependent pathways through cell-surface receptors. Ultimately, these signaling pathways lead to lytic gene expression that results in the reactivation of EBV.24-26
Although S. pyogenes is primarily recognized for causing pharyngitis, skin infections, and various systemic complications, its behavior offers meaningful insight into how bacterial pathogens can influence oral and periodontal health. This organism has been shown to enhance EBV reactivation by activating B cells and disrupting normal immune regulation, emphasizing how bacterial-viral interactions can contribute to broader inflammatory responses within the oral cavity.24
Coinfection with group A Streptococci (GAS) and EBV has also been documented in pharyngitis, where GAS-derived superantigens nonspecifically stimulate T cells and further amplify inflammation.24 While S. pyogenes is not classified as a traditional periodontal pathogen, its capacity to modulate host immunity, promote inflammatory cascades, and increase salivary viral loads suggests that similar mechanisms may intensify periodontal inflammation and accelerate tissue breakdown.24
Dental Hygienists’ Role in Patients With Epstein-Barr Virus
Comprehensive medical and dental histories must be taken to identify if a patient has any potential or known diagnosis of EBV infection or infectious mononucleosis. A thorough patient interview will help with the collection of all relevant information. The dental hygienist should document all findings by noting any signs or symptoms. Because EBV is so contagious, dental hygienists should not treat an infected patient but rather encourage him or her to reschedule the appointment. However, providing appropriate guidance on managing symptoms is vital to supporting patients’ well-being.
In some cases, patients may be unaware of their EBV infection and present to their dental hygiene appointments. Dental hygienists should complete a thorough extraoral and intraoral examination and connect the signs, symptoms, and oral manifestations discussed in Table 1, specifically ulcerations in the oral cavity, swollen lymph nodes in the neck, and hairy leukoplakia/oral candidiasis. Once these conditions have been identified, further questioning of other symptoms, such as fatigue, fever, and sore throat, should be done, as well as the history and duration of symptoms.3–6,11,21 Further questioning of the patient to identify duration is critical because the incubation period of infectious mononucleosis can last for 4 to 6 weeks.27 Once symptoms resolve, patients are safe to resume dental hygiene services.28 Patients should seek their primary care provider for an official diagnosis, but can be educated on EBV management in the dental setting.
In terms of oral hygiene instruction, patients with oral lesions, particularly painful ones, should monitor symptoms closely and seek a referral to the proper healthcare professional, such as physicians or medical virology specialists, if symptoms persist or worsen.5,29 For pain relief associated with EBV, over-the-counter medications, such as acetaminophen or ibuprofen, can be recommended, as well as brushing gently using a soft-bristled toothbrush to minimize irritation. Staying hydrated, the use of saline rinses to soothe throat discomfort, and fluoride treatments to protect tooth enamel in cases of xerostomia are all helpful strategies for patients with EBV.12 Once recovered, patients should replace oral hygiene products to prevent reinfection. Patients currently or previously infected with EBV should be educated about proper self-care to reduce the likelihood of gingivitis and periodontitis.
When caring for patients affected by both EBV and periodontitis, the dental hygienist’s role centers on comprehensive assessment, prevention, and strategic patient education. Due to EBV’s ability to increase inflammatory pathways and interact with periodontal pathogens, clinicians must closely monitor overall periodontal status. This includes meticulous periodontal charting, efficient debridement, and targeted biofilm disruption while remaining aware that viral-related immune dysregulation may negatively influence healing responses. Effective collaboration with medical providers is essential to ensure coordinated management and timely referral when inflammation does not resolve as expected, including referral to a periodontist when deeper probing depths persist. Through this integrated approach, dental hygienists help support improved periodontal outcomes in patients whose disease may be complicated by EBV reactivation and its associated symptoms.
Dental hygienists can provide psychosocial support by listening and acknowledging concerns and encouraging stress-reducing techniques to manage EBV symptoms.22,30 Patients need providers who practice active listening to help empower them, especially with this type of condition.31 Dental hygienists can also help by offering patients resources, including community support and educational resources from organizations such as the National Organization for Rare Disorders.32
Conclusion
EBV poses significant implications for both general and oral health. Dental hygienists play a crucial role in recognizing important oral manifestations, guiding patients in managing symptoms, and providing preventive care. Dental hygienists need to be careful in the event that patients are unaware of a potential infection, so maintaining standard precautions is crucial. Early identification of EBV-related symptoms, along with time-effective referrals to healthcare providers, is essential for effective patient care.
Consideration of a patient’s immunosuppressed health status and potential re-infection with EBV is essential for dental hygienists to identify, especially in patients with periodontitis. The coinfection of EBV with periodontitis intensifies tissue breakdown due to the pro-inflammatory cytokines. These cytokines impair the host’s immune response by triggering a synergistic relationship that contributes to deeper periodontal pockets and advanced loss of soft tissue. Understanding the complexities of EBV, its co-infection rate with periodontal pathogens, and its impact on oral health enables oral health professionals to deliver comprehensive, patient-centered care that addresses both the physical and emotional aspects of the infection.
References
- Sangueza-Acosta M, Sandoval-Romero E. Epstein-Barr virus and skin. An Bras Dermatol. 2018;93:786–799.
- Yu H, Robertson ES. Epstein-Barr virus history and pathogenesis. Viruses. 2023;15:714.
- Damania B, Kenney SC, Raab-Traub N. Epstein-Barr virus: Biology and clinical disease. Cell. 2022;185:3652–3670.
- Chakravorty S, Afzali B, Kazemian M. EBV-associated diseases: Current therapeutics and emerging technologies. Front Immunol. 2022;13:1059133.
- Hoover K, Higginbotham K. Epstein-Barr Virus. Treasure Island, Florida: StatPearls Publishing; 2023.
- Houen G, Trier NH. Epstein-Barr virus and systemic autoimmune diseases. Front Immunol. 2021;11:587380.
- Dasari V, McNeil LK, Beckett K, et al. Lymph node targeted multi-epitope subunit vaccine promotes effective immunity to EBV in HLA-expressing mice. Nat Commun. 2023;14:4371.
- Slots J, Saygun I, Sabeti M, Kubar A. Epstein-Barr virus in oral diseases. J Periodontal Res. 2006;41:235–424.
- Imai K, Ogata Y. How does Epstein-Barr virus contribute to chronic periodontitis? Int J Mol Sci. 2020;21:1940.
- Khanna R. Kissing the Epstein-Barr virus goodbye? Available at science.org.au/curious/people-medicine/epstein-barr-virus. Accessed April 2, 2026.
- Naughton P, Healy M, Enright F, Lucey B. Infectious mononucleosis: diagnosis and clinical interpretation. Br J Biomed Sci. 2021;78:107–116.
- United States Centers for Disease Control and Prevention. About Epstein-Barr Virus. Available at cdc.gov/epstein-barr/about/index.html. Accessed April 2, 2026.
- Talapko J, Juzbašić M, Matijević T, et al. Candida albicans—The virulence factors and clinical manifestations of infection. J Fungi (Basel). 2021;7:79.
- Devcic M, Simonic-Kocijan S, Prpic J, et al. Oral candidal colonization in patients with different prosthetic appliances. J Fungi (Basel). 2021;7:662.
- Manikandan S, Vinesh E, Selvi D, Kannan R, Jayakumar A, Dinakaran J. Prevalence of Candida among denture wearers and nondenture wearers. J Pharm Bioallied Sci. 2022;14(Suppl 1):S702–705.
- Mahalakshmi SKJ, Safreen V, Sivaraj R, Haripriya G, Deepasree B. Oral candiasisis mimicking leukoplakia in an immunocompetent male: A rare case report. Int J Pharm Sci. 2025;3:5343–5346.
- Mohammed F, Fairozekhan AT. Oral Leukoplakia. Treasure Island, Florida: StatPearls Publishing; 2023.
- Gao Z, Lv J, Wang M. Epstein–Barr virus is associated with periodontal diseases. Med (Baltimore). 2017;96:e5980.
- Zeng W, Liu G, Luan Q, et al. Epstein-Barr virus promotes inflammatory cytokine production in human gingival fibroblasts. Int Dent J. 2024;74:607–615.
- Kato A, Imai K, Ochiai K, Ogata Y. Higher prevalence of Epstein–Barr Virus DNA in deeper periodontal pockets of chronic periodontitis in Japanese patients. PLoS One. 2013;8:e71990.
- Che Y, Ding X, Xu L, et al. Advances in the treatment of Hodgkin’s lymphoma (Review). Int J Oncol. 2023;62:61.
- Chen CJ. Epstein-Barr virus reactivation and disease flare of systemic lupus erythematosus. Taiwan J Obstet Gynecol. 2024;63:161–164.
- Lamont R, Hajishengallis G, Koo H, Jenkinson H. Oral Microbiology and Immunology. 3rd ed. Washington, DC: ASM Press; 2019:480.
- Indari O, Ghosh S, Bal A, et al. Awakening the sleeping giant: Epstein-Barr virus reactivation by biological agents. Pathog Dis. 2024;82:ftae002.
- Nørskov-Lauritsen N, Claesson R, Jensen A, Åberg C, Haubek D. Aggregatibacter actinomycetemcomitans: clinical significance of a pathobiont subjected to ample changes in classification and nomenclature. Pathog. 2019;8:243.
- Könönen E, Fteita D, Gursoy U, Gursoy M. Prevotella species as oral residents and infectious agents with potential impact on systemic conditions. J Oral Microbiol. 2022;14:2079814.
- Mayo Clinic. Mononucleosis. Mayo Clinic; 2022. Available at mayoclinic.org/diseases-conditions/mononucleosis/symptoms-causes/syc-20350328. Accessed April 2, 2026.
- College of Dental Hygienists of Ontario. Fact Sheet: Mononucleosis. Available at https://cdho.org/factsheets/mononucleosis. Accessed April 2, 2026.
- Cleveland Clinic. Virologist. Available at https://my.clevelandclinic.org/health/articles/25116-virologist. Accessed April 2, 2026.
- Ackerman B. Communication skills for dental professionals: Do you hear me now? Available at https://www.todaysrdh.com/communication-skills-for-dental-professionals-do-you-hear-me-now. Accessed April 2, 2026.
- Pieren J, Gadbury-Amyot C. Darby and Walsh: Dental Hygiene Theory and Practice. 6th ed. New York: Elsevier; 2024.
- National Organization for Rare Disorders. Community Support. Available at https://rarediseases.org/community-support. Accessesd April 2, 2026.
From Dimensions of Dental Hygiene. May/June 2026; 24(3):36-39