
Reduce the Risk of Adverse Pregnancy Outcomes
Preventive therapies and oral hygiene education before conception and during early pregnancy may help reduce the severity of periodontal diseases and the negative effects of inflammation on pregnancy.
This course was published in the February 2015 issue and expires February 2018. The authors have no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Identify the adverse pregnancy outcomes associated with inflammation and periodontal diseases.
- Explain how maternal oral infection can affect a developing fetus.
- Discuss the current professional recommendations for treating and managing pregnant patients.
Maternal periodontitis has been linked to adverse pregnancy outcomes, such as preterm birth, low birth weight, growth restriction, and preeclampsia.1–3 Although the exact mechanisms remain unclear, strong evidence supports the concept that periodontal infections provide a pathway for oral microorganisms to reach the fetus. In addition, some dental practitioners may be unclear on the proper protocols for the treatment and management of pregnant patients. The purpose of this review is to educate oral health professionals on the effects of periodontal diseases on pregnancy, and to discuss clinical guidelines for managing pregnant patients.
Historically, periodontal diseases are the result of poor oral hygiene and excessive plaque accumulation, resulting in loss of the supporting tissues of the periodontium.4,5 Thus, treatment of periodontitis has largely been focused on plaque reduction and antibacterial therapies. Emerging studies, however, question whether bacterial overgrowth is the cause or result of periodontitis. One study suggests that it is the host’s response, rather than specific bacteria or their virulence factors, that determines whether periodontal diseases develop and progress.1 Although plaque biofilm plays a large role in the initiation of periodontal diseases, an individual’s immune response to the chronic presence of specific bacteria may be more involved in progression of the diseases than previously thought. As the severity of periodontal diseases progresses, the risk of experiencing adverse pregnancy outcomes increases.6
ADVERSE PREGNANCY OUTCOMES ASSOCIATED WITH INFLAMMATION
An estimated one in every nine infants in the United States is born preterm (less than 37 weeks gestation).7 Preterm birth is the second most common cause of death in children younger than 5 and the leading risk factor for deaths caused by neonatal infections.8 In addition, preterm birth can contribute to long-term growth restrictions and cognitive delays.9 In fact, nearly half of long-term neurological complications are associated with preterm delivery.10 Despite significant advances in neonatal medicine, preterm births and complications continue to negatively impact public health.
The reason for the increasing prevalence of preterm births is not clearly understood. Risk factors include smoking, alcohol consumption, obesity, low socioeconomic status, stress, and advanced maternal age.11–13 Furthermore, maternal infections such as periodontal diseases have been associated with an increased risk for preterm birth.1,14 Not only does the presence of periodontal diseases early in pregnancy appear to be a risk factor for preterm birth, but the worsening of periodontal diseases during pregnancy may independently increase the risk of fetal exposure and resultant preterm birth.15
Despite evidence supporting an association between maternal periodontitis and preterm birth, whether periodontal treatment delivered during pregnancy would prevent preterm birth remains unclear. Some findings suggest that treatments may be ineffective at preventing preterm birth, depending on the nature and timing of treatment provided.16 For example, it is unknown if more intensive treatment or treatment targeted at specific components of inflammation, such as Fusobacterium nucleatum, would reduce the rate of preterm births.17,18 Nonetheless, prevention of periodontal diseases remains the goal of oral health care providers in reducing adverse pregnancy outcomes.
Preeclampsia is another adverse pregnancy outcome that affects approximately 3% to 10% of pregnant women.19,20 It is characterized by hypertension and proteinuria that develops after the 20th week of gestation.21 Preeclampsia and other hypertensive disorders are among the most common medical complications of pregnancy and are responsible for an estimated 15% of maternal mortality.21
Research suggests that periodontal diseases may be an independent risk factor for preeclampsia.19–21 One study reports that women with periodontal diseases were almost three times more likely to develop preeclampsia than pregnant women with a healthy periodontium.20 The etiology of preeclampsia is most likely multifactorial; however, research suggests that the presence and severity of periodontal diseases seem to increase the risk for the occurrence and severity of preeclampsia among pregnant women.22 As with preterm birth, oral health education and prevention of disease remain an integral part of reducing women’s risk for preeclampsia during pregnancy.
In addition to preterm birth and preeclampsia, other adverse pregnancy outcomes, such as low birth weight and intrauterine growth restriction, may also be associated with periodontal diseases.23,24 Low birth weight is defined as weighing less than 2,500 grams, or 5.5 pounds.8 In 2012, more than 300,000 babies were born weighing less than 2,500 grams (Table 1).25 Intrauterine growth restriction occurs when the weight is below the 10th percentile for gestational age.8 While the exact mechanism for the association remains a topic of scientific research, maternal infection and/or inflammation seem to play a large role in adverse pregnancy outcomes.26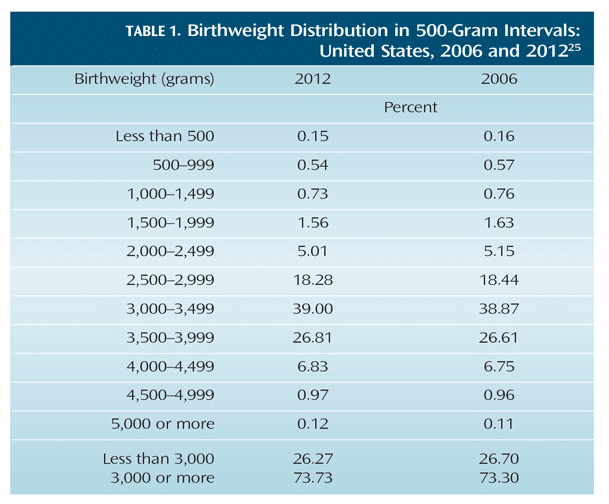
During normal pregnancy, the placenta invades the surrounding uterine tissue and provides an exchange of nutrients and waste between mother and fetus via the umbilical cord. As pregnancy progresses, amniotic fluid levels containing prostaglandin E2 (PGE2) and inflammatory cytokines—tumor necrosis factor (TNF-?) and interleukin 1(IL-1?)—steadily rise to reach the threshold that induces labor.27 Thus, normal labor and delivery are induced by inflammatory signaling. One theory for the association between periodontal diseases and preterm birth is that women with periodontitis, a bacterial infection, exhibit an increase in fluid mediator levels and inflammatory cytokines, which can trigger labor prematurely.28 Furthermore, an increase in other markers of inflammation such as C-reactive protein (CRP) has been associated with an elevated risk for preeclampsia and intrauterine growth restriction.29
![]() PERIODONTAL DISEASES AND PREGNANCY HEALTH
PERIODONTAL DISEASES AND PREGNANCY HEALTH
Periodontitis, which affects an average of 38% of women in the US,7 is characterized by the destruction of connective tissue and alveolar bone due to infection by periodontal pathogens.30 Periodontitis is a complex disease involving multiple causative factors, both modifiable and nonmodifiable.30 In addition to the presence of Gram-negative anaerobes in subgingival biofilm, modifiable risk factors include smoking, undiagnosed or uncontrolled diabetes, cardiovascular diseases, stress, and obesity.30 Pregnancy is among the list of nonmodifiable potential risk factors along with osteoporosis, hematological disorders, and individuals’ immune responses to periodontal diseases.30
Bacterial infections such as intrauterine infections are often associated with adverse pregnancy outcomes.8 Intrauterine infections originate in the lower genital tract and ascend into the amniotic membranes and placenta, and may potentially reach the fetus.16 These bacterial infections cause a rapid increase in the levels of inflammatory mediators and other biological fluids that normally induce labor.16 Likewise, periodontal diseases represent a potential source of microorganisms known to directly or indirectly affect the health of a developing fetus.8,15 Women with periodontal diseases may experience more frequent and severe bacteremias than periodontally healthy women, increasing the chance the uterus will be exposed to oral bacteria or their byproducts.17 One study found that pregnant women with severe periodontitis could be up to seven times more likely to deliver prematurely, depending on whether the fetus or only the placenta was exposed to the infection.15
Perhaps the most compelling data arise from evidence of oral organisms of maternal origin found within the placenta. Katz et al31 found that Porphyromonas gingivalis, a common periodontal pathogen, may colonize placental tissue and contribute to preterm delivery. This study was the first to find P. gingivalis present in the placental tissue of both normal asymptomatic placenta and women with chorioamnionitis—an infection caused by bacteria invading the membranes and amniotic fluid surrounding the fetus. Furthermore, Aagaard et al32 found the presence of a microbiome similar to that of the oral cavity in placentas of healthy pregnant women.32
In the first few weeks of gestation, the placenta is in a state of physiological hypoxia, potentially facilitating the growth of Gram-negative anaerobes such as P. gingivalis.33 Although the definitive role of P. gingivalis and other oral organisms in preterm delivery is not fully understood, the association between preterm labor and bacterial infection is well established.34,35
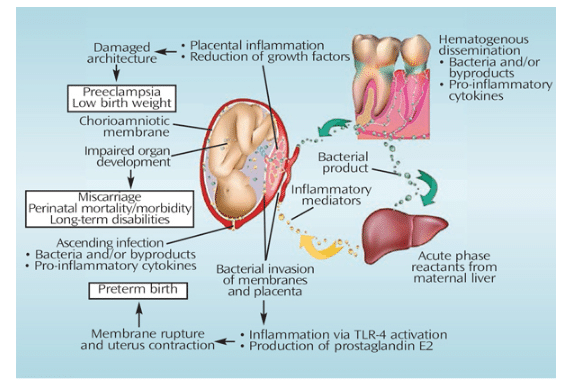
During pregnancy, there is a hormone-induced increase in vascular permeability that facilitates entrance of periodontal pathogens and their byproducts into the bloodstream.28 In patients with periodontal diseases, host cells activate a local inflammatory response against bacteria in an effort to eliminate the infection. It has also been reported that inflammatory cytokines, periodontal bacteria, and/or their virulence factors may enter the blood circulation and disseminate throughout the body—triggering a systemic response to the infection.8 A proposed model (Figure 1) illustrates two possible mechanisms that may mediate this systemic response:
1. Direct Pathway
Periodontal bacteria and their byproducts disseminate to the feto-placental unit, where they trigger an inflammatory response. This results in the elevation of inflammatory cytokines that may contribute to adverse pregnancy outcomes.
2. Indirect Pathway
Inflammatory cytokines produced locally in periodontally diseased tissues enter the blood circulation and reach the feto-placental unit and the liver, which can stimulate cytokine production and acute phase protein responses.8,28 Regardless of the pathway, the host’s response of increasing production of inflammatory mediators in the presence of periodontal diseases could potentially lead to adverse pregnancy outcomes.
IIMPLICATIONS FOR DENTAL HYGIENE PRACTICE
While the effects of periodontal diseases on pregnancy are well documented, it is estimated that less than half of women receive dental care during pregnancy.36,37 Women from low socioeconomic backgrounds are even less likely to see an oral health professional.38 For this reason, dental hygienists need to educate women on the effects of periodontal diseases on pregnancy, especially before conception in women of childbearing age. Studies report conflicting evidence as to whether providing nonsurgical periodontal therapy during pregnancy reduces the incidence of preterm births; however, periodontal therapy can improve maternal oral health.39,40 Providing periodontal therapy during pregnancy may be too late to interfere with inflammatory responses that lead to adverse pregnancy outcomes.41,42 Treating the disease prior to pregnancy would allow for more aggressive therapy and a longer maintenance phase for systemic inflammation to resolve.43
Although studies show that few risks are associated with routine dental care during pregnancy,8,43 general obstetric guidelines suggest that elective dental procedures be avoided during the first and last trimesters of pregnancy. In order to manage periodontal health during pregnancy, dental hygienists should identify the stage of pregnancy and perform a comprehensive oral evaluation and periodontal examination.8 If the pregnant patient is diagnosed as periodontally healthy, treatment should focus on oral health promotion and education for mother and child. The healthy patient should then be scheduled for re-evaluation at a later stage in pregnancy, if necessary. In the presence of gingivitis, treatment should focus on reducing the bacterial load and signs of inflammation. Once periodontal health is established, the patient should be monitored frequently throughout her pregnancy.
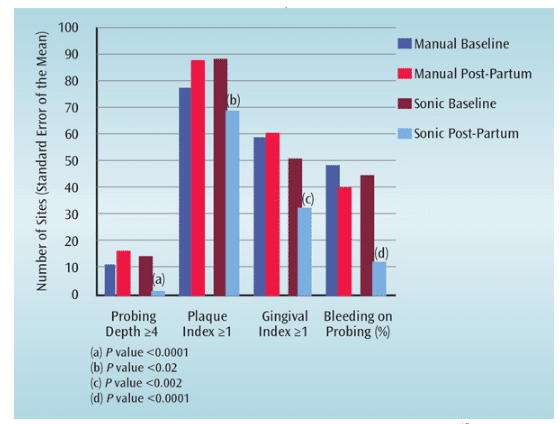
In the pregnant patient with periodontitis, treatment should focus on reducing subgingival biofilm and inflammation with standard nonsurgical periodontal therapy.8 Among pregnant women, the sonic toothbrush provides better reduction in oral plaque and inflammation than a manual brush (Figure 2).8 The benefits of using a sonic toothbrush are accompanied by significant improvements in the extent of probing depths ?4 mm, number of plaque scores ?1, number of gingival scores ?1, and the number of sites with bleeding on probing. Furthermore, one study reported that sonic brushing combined with nonsurgical periodontal therapy was associated with a 3.8-fold reduction in the rate of preterm delivery compared to manual toothbrush use.15
While the treatment of periodontal diseases during pregnancy has not been shown to either increase or decrease the occurrence of adverse pregnancy outcomes, women do not seem to respond optimally to periodontal therapy until after delivery.17,44 Studies do, however, show a positive response to oral hygiene education and preventive therapies before conception and during early pregnancy.45,46 Emphasizing the importance of plaque control during pregnancy and encouraging the daily use of an antimicrobial mouthrinse have been associated with decreased incidence of preterm birth.47 Jeffcoat et al47 found that pregnant subjects with periodontal diseases who rinsed twice daily with an alcohol-free mouthwash containing cetylpyridinium chloride (CPC) experienced a significant decrease in the incidence of preterm birth when compared to the nonrinse group. Therefore, the addition of a sonic toothbrush and alcohol-free mouthrinse to the pregnant patient’s established oral health routine could be beneficial in the management of periodontitis. The results of these studies are encouraging to dental hygienists and provide an effective treatment alternative for pregnant patients who do not regularly access dental care.
CONCLUSION
Despite recent medical advances, preterm birth and other adverse pregnancy outcomes are a growing public health concern and economic burden on the health care system. Periodontal diseases are systemic infections and inflammatory responses that can represent a potential risk for pregnancy complications. Although periodontal treatment during pregnancy has not been proven to reduce adverse pregnancy outcomes, preventive therapies and oral hygiene education before conception and during early pregnancy could lessen the severity of these diseases and their effects. Therefore, dental hygienists should emphasize the importance of oral health and the need for regular dental care before conception and throughout pregnancy.
REFERENCES
- Shanthi V, Vanka A, Bhambal A, et al. Association of pregnant women periodontal status to preterm and low-birth weight babies: A systematic and evidence-based review. Dent Res J. 2012;9:368–380.
- Barak S, Oettinger-Barak O, Machtei EE, et al. Evidence of periopathogenic microorganisms in placentas of women with preeclampsia. J Periodontol. 2007;78:670–676.
- Ide M, Papapanou PN. Epidemiology of association between maternal periodontal disease and adverse pregnancy outcomes—systematic review. J Periodontol. 2013;84(Suppl 4):S181–S194.
- Tatakis DN, Kumar PS. Etiology and pathogenesis of periodontal diseases. Dent Clin North Am. 2005;49:491–516.
- Caton JG, Quinones CR. Etiology of periodontal diseases. Curr Opin Dent. 1991;1:17–28.
- Bartold PM, Van Dyke TE. Periodontitis: a host-mediated disruption of microbial homeostasis. Unlearning learned concepts. Periodontol 2000. 2013;62:203–217.
- Centers for Disease Control and Prevention. Preterm Birth. Available at:?cdc.gov/reproductivehealth/maternalinfanthealth/pretermbirth.htm. Accessed January 26, 2015.
- Sanz M, Kornman K. Periodontitis and adverse pregnancy outcomes: consensus report of the Joint EFP/AAP Workshop on Periodontitis and Systemic Diseases. J Clin Periodontol. 2013;40:S164–S169.
- Chang HH, Larson J, Blencowe H, et al. Preventing preterm births: analysis of trends and potential reductions with interventions in 39 countries with very high human development index. Lancet. 2013;381:223–234.
- McCormick MC. The contribution of low birth weight to infant mortality and childhood morbidity. N Engl J Med. 1985;312:82–90.
- Slattery MM, Morrison JJ. Preterm delivery. Lancet. 2002;360:1489–1497.
- Goldenberg RL, Culhane JF, Iams JD, Romero R. Epidemiology and causes of preterm birth. Lancet. 2008;371:75–84.
- Djelantik AA, Kunst AE, Van der Wal MF, Vrijkotte TG. Contribution of overweight and obesity to the occurrence of adverse pregnancy outcomes in a multi-ethnic cohort: population attributive fractions for Amsterdam. BJOG. 2012;199:283–290.
- Mannem S, Chava VK. The relationship between maternal periodontitis and preterm low birth weight: A case-control study. Contemp Clin Dent. 2011;2:88–93.
- Offenbacher S, Lin D, Strauss R, et al. Effects of periodontal therapy during pregnancy on periodontal status, biologic parameters, and pregnancy outcomes: a pilot study. J Periodontol. 2006;77:2011–2024.
- Barros SP, Offenbacher S, Genco RJ, Williams RC. Adverse pregnancy effects. In: Periodontal Disease and Overall Health: A Clinician’s Guide. 2nd ed. Yardley, Pennsylvania: Professional Audience Communications Inc; 2014:152–175.
- Michalowicz BS, Gustafsson A, Thumbigere-Math V, Buhlin K. The effects of periodontal treatment on pregnancy outcomes. J Clin Periodontol. 2013;40:S195–S208.
- Han YW, Fardini Y, Chen C, et al. Term stillbirth caused by Fusobacterium nucleatum. Obstet Gynecol. 2010;115:442–445.
- Varshney S, Gautam A. Poor periodontal health as a risk factor for development of pre-eclampsia in pregnant women. J Indian Soc Periodontol. 2014;18:321–325.
- Huang X, Wang J, Liu J, et al. Maternal periodontal disease and risk of preeclampsia: A meta-analysis. J Huazhong Univ Sci Technolog Med Sci. 2014;34:729–735.
- Boggess KA, Lieff S, Murtha AP, et al. Maternal periodontal disease is associated with an increased risk for preeclampsia. Obstet Gynecol. 2003;101:227–231.
- Canakci V, Canakci CF, Yildirim A. et al. Periodontal disease increases the risk of severe pre-eclampsia among pregnant women. J Clin Periodontol. 2007;34:639–645.
- Offenbacher S, Lieff S, Boggess KA, et al. Maternal periodontitis and prematurity. Part I: Obstetric outcome of prematurity and growth restriction. Ann Periodontol. 2001;6:164–174.
- Saini R, Saini S, Saini SR. Periodontitis: A risk for delivery of premature labor and low-birth-weight infants. J Nat Sci Biol Med. 2010;1:40–42.
- Martin JA, Hamilton BE, Osterman MJK, Curtin SC, Matthews TJ. Births: final data for 2012. Natl Vital Stat Rep. 2015;64:10.
- Romero R, Espinoza J, Goncalves LF, et al. The role of inflammation and infection in preterm birth. Semin Reprod Med. 2007;25:21–39.
- Haram K, Mortensen JH, Wollen AL. Preterm delivery: an overview. Acta Obstet Gynecol Scand. 2003;82:687–704.
- Madianos PN, Bobetsis YA, Offenbacher S. Adverse pregnancy outcomes (APOs) and periodontal disease: pathogenic mechanisms. J Clin Periodontol. 2013;40:S170–S180.
- Herrera JA, Parra B, Herrera E, et al. Periodontal disease severity is related to high levels of C-reactive protein in pre-eclampsia. J Hypertens. 2007;25:1459–1464.
- AlJehani YA. Risk factors of periodontal disease: review of the literature. Int J Dent. 2014;2014:1–9.
- Katz J, Chegini N, Shiverick KT, Lamont RJ. Localization of P. gingivalis in preterm delivery placenta. J Dent Res. 2009;88:575–578.
- Aagaard K, Ma J, Antony KM, et al. The placenta harbors a unique microbiome. Sci Transl Med. 2014;6:237.
- James JL, Cartwright JE, Whitley GS, et al. The regulation of trophoblast migration across endothelial cells by low shear stress: consequences for vascular remodeling in pregnancy. Cardiovasc Res. 2012;93:152–161.
- Klein LL, Gibbs RS. Infection and preterm birth. Obstet Gynecol Clin North Am. 2005;32:397–410.
- Goldenberg RL, Hauth JC, Andrews WW. Intrauterine infection and preterm delivery. N Engl J Med. 2000;342:1500–1507.
- Mangskau KA, Arrindell B. Pregnancy and oral health: utilization of the oral health care system by pregnant women in North Dakota. Northwest Dent. 1996;75:23–28.
- Gaffield ML, Gilbert BJ, Malvitz DM, Romaquera R. Oral health during pregnancy: an analysis of information collected by the pregnancy risk assessment monitoring system. J Am Dent Assoc. 2001;132:1009–1016.
- Lydon-Rochelle MT, Krakowiak P, Hujoel PP, Peters RM. Dental care use and self-reported dental problems in relation to pregnancy. Am J Public Health. 2004;94:765–771.
- Polyzos NP, Polyzos, IP, Mauri D, et al. Effect of periodontal disease treatment during pregnancy on preterm birth incidence: a meta-analysis of randomized trials. Am J Obstet Gynecol. 2009;200:225–232.
- Kim AJ, Lo AJ, Pullin DA, et al. Scaling and root planing treatment for periodontitis to reduce preterm birth and low birth weight: a systematic review and meta-analysis of randomized controlled trials. J Periodontol. 2012;83:1508–1519.
- Goldenberg RL, Culhane JF. Preterm birth and periodontal disease. N Engl J Med. 2006;355:1925–1927.
- Xiong X, Buekens P, Goldenberg RL, et al. Optimal timing of periodontal disease treatment for prevention of adverse pregnancy outcomes: before or during pregnancy? Am J Obstet Gynecol. 2011;205:e1–e6.
- Michalowicz BS, DiAngelis AJ, Novak MJ, et al. Examining the safety of dental treatment in pregnant women. J Am Dent Assoc. 2008;139:685–695.
- Pirie M, Linden G, Irwin C. Intrapregnancy non-surgical periodontal treatment and pregnancy outcome: a randomized controlled trial. J Periodontol. 2013;84:1391–1400.
- Geisinger ML, Geurs NC, Bain JL, et al. Oral health education and therapy reduces gingivitis during pregnancy. J Clin Periodontol. 2014;41:141–148.
- Kaur M, Geisinger ML, Geurs NC, et al. Effect of intensive oral hygiene regimen during pregnancy on periodontal health, cytokine levels, and pregnancy outcomes: a pilot study. J Periodontol. 2014;85:1684–1692.
- Jeffcoat M, Parry S, Gerlach RW, et al. Use of alcohol-free antimicrobial mouth rinse is associated with decreased incidence of preterm birth in a high-risk population. Am J Obstet Gynecol. 2011;382:e1–e6.
From Dimensions of Dental Hygiene. February 2015;13(2):55–60.


 PERIODONTAL DISEASES AND PREGNANCY HEALTH
PERIODONTAL DISEASES AND PREGNANCY HEALTH
