 GZZZ, CC BY-SA 4.0
GZZZ, CC BY-SA 4.0 Oral Health Considerations for Geographic Tongue
Dental professionals should be aware of the clinical features, etiology and pathogenesis, treatment, and oral health considerations for this unique pathosis.
This course was published in the July 2021 issue and expires July 2024. The authors have no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Identify the etiology and pathogenesis of geographic tongue (GT).
- Describe the clinical presentation of GT.
- Review treatment strategies for improving outcomes for patients with GT.
Geographic tongue (GT) is a benign oral mucosal lesion that usually affects the dorsal and lateral surfaces of the tongue.1 This condition was first described by Rayer2 in 1831 as a wandering rash of the tongue, and has since been known by other names including benign migratory glossitis, erythema migrans, and annulus migrans.3 The disease is characterized by alternating activity, remission, and reactivation at various locations of the tongue. This article will review the clinical features of GT, etiology and pathogenesis, treatment, and oral health considerations. A clinical case of GT will be presented to familiarize oral health professionals with this unique pathosis.
EPIDEMIOLOGY AND ETIOLOGY
GT affects between 1% and 3% of the population, and occurs more frequently in women than men. All age groups can be affected; however, GT tends to occur more often in adults than children. Racial and ethnic predilection have not been reported for this condition.3–5
The etiology for GT is unknown. However, a number of etiological factors have been suggested. A hereditary tendency had been reported in previous studies investigating siblings of a parent affected by GT while a more recent study of 5-year-old monozygotic twins with GT supported a genetic etiology.6–8
GT has also been associated with systemic diseases including atopy, allergy, asthma, stress, eczema, psoriasis, hormonal changes, vitamin deficiencies, and syndromes such as Reiter syndrome and Down syndrome. Another study showed that 25% of patients with geographic tongue had allergy concurrently.9–11
Controversy exists concerning the association between GT and psoriasis. Studies have shown similarities between both conditions and the presence of a common genetic marker, the human leukocyte antigen (HLA) HLA-Cw6.12–16 A weak association has also been noted with HLA-B13 in both GT and psoriasis.9 However, patients without psoriasis can present with GT as well. Picciani et al17 conducted a review of the literature to describe the relationship between psoriasis and GT. They concluded that GT is an oral manifestation of psoriatic disease, presenting histopathological, immunohistochemical, and genetic similarities with that of plaque psoriasis. They recommended patients with psoriasis have a comprehensive intraoral and extraoral examination, and patients with GT have skin and joint examinations.
Vitamin deficiency has been considered as a potential etiology for GT. Specifically, Vitamin D, B6, B12, folic acid, iron, and zinc deficiency have been suggested as contributing factors related to GT.9,17 A study was conducted on the levels of iron, zinc, and vitamin B12 in blood and saliva of patients presenting with GT as compared with healthy subjects in a dental school setting. The authors indicated that a positive effect can be found when zinc sulfate is administered in improving the filiform papillae in patients with GT, but further study is needed to investigate the use of systemic or topical zinc as a treatment option for GT.18
Hormone changes have been considered a potential etiology for GT. Oral contraceptive pills has been associated with GT.9 Ghalayani et al19 examined if salivary levels of estrogen and progesterone in pregnant women with and without GT were different as increased prevalence of GT had been noted in pregnancy. Their study showed no significant difference regarding the level of sex hormones between case and control groups, but other factors including HLA marker, stress, and genetic potential could aggravate the incidence of GT.
Psychological factors, such as stress and anxiety, may play a role in the etiology of GT. Ebrahimi et al20 found an association between stress and GT, noting that decreasing stress can help in the healing of lesions. Other studies have examined the relationship between anxiety and GT indicating that anxiety increases the risk for GT.21,22
Some studies have indicated that GT is less common in smokers.9 This may be due to increased keratinization and decreased inflammatory cytokines, such as tumor necrosis factor-alpha, interleukin (IL)-1, and IL-6 by macrophages, which occur through the activation of nicotine receptors in these cells.17
PATHOPHYSIOLOGY
While the etiology of GT remains unknown, recent studies have revealed varying insights into the pathophysiology of this inflammatory disease. Levels of the inflammatory mediator, IL-8, are increased among individuals with GT, and is thought to contribute to the acute inflammatory response.23 More specifically, the higher salivary levels of IL-8 were positively correlated with the severity of GT. Neutrophils were found in the subepithelial GT lesions, suggesting that IL-8 was the pro-inflammatory cytokine in the pathogenesis of GT. However, the papillary atrophy of GT and the subsequent colonization of bacterial and fungal organisms might also contribute to the neutrophil recruitment.23 Another possible suggestion for the involvement of IL-8 in GT, is the involvement of IL-8 in angiogenesis, the process by which new blood vessels form to assist with the healing process.23,24
Another study demonstrated that salivary calprotectin may be involved in the pathogenesis of GT.25 In this study, increased calprotectin levels were significantly and positively correlated with the number of lesions on the tongue and increased levels of salivary IL-8.
Discussions regarding genetic implications in the pathophysiology of GT have also been explored. A mutation in the IL36RN gene was detected in a three-generational family with GT alone.26 This mutation is carried in an autosomal dominant inheritance pattern with incomplete penetrance meaning that everyone that carries the genetic mutation may not necessarily display the phenotypic characteristics of this disease. Biopsies of GT in individuals demonstrated neutrophil infiltration in the epidermis when compared to a control group. Individuals who manifested with GT without the IL36RN mutations demonstrated decreased expression ratios of IL-36Ra and IL-36y proteins in the lingual tissue suggesting other factors are associated with GT.26
CLINICAL PRESENTATION
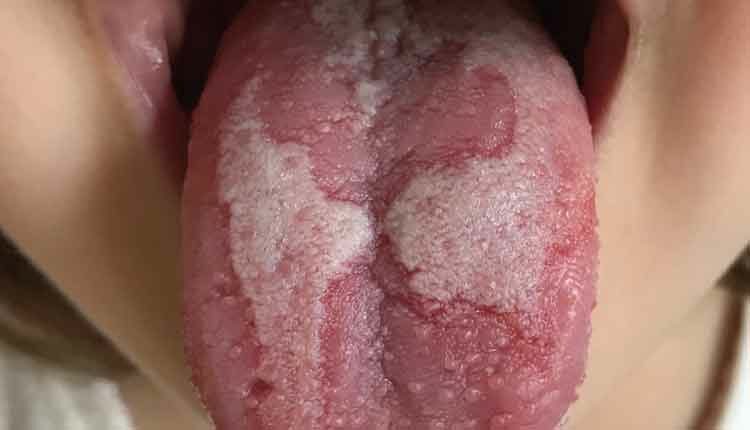
GT lesions present as multiple, well demarcated areas of erythema, surrounded by a white, serpiginous, hyperkeratotic elevated border. There is atrophy of the filiform papillae.4 The characteristic pattern reported by patients is spontaneous resolution of the lesion in one area with return of normal tongue architecture, followed by another lesion appearing in a different location of the tongue. This pattern of resolution and recurrence to a new location on the tongue is what led to the migratory name. Lesions may change in location, pattern, and size, and occur on other mucosal surfaces, including the floor of the mouth and buccal mucosa.4,27 Lesions can last up to 2 weeks.
GT is usually painless, although some patients have complained of discomfort associated with eating acidic or spicy foods. Other symptoms include burning sensations, taste impairment, and tongue pain. A study of patterns of presentation of GT in a Nigerian teaching hospital evaluated the clinical presentations of 21 cases of GT.28 In addition to the classic presentation of GT, five reported itchy tongue, six had painful and burning sensations, and five had a fissured tongue. Ching et al29 assessed the frequency of GT and fissured tongue in a retrospective study of 161 patients with burning mouth syndrome compared to 87 individuals with temporomandibular joint dysfunction used as a control. Findings revealed that 26.7% of the subjects were diagnosed with geographic and fissured tongue as compared to the control group with a male to female ratio of 1:2. Similarly, Picciani et al30 assessed the presence of GT and fissured tongue in patients with psoriasis. Their study compared the prevalence of GT and fissured tongue between 348 individuals with psoriasis and 348 healthy controls. Results showed a higher incidence of GT and fissured tongue in the psoriasis group than the healthy controls with the incidence of GT higher in patients with early psoriasis and fissured tongue noted more in those with late psoriasis. The authors concluded that GT is associated with disease severity and may be considered a marker of psoriasis disease severity.
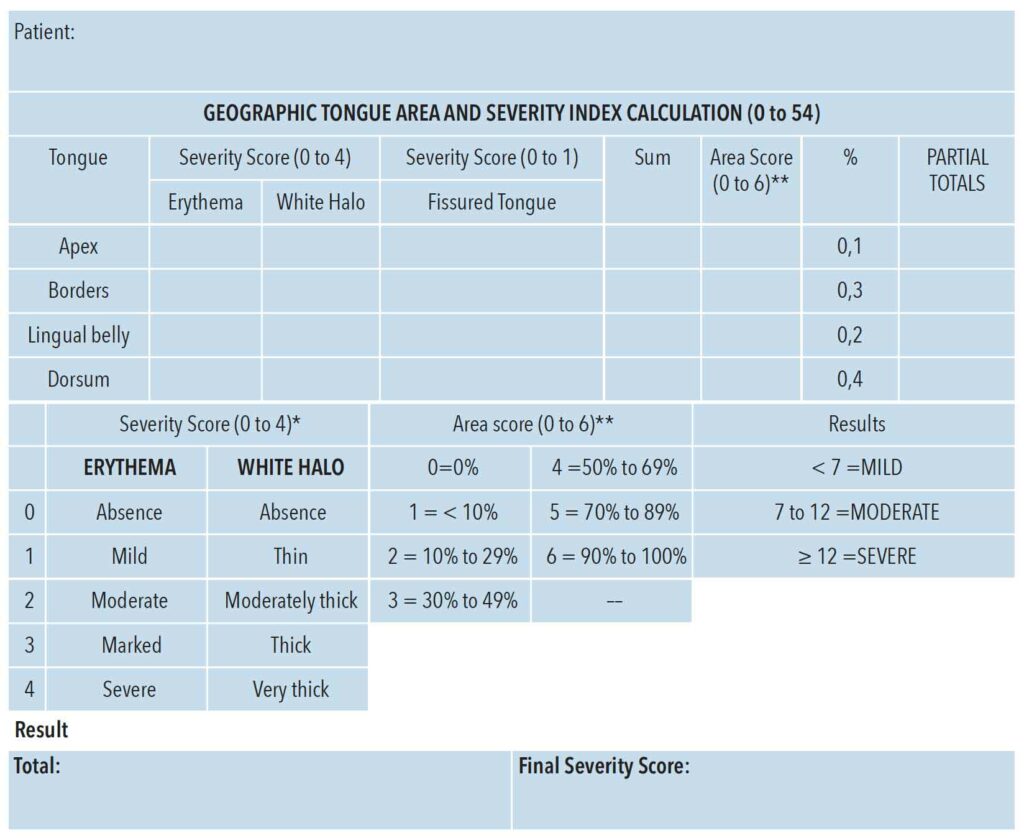
In 2020, Picciani et al31 published a severity index for GT based on a retrospective sample of 100 participants and 500 images of GT that were evaluated by three researchers experienced in both GT and psoriasis. Based on the analysis performed, GT was classified as mild, moderate, or severe. The Geographic Tongue Area and Severity Index offers a way to characterize GT in a reliable manner with three main characteristics of the lesion evaluated: erythema, white halo, and presence of fissured tongue. Each of these three characteristics is scored on a scale of 0 to 4 and the total severity index ranges from 0 to 54, where 0 means total absence of GT lesions and 54 is the highest degree of severity (Figure 1).31
DIAGNOSIS, TREATMENT, PROGNOSIS
Based on the clinical appearance and history, GT is usually diagnostic. In equivocal cases, a differential diagnosis might include candidiasis, lichen planus, contact stomatitis, chemical burns, Reiter syndrome, anemia, and squamous cell carcinoma.3–5
In most cases, GT is asymptomatic, and, because it is a benign condition, GT does not require any treatment. For symptomatic cases of GT, use of topical corticosteroids, antihistamines, cyclosporine, and vitamin A may provide some relief. Triggers that can intensify GT symptoms include alcohol; hot, spicy, and sour foods; and acidic fruits or beverages.3 Consumption of these may cause the existing symptoms to become more uncomfortable. According to a current systematic review, specific recommendations on how to effectively reduce symptoms of GT are not available.32
A study by Aung-Din et al33 evaluated the effectiveness of a tacrolimus swish-and-spit treatment two times a day to treat GT symptoms. According to the study, this technique was successful in managing the symptoms of oral lichen planus. The results showed that after using the solution twice daily, 70% of the patients reported improvement of their symptoms. The authors concluded that the use of the tacrolimus swish-and-spit solution is low cost, easy to use, and may be beneficial in treating symptoms relating to GT.
The prognosis for GT is good because the condition is usually benign and asymptomatic.3 Most patients who present with GT may only have symptoms temporarily and may have knowledge of what to avoid to prevent burning sensations.
CLINICAL CASE
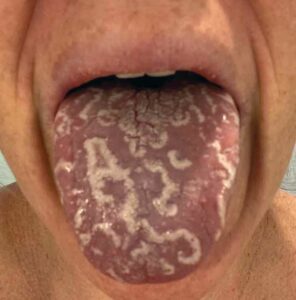
The following case describes a 37-year-old woman in her second trimester of pregnancy with symptoms of GT. This patient presented with lesions on her tongue that appeared as smooth patches of missing filiform papillae surrounded by white, curved, and slightly raised borders on the dorsal and lateral surfaces. The patient reported that this appearance varies throughout her pregnancy; sometimes becoming more severe and then decreasing back to a mild state. The lesions were accompanied by burning sensations on occasion, but presently she was asymptomatic. The patient stated that her tongue has had similar appearance in a previous pregnancy and did not return to normal until she was post-partum (Figure 2).
In the past, the patient was advised to rinse with warm salt water to help with burning tongue sensations, but since the patient reported that it only occasionally bothered her, no other recommendations were made. For her current condition of GT, the patient was asymptomatic. She was advised to avoid spicy or acidic foods and drinks, as the patient self-reported that these foods usually exacerbated burning tongue sensations. The patient was advised to return for further care if she became symptomatic.
CONCLUSION
For many cases of GT, an integrated approach to diagnosis and management is important. The symptoms and severity of GT vary, and co-occurring systemic diseases, periods of remission and exacerbation, vitamin deficiencies, hormone changes, stress, and anxiety may further complicate diagnosis and treatment.
In order to provide a patient-centered approach, communication and collaboration with other healthcare practitioners are needed. For example, individuals with GT should also be evaluated for signs of psoriasis and then provided with appropriate referrals.
As oral health professionals, recommending stress management programs and initiating mental health referrals might be indicated for individuals with GT. Similarly, a referral to an allergist or registered dietitian may be appropriate depending on suspected etiology. Communication with these practitioners and individuals with GT is vital to treat symptoms. Interprofessional collaboration provides an opportunity to create positive health outcomes for individuals with GT.
REFERENCES
- Dafar A, Bankvall M, Cevik-Aras H, et al. Lingual microbiota profiles of patients with geographic tongue. J Oral Microbiol. 2017;9:1355206.
- Goswami M, Verma A, Verma M. Benign migratory glossitis with fissured tongue. J Indian Soc Pedod Prev Dent. 2012;30:173–175.
- Shareef S, Ettefagh L. Geographic tongue. StatPearls. Available at: ncbi.nlm.nih.gov/books/NBK554466. Accessed June 15, 2021.
- Kelsch RD. Geographic tongue. Available at: emedicine.medscape. com/article/1078465-overview. Accessed June 15, 2021.
- Delong L, Burkhart NW. General and Oral Pathology for the Dental Hygienist. 3rd ed. Philadelphia: Wolters Kluwer; 2019:335.
- Edelman E, Chosack A, Cohen T. Scrotal tongue and geographic tongue: polygeneic and associated trials. Oral Surg Oral Med Oral Pathol.1993;76:476–479.
- Redman RS, Shapiro BL, Gorlin RJ. Hereditary component in the etiology of benign migratory glossitis. Am J Hum Genet. 1972;24:124–133.
- Gunashekar M. Geographic tongue in monozygotic twins. J Clin Diagn Res. 2014;8:ZD01–02.
- Nandini DB, Bhavana SB, Deepak, BS, et al. Paediatric geographic tongue: a case report, review and recent updates. J Clin Diagn Res. 2016;10:ZE05–9.
- Ogueta CI, Ramirez PM, Jiménez OC, et al. Geographic tongue: what a dermatologist should know. Actas Dermosifiliogr. 2019;110:341–346.
- Goregen M, Melikoglu M, Miloglu O, et al. Predisposition of allergy in patients with benign migratory glossitis. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2010;110:470–474.
- Marks R, Tait B. HLA antigens in geographic tongue. Tissue Antigens. 1980;15:6–62.
- Ladizinski B, Lee KC, WIlmer E, et al. A review of the clinical variants and the management of psoriasis. Adv Skin Wound Care. 2013;26:271–284.
- Bachelez H. Immunopathogenesis of psoriasis: recent insights on the role of adaptive and innate immunity. J Autoimmun. 2005;25:69–73.
- Chandran V. The genetics of psoriasis and psoriatic arthritis. Clin Rev Allergy Immunol. 2013;44:149–156.
- Ulmansky M, Michelle R, Azaz B. Oral psoriasis: report of six new cases. J Oral Pathol Med. 1995;24:42–45.
- Picciani BLS, Domingas TA, Teixeira-Souza T, et al. Geographic tongue and psoriasis: clinical, histopathological, immunohistochemical and genetic correlation—a literature review. An Bras Dermatol. 2016;91:410–421.
- Khayamzadeh M, Najafi S, Sadrolodabaei P, et al. Determining salivary and serum levels of iron, zinc and vitamin B12 in patients with geographic tongue. J Dent Res Dent Clin Dent Prospect. 2019;13:221–226.
- Ghalayani P, Tavangar A, Nilchian F, et al. The comparison of salivary level of estrogen and progesterone in 1st, 2nd and 3rd trimester in pregnant women with and without geographic tongue. Dent Res J(Isfahan). 2013;10:609–612.
- Ebrahimi H, Pourshahidl S, Tadbir AA, et al. The relationship between geographic tongue and stress. Iran Red Crescent Med J. 2919L12:313–315.
- Scariot R, Sebastiani AM, Meger MN, et al. Anxiety and genetic polymorphisms in cetechol-O-methyltransferase (COMT) and serotonin transportation gene (5HTT) are associated with benign migratory glossitis. Oral Surg Oral Med Oral Pathol Oral Radiol. 2019;127:218–224.
- Slikhani M, Khalighinejad N, Glalaiani P, et al. Immunologic and psychologic parameters associated with geographic tongue. Oral Surg Oral Med Oral Pathol Oral Radiol. 2014;118:68–71.
- Dafar A, Bankvall M, Garsjö V, Jontell M, Çevik-Aras H. Salivary levels of interleukin-8 and growth factors are modulated in patients with geographic tongue. Oral Dis. 2017;23:757–762.
- Qazi BS, Tang K, Qazi A. Recent advances in underlying pathologies provide insight into interleukin-8 expression mediated inflammation and angiogenesis. Int J Inflam. 2011;2011:1–13.
- Garsjo V, Dafar A, Jontell M, et al. Increased levels of calprotectin in the saliva of patients with geographic tongue. Oral Dis. 2019;26:558–565.
- Liang J, Huang P, Li H, et al. Mutations in IL36RN are associated with geographic tongue. Hum Genet. 2017;136:241–252.
- Borrie F, Musthyala R, Macintyre D. Ectopic geographic tongue—a case report. Dent Update. 2007;34:121–122.
- Oyetola EO, Oluwande A, Agho ET. Geographic tongue: pattern of presentation in a south western Nigerian teaching hospital. Ann Ibd Pg Med. 2018;16:131–135.
- Ching V, Grushka M, Darling M, et al. Increased prevalence of geographic tongue in burning mouth complaints: a retrospective study. Oral Surg Oral Med Oral Pathol Oral Radiol. 2012;114:444–448.
- Picciani BLS, Souza TT, Santos VCB, et al. Geographic tongue and fissured tongue in 348 patients with psoriasis: correlation with disease severity. Scientific World Journal. 2015;2015:564326.
- Picciani BLS, Santos LR, Teixeira-Souza T, et al. Geographic tongue severity index: a new and clinical scoring system. Oral Surg Oral Med Oral Pathol Oral Radiol. 2020:129:330–338.
- de Campos WG, Esteves CV, Fernandes LG, Domaneschi C, Lemos Júnior CA. Treatment of symptomatic benign migratory glossitis: a systematic review. Clin Oral Investig. 2018;22:2487–2493.
- Aung-Din D, Heath M, Wechter T, et al. Effectiveness of the tacrolimus swish-and-spit treatment regimen in patients with geographic tongue. JAMA Derm. 2018;154:1481–1482.
From Dimensions of Dental Hygiene. July 2021;19(7):32-35.



