 SOMEONE25/ISTOCK/GETTY IMAGES PLUS
SOMEONE25/ISTOCK/GETTY IMAGES PLUS
Noting the Oral Signs of Tuberculosis
In light of expanding globalization and drug resistance, the resurgence of tuberculosis in some regions increases the possibility of infected individuals presenting for dental care—thus the need to recognize the disease’s oral manifestations.
This course was published in the July 2021 issue and expires July 2024. The authors have no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Describe the reasons for the resurgence of tuberculosis in some areas, the impact of globalization, and ramifications of the increasing prevalence of drug-resistant tuberculosis.
- Explain the pathogenesis of this disease, its various forms, clinical presentation, and mortality.
- Discuss the ways in which tuberculosis manifests in the oral cavity.
The COVID-19 pandemic has brought about many changes, not the least of which is a renewed attention to infectious diseases. Preventive efforts, such as mask wearing, have slowed the occurrence of diseases, such as the seasonal flu. However, other infectious diseases, including tuberculosis, continue to spread and adversely affect global populations, causing undue economic and health burdens. As the current pandemic has shown, globalization has made the world a smaller place, making it even more crucial to prevent the spread of pathogenic organisms. This article will update the reader on tuberculosis and its oral manifestations.
After decades of decreasing incidence, tuberculosis resurgence is being seen in some regions due to factors that include the human immunodeficiency virus (HIV) epidemic, increased immigration from countries with endemic tuberculosis, transmission of tuberculosis in crowded or unsanitary environments, and poor healthcare infrastructure. Additionally, this review is especially prescient considering the increasing prevalence of drug-resistant tuberculosis.
Tuberculosis is an infectious granulomatous disease caused by Mycobacterium tuberculosis and, less frequently, by other two species of bacteria, M. bovis and M. africanum.1,2 Primary infection occurs in the lungs via inhalation of droplets generated during coughing, sneezing, shouting, and singing. Primary tuberculosis is usually asymptomatic, with limited cases exhibiting fever and pleural effusion. In primary infection, the bacterium enters an unsensitized host, inducing an immune response. In the case of tuberculosis infection, the immune response—which causes granuloma formation—can also severely damage tissues.
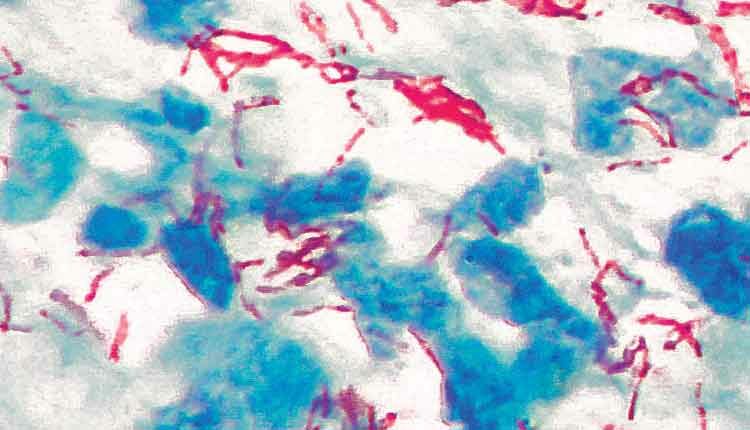
Secondary tuberculosis occurs in a previously sensitized host, usually as a reactivation of a latent infection. Only 5% to 10% of infections develop into active disease (Figure 1).3 Active tuberculosis usually develops from the secondary reactivation. Symptoms include fever, chills, fatigue, malaise, weight loss, and persistent cough.3 In the latter stages, if untreated, the host dies due to progressive pulmonary and extrapulmonary inflammation and, ultimately, multi-organ failure. That is, the disease induces immunopathology which results in death.4 M. tuberculosis can also travel to other parts of the body via diffuse dissemination through the vascular system, often producing multiple small foci of infection that resemble millet seeds. This results in infection of organs, such as the kidney and liver, and is called miliary tuberculosis.3
LEADING CAUSE OF DEATH
Tuberculosis is the leading cause of death from an infectious disease, affecting men (56%), women (32%), and children (12%).1,5 Despite reductions in incidence rates in many areas, tuberculosis remains a global health concern, as an estimated 10.4 million new cases were reported in 2015 (10% of cases occurred in children, and 12% involved HIV coinfection).1 In 2015, there were an estimated 1.8 million deaths due to tuberculosis globally, and more than 95% of cases and deaths took place in developing countries.1
infection.COURTESY SAMI EL TOUM, DDS
In 2019, 8916 cases were reported in the United States; 51.5% of these cases occurred in four states: California (23.7%), Texas (13.0%), New York (8.5%), and Florida (6.3%). In addition, 71.4% of the overall US cases occurred among people born outside of the US.6 In countries with a high tuberculosis burden—including regions of Southeast Asia (44%), Africa (25%), and the Western Pacific (18%)—transmission more often occurs in schools, public transportation settings, workplaces, healthcare facilities, mines, and prisons.1,5 It is possible to effectively reduce a country’s tuberculosis burden by implementing a comprehensive strategy, at scale, using a combination of treatment and prevention. The World Health Organization (WHO) characterizes tuberculosis as “intimately linked to poverty,” with its control and eradication being “ultimately a question of justice and human rights.”6
The characteristic pathology that develops as a result of M. tuberculosis infection is the granuloma, a collection of inflammatory cells, and immune cells (macrophages, lymphocytes, epithelioid histiocytes, and multinucleated giant cells). In other cases, the host clears the infection, but still harbors the microorganism—which can be reactivated later in life.3,5 Asymptomatic individuals are able to transmit the bacterium to others without realizing it. In the process, they may develop immunity that effectively limits proliferation of the bacterium in a few weeks—although infected hosts are unable to eliminate all organisms.
An important thing to note about M. tuberculosis is its ability to become resistant to the drugs used to treat it. This increases the focus on preventing transmission, as resolution of infection via antibiotics or other medications may be less effective than in the past.
DIAGNOSIS AND TREATMENT
Two tests are administered to confirm if a person has been infected with M. tuberculosis: a Mantoux tuberculin skin test and tuberculosis blood test. In order to find out if the person has latent tuberculosis infection or if the infection has progressed to tuberculosis disease, other diagnostic methods—such as a chest X-ray and sputum sampling—are used.7
About 85% of people who develop tuberculosis can be successfully treated with a 6-month antibiotic drug regimen (usually four first-line antibiotics: isoniazid, rifampicin, pyrazinamide, and ethambutol).5 Unfortunately, patient compliance with such regimens is low. Commonly, isoniazid is started as a prophylaxis, normally in individuals with a positive purified protein derivative skin test and a normal chest X-ray. In most circumstances, a multidrug therapy is started with two or more drugs to kill the tubercle bacilli quickly. This is to reduce the development of drug resistance, which is a worldwide health issue.7
DRUG RESISTANCE
The WHO considers drug-resistant tuberculosis as a public health crisis, with approximately half a million people across the globe falling ill with this disease annually.8 In the United States, the Centers for Disease Control and Prevention (CDC) lists drug-resistant tuberculosis as a “serious threat” to human health.9–11
Drug-resistant tuberculosis can occur due to a lack of availability of drugs for proper treatment, incorrect dosage or duration of treatment, and/or poor patient compliance with treatment regimens.12 There are three types of drug-resistant tuberculosis: drug-resistant, multidrug-resistant, and extensively drug-resistant tuberculosis.12 In 2019, 631 cases of drug-resistant tuberculosis were reported in the US.11
There has been interest in the use of bacille Calmette-Guerin (BCG) vaccine throughout the world, but its efficacy has raised concern, as it does not prevent primary infection or reactivation of latent pulmonary infection, which is the principal source of infection in many communities.13 However, in children, the BCG vaccine has a protective effect against meningitis and disseminated tuberculosis.13
ORAL MANIFESTATIONS
Oral manifestations of tuberculosis are rare—seen in 0.05% to 5% of all infections—and usually appear as a chronic ulcer that may be multiple or singular, painful, or painless.2,3,14 Primary oral tuberculosis lesions (those appearing without pulmonary involvement) present on the gingiva, mucobuccal fold, in areas of soft tissue inflammation, or in extraction sites.14 These lesions occur most frequently in younger people, and while the mechanism of primary inoculation is unknown, it is hypothesized the Mycobacterium is inoculated directly into the oral mucosa.2 There may also be palpable cervical lymph nodes.2,14 Secondary oral tuberculosis lesions (those appearing secondary to pulmonary involvement) are more commonly seen on the tongue, lips, buccal mucosa, and, less frequently, on the palate, gingival mucosa, and lingual frenum.2
CASE REPORT 1
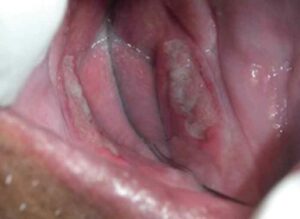
Figure 1 is from a case involving a 65-year-old man with type II diabetes. The patient was a nonsmoker who was not taking any medications. His chief complaint was of a tender, nonhealing ulcer that had been present for 3 months, increasing in size during that time. The ulcer was unsuccessfully treated with miconzaole and various mouthrinses (undisclosed formulas). The patient was referred to an oral pathologist, who detected a single enlarged submandibular lymph node on the right side, which was firm, tender, and fixed. A biopsy was done and histopathological examination disclosed granulomatous inflammation in the connective tissue containing epithelioid and multinuclear giant cells.
Subsequently, a real-time polymerase chain reaction was performed to ascertain the lesion’s DNA. The lesion tested positive for Mycobacterium complex and the patient was referred to a pulmonologist for continued care. Subsequently, further tests were performed and the findings were consistent with those of primary extrapulmonary tuberculosis.14 The patient completed a regimen of isoniazid, rifampicin, pyrazinamide, and ethambutol daily for 6 months and the lesion healed.
CASE REPORT 2
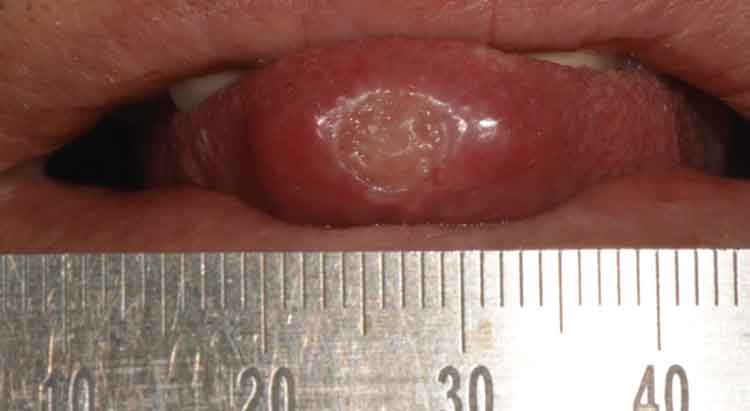
Figure 2 is from a case involving a 57-year-old man whose past medical history revealed a diagnosis and treatment of tuberculosis more than 40 years prior to development of an oral lesion.15 The patient’s chief complaint was of a painful ulcer on the tip of his tongue that developed—without any incident of trauma, stress, or systemic illness—approximately 4 weeks prior to presentation in the dental clinic. Upon examination, a slightly elevated, round lesion with a granulomatous center and a whitish, well-defined border, measuring approximately 0.7 cm in diameter, was found on the tip of the tongue.
Extraorally, lymph node enlargement was not evident and there were no other systemic symptoms; a subsequent panoramic radiograph revealed no additional findings. Initial differential diagnosis included major aphthous ulcer, traumatic ulcer, granulomatous diseases, and infections. Quickly, aphthous ulcer and traumatic ulcer were ruled out because of the long appearance of the ulcer and absence of trauma in the history.
Initially, a topical mouthrinse with a mixture of amoxicillin 1.0 g and prednisolone 30 mg in 500 ml distilled water was prescribed for 7 weeks. Additionally, triamcinolone acetonide 5 mg was injected into the lesion twice. The lesion’s appearance improved slightly, but the ulcer was still present, so a biopsy was performed. Histologically, the biopsy revealed the presence of numerous epithelioid cells and multiple Langhans giant cells. Ziehl-Neelsen staining demonstrated acid-fast bacilli and the lesion was diagnosed as M. tuberculosis. The patient was immediately referred to a pulmonologist, who ordered additional testing (polymerase chain reaction and blood tests, plus a chest radiograph) that confirmed the diagnosis of tuberculosis. The patient was prescribed anti-tuberculosis medications and, within 2 months, the lesion had almost completely healed.
![TABLE 1. Why Is Tuberculosis Difficult to Eradicate in High Burden Countries?]() ADDITIONAL CONSIDERATIONS
ADDITIONAL CONSIDERATIONS
According to the CDC, oral healthcare providers must report confirmed cases of tuberculosis or clinically suspected tuberculosis to the local health department.16 Clough et al17 suggest that patients with tuberculosis should be treated with the ”same high standard of care for patients with infectious diseases as available to any other patient,” and that “when caring for people who may have active tuberculosis, aerosol-generating procedures should only be carried out in an appropriately engineered and ventilated area, ideally a negative pressure room.” The CDC recommends dental office staff include protocols for tuberculosis infection control in their offices’ written infection prevention program.18
Why does the human immune system respond to M. tuberculosis in such an opposite manner, providing both protection and initiating tissue damage? Scientists have attempted to identify the specific immune responses responsible for these startling differences, but thus far have been unsuccessful. This is only one of the reasons why tuberculosis is still so prevalent in world populations. Tuberculosis is a disease of the poor and eradication efforts are thwarted by some of the challenges listed in Table 1.
CONCLUSION
The COVID-19 pandemic has elucidated the importance of restraining and monitoring the spread of infectious diseases in all parts of the world. As oral lesions associated with tuberculosis most commonly present as a chronic tongue ulcer, oral healthcare professionals should strive to consider this in their differential diagnosis of such lesions. A thorough review of the medical history can provide important clues as to possible diagnosis of these lesions.
REFERENCES
- Churchyard G, Kim P, Shah NS, et al. What we know about tuberculosis transmission: an overview. J Infect Dis. 2017;216:S629–S635.
- Ito FA, de Andrade CR, Vargas PA, Jorge J, Lopes, MA. Primary tuberculosis of the oral cavity. Oral Dis. 2005;11:50–53.
- Neville BW, Damm DD, Allen CM, Chi AC. Oral and Maxillofacial Pathology. 4th ed. Philadelphia: Saunders; 2015.
- Basaraba RJ, Hunter RL. Pathology of tuberculosis: How the pathology of human tuberculosis informs and directs animal models. Microbiol Spectr. 2017;5:117–129.
- World Health Organization. Global Tuberculosis Report 2020. Available at: apps.who.int/iris/bitstream/handle/10665/336069/9789240013131-eng.pdf. Accessed June 14, 2021.
- United States Centers for Disease Control and Prevention. Reported Tuberculosis in the United States, 2019. Available at: cdc.gov/tb/statistics/reports/2019/default.htm. Accessed June 14, 2021.
- Weinberg M, Thiele CW, Fine JB. Oral Pharmacology for the Dental Hygienist. 2nd ed. London: Pearson; 2013;132–134.
- World Health Organization. Tackling the drug-resistant TB crisis. Available at: who.int/activities/tackling-the-drug-resistant-tb-crisis. Accessed June 14, 2021.
- United States Centers for Disease Control and Prevention. Antibiotic Resistance Threats in the United States. Available at: cdc.gov/drugresistance/pdf/threats-report/2019-ar-threats-report-508.pdf. Accessed June 14, 2021.
- Unite States Centers for Disease Control and Prevention. Drug-Resistant TB. Available at: cdc.gov/TB/Topic/DRTB. Accessed June 14, 2021.
- United States Centers for Disease Control and Prevention. Trends in Tuberculosis, 2019. Available at: cdc.gov/tb/publications/factsheets/statistics/tbtrends.htm. Accessed June 14, 2021.
- United States Centers for Disease Control and Prevention. Treatment for TB Disease. Available at: cdc.gov/tb/topic/treatment/tbdisease.htm. Accessed June 14, 2021.
- World Health Organization. BCG Vaccine. Available at: who.int/teams/health-product-and-policy-standards/standards-and-specifications/vaccines-quality/bcg. Accessed June 14, 2021.
- Aoun N, El‐HajJ G, El Toum S. Oral ulcer: an uncommon site in primary tuberculosis. Aust Dent J. 2015;60:119–122.
- Kim SY, Byun JS, Choi JK, Jung JK. A case report of a tongue ulcer presented as the first sign of occult tuberculosis. BMC Oral Health. 2019;19,1–5.
- United States Centers for Disease Control and Prevention. Menu of Suggested Provisions for State Tuberculosis Prevention and Control Laws. Available at: cdc.gov/tb/programs/laws/menu/caseid.htm#communicableDisease. Accessed June 14, 2021.
- Clough S, Shaw A, Morgan C. Tuberculosis and oral healthcare provision. Br Dent J. 2018;224:931–936.
- 18. United States Centers for Disease Control and Prevention. Tuberculosis: Health Care Settings. Available at: cdc.gov/tb/topic/infectioncontrol/default.htm. Accessed June 14, 2021.
- Su Y, Baena IG, Harle AC, et al. Tracking total spending on tuberculosis by source and function in 135 low-income and middle-income countries, 2000–17: a financial modelling study. Lancet Infect Dis. 2020;20:929–942.
From Dimensions of Dental Hygiene. July 2021;19(7):26-28, 31.




