Individualizing Care For Pregnant and Lactating Patients
Remaining up to date about the FDA’s Pregnancy and Lactation Labeling Rule is key to the provision of safe dental treatment.
Planning safe individualized care for pregnant and lactating patients requires up-to-date knowledge of current guidance from the United States Food and Drug Administration (FDA) on drug precautions and contraindication. To reduce risks associated with drugs administered for dental treatment, dental hygienists share in the responsibility to identify and discuss these implications with patients as they relate to planned treatment, including the appropriate integration of local anesthetics into patient care. To do this, oral health professionals traditionally referenced the long-standing “FDA Pregnancy and Lactation Drug Categories” in use since 1979. This reference, however, is no longer available.
Update to Drug Labeling for Pregnancy and Lactation
In 2015, the FDA implemented a change to the long-standing system for rating drug risks during pregnancy and lactation. In an effort to provide product information that is more meaningful to both patients and healthcare providers, the widely recognized system of letter-based risk categories (A, B, C, D, and X) on prescription and biological drug labels was replaced with a new format. The impetus for this change was to address concerns that the old letter system resulted in false assumptions about the actual meaning of the letters and left patients and providers ill-informed. 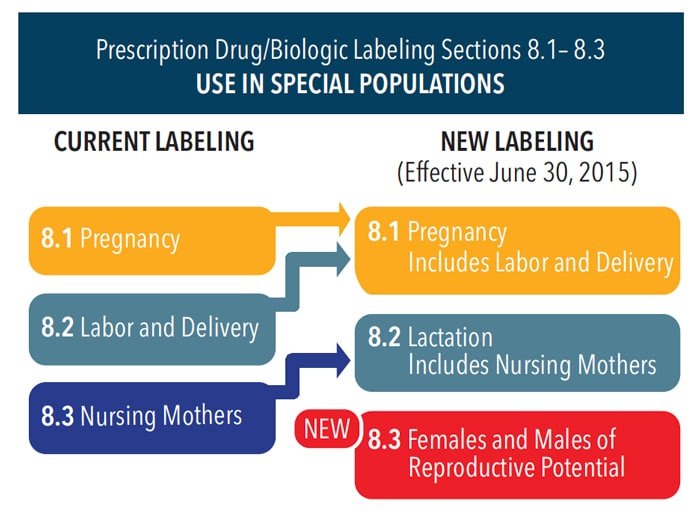
New Labeling Format
The new labeling system (Figure 1) is designed to facilitate better patient-centered counseling and informed decision making for pregnant individuals considering drugs recommended for their healthcare needs.1 Although the new labeling format does guide improved individualized care decisions, it still does not provide a definitive “yes” or “no” answer in most cases. Clinical interpretation is still required on a case-by-case basis. For example, selecting local anesthetics is not as simple as one of two “favored categories.” Each drug should be considered based on its unique pharmacological properties and pharmacokinetics, in addition to specific patient history.
While the new Pregnancy and Lactation Labeling Final Rule went into effect on June 30, 2015, the implementation timeline of changes in product directions for use—including drug labels and package inserts—varies (Table 1). The A, B, C, D, and X risk categories have been replaced with narrative presentations of information related to the use of a drug during pregnancy and lactation including the following:
- Risk summary
- Clinical considerations for use
- Supporting data
A lactation subsection provides information about using the drug while breastfeeding, including the amount of drug in breast milk and potential effects on the breastfed infant, and a subsection on reproductive potential for both men and women with information about:
- Pregnancy testing
- Contraception
- Infertility
All anesthetics currently available in dental cartridges were approved by the FDA prior to 2001. These companies will not be required to include the new Pregnancy and Lactation Labeling Final Rule risk sections, but they are required to remove any reference to the A, B, C, D, and X risk categories.
Conclusion
Oral health professionals should visit: fda.gov/drugs/labeling-information-drug-products/pregnancy-and-lactation-labeling-drugs-final-rule for additional information. The rule is continually updated with the most recent occurring on March 5, 2021.1
![TABLE 1. United States Food and Drug Administration (FDA) Pregnancy and Lactation Labeling Final Rule Implementation1]() Reference
Reference
- United States Food and Drug Adminstration. Pregnancy and Lactation Labeling (Drugs) Final Rule. Available at: fda.gov/drugs/labeling-information-drug-products/pregnancy-and-lactation-labeling-drugs-final-rule. Accessed June 14, 2022.
From Dimensions of Dental Hygiene. July 2022; 20(7)12.


