Hydroxyapatite Offers a New Option in Caries Prevention and Sensitivity Relief
This calcium and phosphate compound provides a safe and effective alternative to fluoride.
As patients seek alternatives to traditional fluoride-based products, oral health professionals are increasingly fielding questions about the best options for preventing dental caries and reducing tooth sensitivity.1 With ongoing public scrutiny of fluoride use, hydroxyapatite (HA) has emerged as a promising agent due to its unique properties. However, dental provider adoption of HA has been tempered by questions regarding efficacy, safety, and regulatory approval. The prudent clinician is tasked with navigating evidence-based research while considering patient preferences and ensuring efficacy for maximum clinical outcomes.
Fluoride
Fluoride remains the standard for caries prevention and enamel remineralization. Fluoride’s effectiveness is supported by years of research and regulatory endorsement from agencies such as the United States Centers for Disease Control and Prevention,US Food and Drug Administration (FDA), the American Dental Association (ADA), and the American Academy of Pediatrics.2,3
Fluoride forms a robust, acid-resistant fluorapatite layer on teeth, making the tooth structure stronger and more resistant to acid attacks. By increasing salivary fluoride concentration and raising salivary pH, fluorapatite inhibits the breakdown of the tooth surface.2,3 Standard recommendations vary by agent and are based on patient needs. Sodium fluoride (NaF) and sodium monofluorophosphate are used for caries prevention, while stannous fluoride is recommended for caries prevention, antimicrobial control, and sensitivity protection. Potassium nitrate can be combined with NaF for sensitivity management and caries protection.4
In 1997, the FDA placed warning labels on fluoride-containing toothpastes in response to concerns about fluoride toxicity in young children. In 2014, the ADA recommended lowering the amount of toothpaste to a rice-sized smear for children under age 3 and a pea-sized amount for older children and adults.5 However, some evidence-based research has questioned whether a rice-sized smear provides sufficient caries-preventive benefit.6
Excess ingestion of fluoride beyond the recommended dose can lead to an upset stomach, enamel fluorosis, and potential neurotoxic effects on the developing brains of children.3,7 The American Academy of Pediatrics affirms that extensive research validates the safety and efficacy of fluoride when following current dosing regimens. Additionally, community water fluoridation at 0.7 ppm is deemed safe from both fluorosis and adverse neurocognitive effects.8,9 Yet, patient preferences and public perception continue to drive interest in alternative options.
Hydroxyapatite
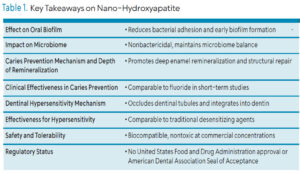
Calcium and phosphate are essential minerals present in bones and teeth as carbonated hydroxyapatite. HA is the main inorganic component of both enamel (97%) and dentin (70%). While bone and dentin share many similarities, enamel is considerably harder due to the composition, size, and crystalline structure of HA.10–12 Research shows that synthetic HA integrates into enamel and dentin defects, builds upon existing crystalline structures, forms a protective mineral layer on the tooth surface, supports ongoing deposition of calcium and phosphate from saliva, and enhances overall tooth hardness.1
Due to the biocompatibility and osteoconductivity of HA, synthetic HA has been explored for use in medical and dental applications since its development by the US National Aeronautics and Space Administration in the 1970s.1,10 A Japanese company acquired the patent and developed the first commercially marketed HA dentifrice. Japan formally recognized HA as an anticaries agent in 1993. Broader adoption in Europe, Canada, and the US has been slow to follow.3,6,7,12-14 In recent years, HA has gained popularity in dentistry and medicine. Current indications for use include bone grafts, tissue repair, sensitivity reduction, repairing and strengthening enamel, medication delivery, and coating dental implants.1,14,15
Synthetic HA is available in micro (mHA 5-10 microns) and nano (nHA 20-100 nanometers) particle forms. nHA is used more frequently in dental products because the size and structure more closely resemble natural HA , allowing deeper penetration into the enamel and dentin tubules than mHA.12
Currently, nHA is not FDA-approved for caries prevention in the US and does not carry the ADA Seal of Acceptance.16 The Journal of the American Dental Association addressed the ethical considerations of recommending HA products as alternatives to fluoride, noting professional ethics, such as veracity, autonomy, and nonmaleficence, must be integrated with standards of care and clinical practice guidelines. The ADA maintains that fluoride is safe, effective, and recommended for caries prevention, while acknowledging that alternative options, such as HA, may be offered to patients.16 Patients must be well informed that HA has not been as extensively researched as fluoride.1
Biofilm and the Microbiome
The oral microbiome maintains symbiosis with the host under healthy conditions. However, local and systemic factors can shift this balance toward dysbiosis, increasing the risk of caries or periodontal disease. In either condition, oral biofilm accumulation on the tooth surface and gingival tissues facilitates the disease process.17 Given its biocompatibility, HA represents a reasonable alternative or adjunct to traditional methods of biofilm control.12,13,18–20
Although literature is limited, HA binds to bacteria and proteins, disrupting the bacterial growth process. The smaller the particle size, the more surface area available for bacterial interaction, making nHA the preferred formulation. Compared to chlorhexidine, HA is as equally effective at disrupting biofilm accumulation.1,12,13,18,21
Caries Prevention
When combined with zinc, strontium, magnesium, or fluoride, nHa was more effective against S. mutans than nHA alone.22 Additionally, HA can decrease lactic acid production, an important factor in caries prevention.19,20 While fluoride acts primarily at the enamel surface, nHA can penetrate enamel lesions more deeply, inhibiting further demineralization. Remineralization of caries with a 10% HA toothpaste was as effective as 1,400 ppm stannous toothpaste in preventing caries progression.23 nHA can penetrate underlying tooth structure for effective enamel repair, sensitivity reduction, and restoration of surface smoothness, independent of salivary conditions.12,24–26 In comparison to mHA, nHA demonstrates more favorable biocompatibility and bioactivity, making it a safe and effective over-the-counter choice for remineralizing teeth and preventing caries as a fluoride-free option.12,24
Dentinal Hypersensitivity
Dentinal hypersensitivity (DH) is a common, multifactorial condition, typically occurring near the cementoenamel junction. Causes include toothbrush abrasion due to dentin exposure and demineralization of cementum from an acidic oral environment.7 While stannous fluoride and potassium nitrate provide relief from DH, nHA provides both caries protection and sensitivity reduction.27 nHA crystals adhere to the dentin surface, coating the surface and occluding dentinal tubules to prevent temperature or tactile stimuli from reaching the pulp, similar to that of stannous fluoride. One study found HA to be an effective desensitizer and, in some cases, superior to stannous fluoride.7 A 2019 meta-analysis identified nHA toothpaste as the best treatment for DH.28
Recommending Hydroxyapatite
Given its biocompatibility, ability to prevent biofilm adhesion, anti-caries properties, desensitizing effects, and capacity to maintain homeostatic balance within the oral cavity, nHA is an option for patients who prefer fluoride-free alternatives. While additional studies are needed, nHA serves as a favorable oral care agent to combat cariogenic bacteria and support oral health.12,14,18–20 Ultimately, nHA toothpaste is not definitively superior to fluoride, but stands as a strong alternative. The choice between fluoride and nHA should be guided by patient ingredient preference and specific dental needs, particularly dentinal hypersensitivity or caries risk.
Comparative Effectiveness and Safety
Oral health professionals have expressed concerns about nHA absorption and toxicity. In comparison to fluoride, which has been associated with dental fluorosis, bone weakening, and accidental poisoning from over-ingestion, available evidence indicates that HA is essentially nontoxic and nonimmunogenic.6,11,21,29,30 When ingested, HA particles are broken down by stomach acid into calcium and phosphate ions and easily eliminated; thereby making nHA a biocompatible option that is considered safe if swallowed and a preferable option for children.1
Nano-sized particles are preferred for their bioactivity, larger surface area, and crystalline structure closely resembling enamel.1,12,15,22 An in vitro study on nHA cytotoxicity found no harm to human gingival cells, specifically fibroblasts. Moreover, cells appeared normal and healthy, with no signs of damage, cell death, oxidative stress, or tissue irritation. nHA-treated cells were more metabolically active than untreated controls.31 Systemic cytotoxicity of nHA via oral epithelium exposure is considered unlikely due to cell turnover of nonkeratinized oral mucosa.14 However, another study found that inhaled nHA particles may interfere with pulmonary surfactant function, though lung cell damage was not observed. The risk of inhalation of nHA from currently available dental products is low.32
The shape of nHA particles warrants consideration. Different crystalline morphologies, H-sphere, H-needle, H-rod, and H-plate, vary in cytotoxicity. In vitro studies indicate H-plate shapes exhibit the highest and H-rod shapes the lowest levels of cytotoxicity.12,33 European Union guidelines classify rod-shaped nHA as safe in concentrations < 10% in dentifrices and < 0.465% in mouthrinses.34 While further research is needed, nHA morphology needs to be considered when recommending oral care products to patients.32,33
Products and Limitations
A variety of nHA oral care products are commercially available, including toothpastes, gels, mouthrinses, gums, and professionally applied varnishes. While marketed to promote remineralization, protect enamel, prevent caries, and decrease hypersensitivity, no nHA dental products currently carry FDA approval.3,12,13,21,29,35–41
Cost, limited manufacturing capacity, and variable regulatory oversight influence the production and availability of nHA. Inconsistencies among brands in particle size, shape, and concentration contribute to differences in product efficacy.40 The recommended over-the-counter concentration is 5% to 10% nHA for dentifrices and 0.465% for mouthrinses, yet commercially available products may fall outside these ranges due to limited regulatory oversight. While 10% is optimal for caries prevention and remineralization, higher concentrations provide no additional benefit.3,12,14,34,41 Because nHA requires more advanced manufacturing processes and lacks the global scale of fluoride production, raw material and manufacturing costs are higher. This additional cost cascades down to the consumer, reflecting retail prices three to five times higher than fluoride toothpaste.40
The body of research supporting nHA is limited in comparison to fluoride, contributing to hesitancy among clinicians.40 Best practices include reviewing product labels for nHA concentration, noting where the active ingredient appears in the ingredient list, and identifying pharmaceutical-grade designations. Dental professionals should strive to recommend reputable products supported by clinical studies and known to produce high-quality, appropriately concentrated nHA dental products.41
Conclusion
Synthetic HA, in either micro or nano formulations, serves as a suitable agent for caries prevention, treatment of hypersensitivity, and the prevention of biofilm adhesion without the potential toxicity concerns associated with fluoride ingestion (Table 1). Its biocompatibility makes nHA a viable option across all ages, particularly those who prefer nonfluoridated products. The higher production cost of nHA is a practical consideration for many patients, and best practice suggests that dental hygienists independently review scientific data to make evidence-based recommendations, regardless of marketing claims about specific products.
References
- Meyer F, Enax J, Amaechi BT, et al. Hydroxyapatite as remineralization agent for children’s dental care. Front Dent Med. 2022; 3:859560.
- Yeh CH, Wang YL, Vo TTT, Lee YC, Lee IT. Fluoride in dental caries prevention and treatment: mechanisms, clinical evidence, and public health perspectives. Healthc Basel. 2025;13(17):2246.
- Naim J, Sen S. The remineralizing and desensitizing potential of hydroxyapatite in dentistry: a narrative review of recent clinical evidence. J Funct Biomater. 2025;16:325.
- National Institutes of Health (NIH). Fluoride: Fact Sheet for Health Professionals. Availalbe at https://ods.od.nih.gov/factsheets/Fluoride-HealthProfessional. Accessed April 9, 2026.
- American Dental Association. Fluoride toothpaste use for young children clinical practice guideline. J Am Dent Assoc. 2014;145:190-191.
- Limeback H, Enax J, Meyer F. Biomimetic hydroxyapatite and caries prevention: a systematic review and meta-analysis. Can J Dent Hyg. 2021;55:148-159.
- Limeback H, Enax J, Meyer F. Clinical evidence of biomimetic hydroxyapatite in oral care products for reducing dentin hypersensitivity: an updated systematic review and meta-analysis. Biomim 2313-7673. 2023;8(1):23.
- American Academy of Pediatrics. Fact Checked: Fluoride is a Powerful Tool for Preventing Tooth Decay. Available at aap.org/en/news-room/fact-checked/fact-checked-fluoride-is-a-powerful-tool-for-preventing-tooth-decay/#:~:text=The%20AAP%2C%20the%20Centers%20for,%2C%20focus%2C%20and%20attend%20school. Accessed April 9, 2026.
- American Academy of Pediatric Dentistry. The Reference Manual of Pediatric Dentistry. Fluoride Therapy. Chicago: American Academy of Pediatric Dentistry; 2023:372-378.
- Dorozhkin SV, Epple M. Biological and medical significance of calcium phosphates. Angew Chem Int Ed Engl. 2002;41(17):3130-3146.
- Meyer F, Amaechi BT, Fabritius HO, Enax J. Overview of calcium phosphates used in biomimetic oral care. Open Dent J. 2018; 12:406-423.
- Chen L, Al-Bayatee S, Khurshid Z, Shavandi A, Brunton P, Ratnayake J. Hydroxyapatite in oral care products—a review. Mater 1996-1944. 2021;14:4865.
- O’Hagan-Wong K, Enax J, Meyer F, Ganss B. The use of hydroxyapatite toothpaste to prevent dental caries. Odontology. 2022;110:223-230.
- Pushpalatha C, Gayathri VS, Sowmya SV, et al. Nanohydroxyapatite in dentistry: A comprehensive review. Saudi Dent J. 2023;35:741-752.
- Juntavee A, Juntavee N, Hirunmoon P. Remineralization potential of nanohydroxyapatite toothpaste compared with tricalcium phosphate and fluoride toothpaste on artificial carious lesions. Int J Dent. 2021;2021:5588832.
- Ambrosino B. Ethical guidance for dentists about offering nonfluoridated toothpaste to patients who request it. J Am Dent Assoc. 2025;156:338-339.
- Marsh PD, Zaura E. Dental biofilm: ecological interactions in health and disease. J Clin Periodontol. 2017;44:S12-S22.
- Meyer F, Enax J. Hydroxyapatite in oral biofilm management. Eur J Dent. 2019;13:287-290.
- Huang Y, Han Q, Peng X, et al. Disaggregated nano-hydroxyapatite (dnhap) with inhibitory effects on biofilms and demineralization. J Dent Res. 2023;102:777-784.
- Luo W, Huang Y, Zhou X, et al. The effect of disaggregated nano-hydroxyapatite on oral biofilm in vitro. Dent Mater. 2020;36:e207-e216.
- Amaechi BT, AbdulAzees PA, Okoye LO, Meyer F, Enax J. Comparison of hydroxyapatite and fluoride oral care gels for remineralization of initial caries: a pH-cycling study. BDJ Open. 2021;6(1):1-7.
- Imran E, Cooper PR, Ratnayake J, Ekambaram M, Mei ML. Potential beneficial effects of hydroxyapatite nanoparticles on caries lesions in vitro-a review of the literature. Dent J. 2023;11:2.
- Schlagenhauf U, Kunzelmann K, Hannig C, et al. Impact of a non‐fluoridated microcrystalline hydroxyapatite dentifrice on enamel caries progression in highly caries‐susceptible orthodontic patients: A randomized, controlled 6‐month trial. J Investig Clin Dent. 2019;10:e12399.
- Mehrjoo M, Haghgoo R, Ahmadvand M. Effect of a nano-hydroxyapatite toothpaste on enamel erosive lesions of third molars induced by exposure to orange juice. Contemp Clin Dent. 2024;15:17-21.
- Grocholewicz K, Matkowska-Cichocka G, Makowiecki P, et al. Effect of nano-hydroxyapatite and ozone on approximal initial caries: a randomized clinical trial. Sci Rep. 2020;10:11192.
- Daas I, Badr S, Osman E. Comparison between fluoride and nano-hydroxyapatite in remineralizing initial enamel lesion: An in vitro study. J Contemp Dent Pract. 2018;19:306-312.
- Smith TL. Tooth remineralization agents: an evidence-based review to make informed patient recommendations. Available at todaysrdh.com/tooth-remineralization-agents-an-evidence-based-review-to-make-informed-patient-recommendations. Accessed April 9, 2026.
- Hu ML, Zheng G, Lin H, Yang M, Zhang YD, Han JM. Network meta-analysis on the effect of desensitizing toothpastes on dentine hypersensitivity. J Dent. 2019;88:103170.
- Bossù M, Saccucci M, Salucci A, et al. Enamel remineralization and repair results of biomimetic hydroxyapatite toothpaste on deciduous teeth: an effective option to fluoride toothpaste. J Nanobiotechnology. 2019;17:17.
- Anil A, Ibraheem WI, Meshni AA, Preethanath RS, Anil S. Nano-hydroxyapatite (nHAp) in the remineralization of early dental caries: a scoping review. Int J Environ Res Public Health. 2022;19:9.
- Coelho CC, Grenho L, Gomes PS, Quadros PA, Fernandes MH. Nano-hydroxyapatite in oral care cosmetics: characterization and cytotoxicity assessment. Sci Rep. 2019;9:N.PAG.
- Fan Q, Wang YE, Zhao X, Loo JSC, Zuo YY. Adverse biophysical effects of hydroxyapatite nanoparticles on natural pulmonary surfactant. ACS Nano. 2011;5:6410-6416.
- Huang L, Sun X, Ouyang J. Shape-dependent toxicity and mineralization of hydroxyapatite nanoparticles in A7R5 aortic smooth muscle cells. Sci Rep. 2019;9:18979.
- Scientific Committee on Consumer Safety. Opinion on Hydroxyapatite (Nano). Available at https://health.ec.europa.eu/publications/hydroxyapatite-nano-0_en. Accessed April 9, 2026.
- Patel D. Nano hydroxyapatite toothpaste market research report. Available at https://dataintelo.com/report/nano-hydroxyapatite-toothpaste-market. Accessed April 9, 2026.
- Amaechi BT, Alshareif DO, Azees PAA, et al. Anti-caries evaluation of a nano-hydroxyapatite dental lotion for use after toothbrushing: An in situ study. J Dent. 2021;115:103863.
- Cocco F, Salerno C, Wierichs RJ, et al. Hydroxyapatite-fluoride toothpastes on caries activity: a triple-blind randomized clinical trial. Int Dent J. 2025;75:632-642.
- Limeback H, Enax J, Meyer F. Improving oral health with fluoride-free calcium-phosphate-based biomimetic toothpastes: an update of the clinical evidence. Biomim Basel Switz. 2023;8:331.
- Hatfield S. Nano-hydroxyapatite varnish: does research support caries prevention efficacy and safety? Available at .todaysrdh.com/nano-hydroxyapatite-varnish-does-research-support-caries-prevention-efficacy-and-safety. Accessed April 9, 2026.
- Nano-Hydroxyapatite Downsides: Limitations and Important Considerations. Available at https://nanohydroxyapatite.org/article/nano-hydroxyapatite-downsides-limitations-considerations. Accessed April 9, 2026.
- Toothpaste with Highest Nano-Hydroxyapatite: Complete Concentration Guide. Available at https://nanohydroxyapatite.org/article/highest-nano-hydroxyapatite-toothpaste-concentration-guide. Accessed April 9, 2026.
From Dimensions of Dental Hygiene. May/June 2026; 24(3):16-21