 97/ISTOCK/GETTY IMAGES PLUS
97/ISTOCK/GETTY IMAGES PLUS
The Role of Attached Gingiva in Implant Therapy
While there is insufficient evidence to routinely recommend gingival augmentation to maintain implants, gingival grafting may benefit certain patient populations.
Historically, a wide zone of attached gingiva has been considered beneficial, as the gingiva plays an important role in maintaining the health of the periodontium. It provides a physical barrier to oral biofilm, dissipates masticatory forces, and protects the periodontium from injury.1 It also mitigates forces from frena pull,2 deepens the vestibule, and provides an environment conducive to successful self-care and biofilm control.3 Additionally, a wide zone of attached gingiva helps prevent gingival recession, root exposure, and root caries.4
The question of what constitutes an adequate zone of attached gingiva has been studied in clinical trials. Lang and Löe5 advocated a minimum of 2 mm of gingiva (1 mm free gingiva and 1 mm attached gingiva) to maintain gingival health, as teeth with less than 2 mm exhibited clinical signs of inflammation, even in the presence of good oral hygiene. In contrast, Bowers6 observed healthy sites with less than 1 mm of attached gingiva. Longitudinal clinical trials ranging from 5 years to 18 years demonstrated the importance of good oral hygiene, rather than the width (apico-coronally) of the attached gingiva.8–10 The subjects in these studies were young with a healthy periodontium, however, and were placed on rigorous plaque control regimens and maintenance protocols.
In an animal study, Ericsson and Lindhe11 assessed the clinical impact of subgingival restorations on teeth with a narrow zone of attached gingiva. They reported that the presence of biofilm in these areas may lead to attachment loss and gingival recession. In a human study, Stetler and Bissada12 demonstrated similar findings, reporting significantly higher gingival index scores compared with restored teeth with a wide zone of attached gingiva. Although the consensus of available reports supports the concept that periodontal attachment can be maintained longitudinally with proper plaque control— regardless of the width of the attached gingiva—research suggests the majority of patients do not perform consistent or proper plaque control, or comply with a regular periodontal maintenance schedule.13 Hence, the decision for gingival augmentation should be based on clinical evaluation and the specific needs of the patient.
The following conditions may help clinicians decide whether to incorporate gingival augmentation into treatment plans:
- Lack of attached gingiva preventing adequate plaque control, especially in areas in which the marginal tissue is composed of alveolar mucosa, as these can become chronically inflamed, and may further be compromised by a frenum pull or shallow vestibule14
- Progressing gingival recession seen during maintenance recare appointments15
- Expansion of the dental arch by orthodontic procedures that may relocate a tooth out of the bony housing, creating bony fenestrations, dehiscences, and subsequent gingival recession16
- Areas in which the margins of restorations will be placed subgingivally;12 these areas require the presence of 5 mm of gingiva (of which 2 mm is composed of free gingiva and 3 mm of attached gingiva)17
- Abutment teeth for a removable partial denture (in which the connector may irritate marginal gingival tissue)
Many surgical procedures are available to increase the zone of attached gingiva, including free gingival grafts18–20 (see the case history on page 16), subepithelial connective tissue grafts,21 acellular dermal matrices,22 and xenogenic collagen matrices.23
CASE HISTORY: FREE GINGIVAL GRAFT
The following case illustrates a free gingival graft procedure to increase the zone of keratinized gingiva around two mandibular implants.
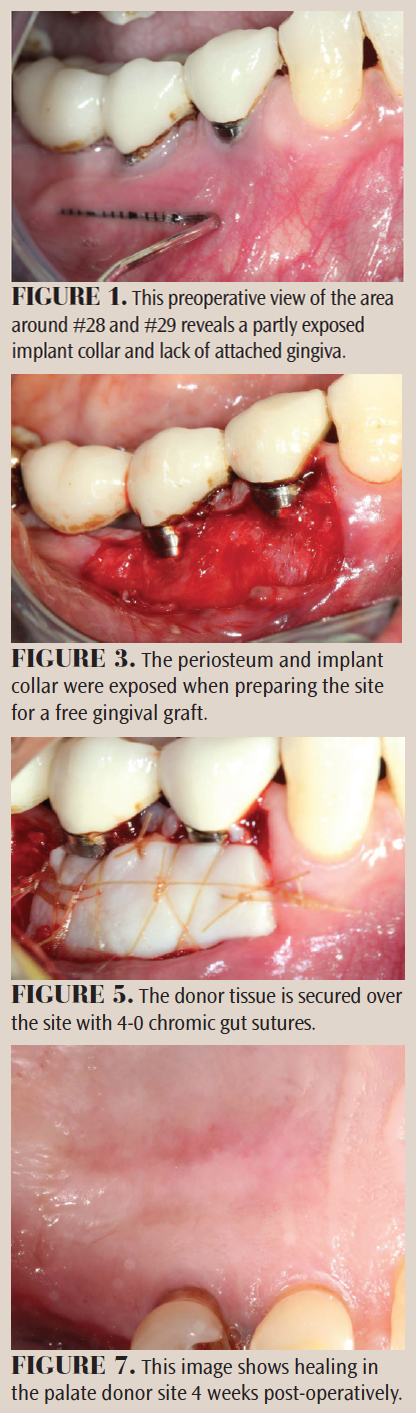
A 65-year-old man was referred for gingival grafting around dental implants in the area of #28 and #29. These implants had been placed 7 years prior. He had hypertension that was well con trolled with hydrochlorothiazide. The patient’s chief complaint was of tenderness and pain during toothbrush ing. He was also concerned about “thinning gums” around his implants.

A clinical examination revealed thin gingiva with a narrow band of keratinized mucosa, and gingival recession on the facial of implants #28 and #29 (Figure 1). A radiographic survey revealed well-integrated implants with mild crestal bone loss (Figure 2). The probing depths ranged from 3 mm to 4 mm. Several options were discussed, including risks and benefits. The patient consented to a free gingival graft on the facial aspect of the implants.

The recipient site was anesthetized with local infiltration of lidocaine with 1:100,000 epinephrine, and a partial thickness flap was reflected from the distal of #27 to the mesial of #30, exposing the periosteum (Figure 3). A free gingival graft measuring 18 mm by 10 mm was removed from the hard palate (Figure 4) and secured to the recipient site with chromic gut sutures (Figure 5). The graft was covered with a periodontal dressing.

The patient was issued oral and written post-operative instructions, and was seen several times over the following 2 months (Figure 6 and Figure 7). The patient responded well to the procedure. The zone of attached gingiva increased significantly in thickness and width. In addition, the periodontal tissue around the implants appeared healthy and asymptomatic.

ATTACHED GINGIVA AND IMPLANTS
Despite the wide acceptance of dental implants, the role of attached gingiva has not been established in implant dentistry. This is due, in part, to the differences in the soft tissue/implant interface (compared with natural dentition). The oral mucosa is attached to the implant surface by the junctional epithelium, but without the connective tissue attachment apical to it, as seen in natural dentition. The collagen fibers around an implant are parallel or oblique24 to its long axis, but are not embedded in the implant surface. Implants lack periodontal ligament and its rich vasculature, as well as the ability of the peri-implant mucosa to recover from bacterial or mechanical insults.25,26 Studies in animal models have demonstrated the difference in tissue response to bacterial challenge between implants and teeth. In the absence of plaque control, the inflammation was more severe and the infiltrate extended more apically around implants.27 Lindhe et al28 placed a ligature wire around dental implants in order to induce periodontitis and reported significant bone loss around implants compared to natural dentition. Warrer et al29 found that the absence of attached gingiva increases the susceptibility of the implant site to inflammation and bone loss. In a human study, Bouri et al30 examined the significance of attached gingiva in subjects with dental implants. The team found higher plaque index scores around implants with a narrow zone of attached gingiva (<2 mm) compared with implants with a wide zone (>2 mm). Subjects reported an inability to brush these areas due to mucosal mobility and tenderness. The implants with a narrow zone of attached gingiva had a higher incidence of bleeding on probing and exhibited more significant bone loss. Chung et al31 reported that lack of attached gingiva was associated with high plaque and gingival indices, especially on implants placed in posterior sextants.
Controversy also exists regarding the effect of keratinized gingiva on probing depths, clinical attachment levels, and bone loss around implants. Compared to areas with keratinized gingiva, several clinical trials associated its absence with a statistically significant increase in bone loss or attachment loss.32–34 In contrast, the studies of Roos-Jansåker et al35 and Chung et al31 did not support these findings. Furthermore, some studies found the absence of keratinized gingiva to be associated with a statistically significant increase in gingival recession.33,34 Schrott et al36 reported greater soft-tissue recession on buccal sites when the width of keratinized mucosa was <2 mm (compared with sites with >2 mm). These findings stand in contrast to studies by Bengazi et al37 and Chung et al31 that did not note significant effects. These differences could be due to variations in implant body surface finishes, such as rough vs smooth surfaces.38,39 While smooth implant surfaces were not negatively affected by the lack of attached gingiva, once bone is lost, implants with rough finishes harbor more plaque and proinflammatory factors.40–42
SUMMARY
As is evident from the literature, the role of attached gingiva in implant dentistry and its impact on long-term outcomes are controversial. A systematic review by Esposito et al43 recommends the need for more research in this area. Currently, there is insufficient evidence to routinely recommend gingival augmentation to maintain implants. That said, several clinical conditions could indicate situations in which gingival grafting might benefit implant patients.
- Areas of thin gingiva and progressing gingival recession, despite good oral hygiene and maintenance recare
- Sites that are a source of pain and discomfort during brushing
- Tissue mobility under overdentures44
- Coverage of exposed metal margins in the esthetic zone
Implantologists can choose many surgical procedures to manage these conditions, including pedicle grafts, free gingival grafts,45connective tissue grafts46 or acellular dermal matrices.47 These procedures can be performed prior to implant placement, during the second-stage surgery, or after delivery of the final prosthesis. As with all therapies, the final treatment plan will depend on the specific case and the patient’s wishes.
REFERENCES
- Miyasato M, Crigger M, Egelberg J. Gingival condition in areas of minimal and appreciable width of keratinized gingiva. J Clin Periodontol. 1977;4:200–209.
- Friedman N. Mucogingival surgery: The apically repositioned flap. J Perio. 1962;33:328–340.
- Nabers JM. Extension of the vestibular fornix utilizing a gingival graft — case history. Periodontics. 1966;4:77–79.
- Agudio G, Nieri M, Rotundo R, Franceschi D, Cortellini P, Pini Prato GP. Periodontal conditions of sites treated with gingival-augmentation surgery compared to untreated contralateral homologous sites: a 10- to 27-year long-term study. J Periodontol. 2009;80:1399–1405.
- Lang NP, Löe H. The relationship between the width of keratinized gingiva and gingival health. J Periodontol. 1972;43:623–627.
- Bowers GM. A study of the width of the attached gingiva. J Periodontol. 1963;34:201–209.
- Wennström JL. Lack of association between width of attached gingiva and development of soft tissue recession. A 5-year longitudinal study. J Clin Periodontol. 1987;14:181–184.
- Freedman AL, Green K, Salkin LM, Stein MD, Mellado JR. An 18-year longitudinal study of untreated mucogingival defects. J Periodontol. 1999;70:1174–1176.
- Kennedy JE, Bird WC, Palcanis KG, Dorfman HS. A longitudinal evaluation of varying widths of attached gingiva. J Clin Periodontol. 1985;12:667–675.
- Freedman AL, Salkin LM, Stein MD, Green K. A 10-year longitudinal study of untreated mucogingival defects. J Periodontol. 1992;63:71–72.
- Ericsson I, Lindhe J. Recession in sites with inadequate width of the keratinized gingival An experimental study in the dog. J Clin Periodontol. 1984;11:95–103.
- Stetler KJ, Bissada NF. Significance of the width of keratinized gingiva on the periodontal status of teeth with submarginal restorations. J Periodontol. 1987;58:696–700.
- Wilson TG Jr, Glover ME, Schoen J, Baus C, Jacobs T. Compliance with maintenance therapy in a private periodontal practice. J Periodontol. 1984;55:468–473.
- Gorman WJ. Prevalence and etiology of gingival recession. J Periodontol. 1967;38:316–322.
- Baker DL, Seymour GJ. The possible pathogenesis of gingival recession. A histological study of induced recession in the rat. J Clin Periodontol. 1976;3:208–219.
- Wennström JL, Lindhe J, Sinclair F, Thilander B. Some periodontal tissue reactions to orthodontic tooth movement in monkeys. J Clin Periodontol. 1987;14:121–129.
- Maynard JG Jr, Wilson RD. Physiologic dimensions of the periodontium significant to the restorative dentist. J Periodontol.1979;50:170–174.
- Bjorn H. Free transplantation of gingival propria. Odontol Revy. 1963;14:523.
- King K, Pennel B. Evaluation of attempts to increase the width of attached gingiva. Presented at the Philadelphia Society of Periodontology; 1964.
- Sullivan HC, Atkins JH. Free autogenous gingival grafts. 3. Utilization of grafts in the treatment of gingival recession. Periodontics. 1968;6:152–160.
- Langer B, Langer L. Subepithelial connective tissue graft technique for root coverage. J Periodontol. 1985;56:715–720.
- Wei PC, Laurell L, Geivelis M, Lingren MW, Maddalozzo D. Acellular dermal matrix allografts to achieve increased attached gingiva. Part 1. A clinical study. J Periodontol. 2000;71:1297–1305.
- Nevins M, Nevins ML, Kim SW, Schupbach P, Kim DM. The use of mucograft collagen matrix to augment the zone of keratinized tissue around teeth: a pilot study. Int J Periodontics Restorative Dent. 2011;31:367–373.
- Berglundh T, Lindhe J, Ericsson I, Marinello CP, Liljenberg B, Thomsen P. The soft tissue barrier at implants and teeth. Clin Oral Implants Res. 1991;2:81–90.
- Berglundh T, Lindhe J, Jonsson K, Ericsson I. The topography of the vascular systems in the periodontal and peri-implant tissues in the dog. J Clin Periodontol. 1994;21:189–193.
- Lindhe J, Berglundh T. The interface between the mucosa and the implant. Periodontol 2000. 1998;17:47–54.
- Ericsson I, Berglundh T, Marinello C, Liljenberg B, Lindhe J. Long-standing plaque and gingivitis at implants and teeth in the dog. Clin Oral Implants Res. 1992;3:99–103.
- Lindhe J, Berglundh T, Ericsson I, Liljenberg B, Marinello C. Experimental breakdown of peri-implant and periodontal tissues. A study in the beagle dog. Clin Oral Implants Res. 1992;3:9–16.
- Warrer K, Buser D, Lang NP, Karring T. Plaque-induced peri-implantitis in the presence or absence of keratinized mucosa. An experimental study in monkeys. Clin Oral Implants Res. 1995;6:131–138.
- Bouri A Jr, Bissada N, Al-Zahrani MS, Faddoul F, Nouneh I. Width of keratinized gingiva and the health status of the supporting tissues around dental implants. Int J Oral Maxillofac Implants. 2008;23:323–326.
- Chung DM, Oh TJ, Shotwell JL, Misch CE, Wang HL. Significance of keratinized mucosa in maintenance of dental implants with different surfaces. J Periodontol. 2006;77:1410–1420.
- Block MS, Kent JN. Factors associated with soft- and hard-tissue compromise of endosseous implants. J Oral Maxillofac Surg. 1990;48:1153–1160.
- Kim BS, Kim YK, Yun PY, et al. Evaluation of peri-implant tissue response according to the presence of keratinized mucosa. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2009;107:24–28.
- Zigdon H, Machtei EE. The dimensions of keratinized mucosa around implants affect clinical and immunological parameters. Clin Oral Implants Res. 2008;19:387–392.
- Roos-Jansåker AM, Renvert H, Lindahl C, Renvert S. Nine to fourteen-year follow-up of implant treatment. Part III: factors associated with peri-implant lesions. J Clin Periodontol. 2006;33:296–301.
- Schrott AR, Jimenez M, Hwang JW, Fiorellini J, Weber HP. Five-year evaluation of the influence of keratinized mucosa on peri-implant soft-tissue health and stability around implants supporting full-arch mandibular fixed prostheses. Clin Oral Implants Res. 2009;20:1170–1177.
- Bengazi F, Wennström JL, Lekholm U. Recession of the soft tissue margin at oral implants. A 2-year longitudinal prospective study. Clin Oral Implants Res. 1996;7:303–310.
- Adell R, Lekholm U, Rockler B, et al. Marginal tissue reactions at osseointegrated titanium fixtures (I). A 3-year longitudinal prospective study. Int J Oral Maxillofac Surg. 1986;15:39–52.
- Wennström JL, Bengazi F, Lekholm U. The influence of the masticatory mucosa on the peri-implant soft tissue condition. Clin Oral Implants Res. 1994;5:1–8.
- Abrahamsson I, Berglundh T, Wennström J, Lindhe J. The peri-implant hard and soft tissues at different implant systems. A comparative study in the dog. Clin Oral Implants Res. 1996;7:212–219.
- Van Steenberghe D. The influence of titanium abutment surface roughness on plaque accumulation and gingivitis: Short-term observations. Int J Oral Maxillofac Impl. 1996;11:169–178.
- Quirynen M, van der Mei HC, Bollen CM, et al. An in vivo study of the influence of the surface roughness of implants on the microbiology of supra- and subgingival plaque. J Dent Res. 1993;72:1304–1309.
- Esposito M, Grusovin MG, Maghaireh H, Coulthard P, Worthington HV. Interventions for replacing missing teeth: management of soft tissues for dental implants. Cochrane Database Syst Rev. 2007;18:CD006697.
- Adibrad M, Shahabuei M, Sahabi M. Significance of the width of keratinized mucosa on the health status of the supporting tissue around implants supporting overdentures. J Oral Implantol. 2009;35:232–237.
- Grover HS, Yadav A, Yadav P, Nanda P. Free gingival grafting to increase the zone of keratinized tissue around implants. Int J Oral Implantol Clinical Res. 2011;2:117–120.
- Wiesner G, Esposito M, Worthington H, Schlee M. Connective tissue grafts for thickening peri-implant tissues at implant placement. One-year results from an explanatory split-mouth randomized controlled clinical trial. Eur J Oral Implantol.2010;3:27–35.
- Yan JJ, Tsai AY, Wong MY, Hou LT. Comparison of acellular dermal graft and palatal autograft in the reconstruction of keratinized gingiva around dental implants: a case report. Int J Periodontics Restorative Dent. 2006;26:287–292.
From Dimensions of Dental Hygiene. May 2017;15(5):14, 16-17.

