
Xerostomia from the Beginning
We explore dry mouth’s etiology, symptoms, and methods of diagnosis.
This course was published in the October 2010 issue and expires October 31, 2013. The authors have no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Provide a definition of xerostomia.
- Discuss the five categories of saliva function.
- List the effects of xerostomia on quality of life.
- Discuss the other potential health risks associated with xerostomia.
- Understand the steps to making an accurate diagnosis of xerostomia.
 Although the definition of xerostomia—dry mouth due to lack of saliva—may be simple, the effects and etiology of this common oral complaint are far from it. Its effects on the oral cavity range from complete devastation, such as with methamphetamine abusers, to minor irritation that can be addressed with over-the-counter remedies. One of the most common complaints of the oral cavity, the diagnosis of xerostomia is often subjective and a myriad of health issues, lifestyle factors, and medical treatments may contribute to the condition.1-3 As such, dental hygienists need to be truly well-versed in the etiology, symptoms, and methods of diagnosis.
Although the definition of xerostomia—dry mouth due to lack of saliva—may be simple, the effects and etiology of this common oral complaint are far from it. Its effects on the oral cavity range from complete devastation, such as with methamphetamine abusers, to minor irritation that can be addressed with over-the-counter remedies. One of the most common complaints of the oral cavity, the diagnosis of xerostomia is often subjective and a myriad of health issues, lifestyle factors, and medical treatments may contribute to the condition.1-3 As such, dental hygienists need to be truly well-versed in the etiology, symptoms, and methods of diagnosis.
SALIVA
Xerostomia is the subjective complaint of dry mouth resulting from a decrease in the production of saliva, also known as hyposalivation. Whole saliva is a mixture of saliva from both the major and minor salivary glands and crevicular gingival fluid that suspends nonadherent oral bacteria and food debris.4 Composed of more than 99% water, saliva also contains sodium, potassium, calcium, magnesium, bicarbonate, and phosphate. Salivary function falls into five categories: lubrication and protection; buffering action and clearance; maintenance of tooth integrity; antibacterial activity; and taste and digestion.
Lubrication and protection: Saliva lubricates the oral cavity, facilitating mastication, swallowing, and speech—all while protecting the tissues from mechanical trauma. Saliva also protects the oral mucosa, gingivae, and teeth from harmful irritants and antimicrobial activity.
Buffering and clearance: The buffering and clearing action of saliva is attributed to the bicarbonate buffering system, although phosphate, urea, and amphoteric proteins and enzymes also contribute to the buffering action of saliva. The diffusion of bicarbonate ions into plaque neutralizes microbial acids through their buffering ability. As a result, ammonia is generated that subsequently forms amines, which serve as buffers.5 Ammonia is also released from urea after it has been metabolized by bacterial plaque, therefore increasing plaque pH and decreasing the likelihood of caries progression.6 The ability of salivary buffers to alter plaque pH is determined by plaque thickness and number of bacteria present.4 The buffering action of saliva is more effective during times of stimulated high salivary rates, as compared to periods of low flow with unstimulated saliva where its effects are nearly negligible.1,7 In other words, when reduced levels of saliva are present, the saliva’s inherent ability to buffer, which reduces the risk of caries, is hindered.
Maintenance of tooth integrity: Saliva supports the integrity of teeth by facilitating remineralization. This process of replacing lost minerals through the organic matrix of the enamel is provided by high, supersaturated concentrations of calcium and phosphate controlled by salivary proteins.7 Fluoride ions present within the saliva increase the rate of crystal precipitation. The assimilation of fluoride ion into enamel structure forms a fluorapatite-like coating that makes the tooth structure more resistant to caries.
Antibacterial activity: The components of saliva, such as the mucosal antibody immunoglobulin A (IgA) and the protein-based enzyme peroxidase, provide its antibacterial qualities, which protect the teeth from physical, chemical, and microbial attacks. Gingival crevicular fluid from within the parotid glands produce the enzymes—lysozymes—that are capable of destroying bacteria cell walls, in effect killing the bacteria. Saliva also inhibits cysteine-proteinase, an enzyme involved in periodontitis.8
 Taste and digestion: Saliva is capable of enhancing taste and is responsible for the initial steps of digestion.9 The composition of saliva augments the tasting capacity for nutritional sources and salty foods.4 Salivary amylase begins the initial breakdown of starch into smaller sugar molecules. The majority of starch breakdown occurs from the action of pancreatic amylase. Fat digestion also appears to be initiated in the oral cavity by salivary enzymes.10 The lubricating function of saliva, which aids in swallowing, is saliva’s most important contribution to the digestive system.4
Taste and digestion: Saliva is capable of enhancing taste and is responsible for the initial steps of digestion.9 The composition of saliva augments the tasting capacity for nutritional sources and salty foods.4 Salivary amylase begins the initial breakdown of starch into smaller sugar molecules. The majority of starch breakdown occurs from the action of pancreatic amylase. Fat digestion also appears to be initiated in the oral cavity by salivary enzymes.10 The lubricating function of saliva, which aids in swallowing, is saliva’s most important contribution to the digestive system.4
SYMPTOMS AND SEQUELAE
Xerostomia affects millions and its prevalence increases with age—affecting about 30% of those 65 years and older.11 Saliva is not required for any life-sustaining functions, however, a decrease or complete absence of saliva production can significantly affect people’s quality of life.12 Xerostomia may affect patients’ dietary habits, speech, mastication, as well as the ability to swallow (dysphagia), taste (dysgeusia), and retain food in the oral cavity. An aversion to dry foods, sore tongue (glossodynia), thirst, halitosis, oropharyngeal burning, and intolerance to spicy foods are also effects of xerostomia.1,2,12,13
Problems with speech and eating can affect patients’ ability to interact socially, which may cause them to avoid social situations.11 Xerostomia is also associated with mucositis, changes in the patient’s oral microbial flora, and the tongue sticking to the top of the palate. Patients may also experience difficulty wearing their oral prosthetics, development of denture sores, nighttime oral discomfort, and mucus and plaque accumulation. Patients presenting with xerostomia are at increased risk for hyposalivation-associated caries, periodontitis, tooth loss, and the development of oral and oropharyngeal infections, such as candidiasis.9
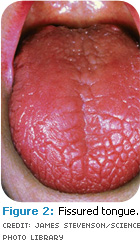
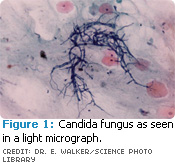 The opportunistic fungus candida albicans (Figure 1), which produces candidiasis, can manifest into other oral conditions such as erythema (erythematous candidiasis) of the oral mucosa, forming white adherent patches. The fungal infection can also lead to angular chelitis, a condition where the fissures at the corners of the mouth become inflamed. A furrowed, erythematous pebbled, cobblestoned, or fissured tongue (Figure 2) and complete or partial depapillation of the dorsum of the tongue are also characteristic of progressive xerostomia.9,14
The opportunistic fungus candida albicans (Figure 1), which produces candidiasis, can manifest into other oral conditions such as erythema (erythematous candidiasis) of the oral mucosa, forming white adherent patches. The fungal infection can also lead to angular chelitis, a condition where the fissures at the corners of the mouth become inflamed. A furrowed, erythematous pebbled, cobblestoned, or fissured tongue (Figure 2) and complete or partial depapillation of the dorsum of the tongue are also characteristic of progressive xerostomia.9,14
The prevalence of dental caries is also related to salivary flow.15,16 Those presenting with xerostomia exhibit recurrent decay at cervical margins (class V), incisal margins, and cusp tips (class VI).9 These class V and VI lesions are seen predominantly in older adults mainly because patients are retaining their natural or previously restored teeth longer than ever before.9
Occlusal pathology and associated noncarious cervical lesions occur at sites unprotected by saliva.17 Patients, however, are usually unaware of a decrease in salivary flow until they present with one or more of these complications. Consequently, patients don’t usually seek treatment from a dental practitioner until the development of symptoms.14
DIAGNOSIS
The diagnosis of xerostomia is based on clinical examination and thorough medical, drug, and family histories.9 Dental practitioners can facilitate the diagnostic process by asking specific questions. Table 1 provides some examples.9 A correct response to any of the questions may indicate a decrease in patient salivary flow.18,19 However, a complaint of oral dryness is not always a direct indicator of salivary gland dysfunction.20
Successful clinical diagnosis of xerostomia in patients with salivary gland hypofunction is possible with evidence of four indicators: 1) dry lips; 2) buccal mucosal dryness; 3) delayed or absence of salivation on palpation; 4) high total score on the decayed, missing, or filled teeth index (DMFT).4 If a patient has all of these factors, further diagnostic evaluations, such as measurements of salivary flow rate (sialometry), biopsy of minor salivary glands, and/or sialography, should be performed.18
In patients with xerostomia, external palpitation of the submandibular and parotid glands may cause delayed or no salivary flow. Palpitation of oral mucosa may cause the adherence of the finger to the oral mucosa as opposed to having the finger slide over the tissue.14
 Great variability exists in individual salivary flow rates. Ideally a base reference should be recorded after the age of 15 years.21 Due to this large variability, it is difficult to objectively evaluate salivary gland dysfunction in patients complaining of xerostomia.14 Sialometric analysis of unstimulated saliva can be performed by having the patient dribble saliva into a measuring vessel for a specified amount of time (between 3 minutes and 5 minutes). Likewise, stimulated saliva flow rates can be evaluated by having the patient chew unflavored wax or unflavored chewing gum base during the specified collection period.9 Unstimulated salivary flow rates of <0.1 ml/min are considered to be diagnostic of xerostomia/hypofunction.4 In the case where individualized salivary base rates have been confirmed, a 50% reduction in salivary flow should be considered hypofunction.22 A 50% reduction in salivary flow will also cause a patient to experience symptoms of dry mouth.23 Xerostomia is a subjective experience. A patient with oral mucosa that appears moist and a salivary flow rate >0.15 ml/min may still report a dry feeling in his or her mouth.
Great variability exists in individual salivary flow rates. Ideally a base reference should be recorded after the age of 15 years.21 Due to this large variability, it is difficult to objectively evaluate salivary gland dysfunction in patients complaining of xerostomia.14 Sialometric analysis of unstimulated saliva can be performed by having the patient dribble saliva into a measuring vessel for a specified amount of time (between 3 minutes and 5 minutes). Likewise, stimulated saliva flow rates can be evaluated by having the patient chew unflavored wax or unflavored chewing gum base during the specified collection period.9 Unstimulated salivary flow rates of <0.1 ml/min are considered to be diagnostic of xerostomia/hypofunction.4 In the case where individualized salivary base rates have been confirmed, a 50% reduction in salivary flow should be considered hypofunction.22 A 50% reduction in salivary flow will also cause a patient to experience symptoms of dry mouth.23 Xerostomia is a subjective experience. A patient with oral mucosa that appears moist and a salivary flow rate >0.15 ml/min may still report a dry feeling in his or her mouth.
To confirm the subjective complaint of xerostomia, other methods of examination are available including the imaging modalities of scintigraphy and sialography. Sialographic studies involve the injection of a radiopaque dye into the major salivary glands to determine if salivary gland obstruction and/or damage is present, which is often seen in patients with Sjogren’s syndrome.9,24 To assess salivary gland function, quantitative salivary scintigraphy can be used.9 A biopsy from the lower lip may be helpful in the diagnosis of Sjogren’s syndrome if the biopsy reveals focal lymphocytic infiltrates in the minor salivary glands.24 Biopsy of minor salivary glands is also required to diagnosis salivary gland neoplasms.9 The use of fine needle aspiration can aid in the identification of malignant lesions.14
Effective management and treatment of xerostomia cannot begin without an accurate diagnosis. Dental practitioners must be familiar with the symptoms of xerostomia so they can note the presence of hyposalivation in patients before the deleterious effects of this condition are sustained.
REFERENCES
- Nederfors T. Xerostomia and hyposalivation. Adv Dent Res. 2000;14:48-56.
- Guggenheimer J, Moore PA. Xerostomia: etiology, recognition and treatment. J Am Dent Assoc. 2003;134:61-9.
- Sreebny LM. Saliva in health and disease: an appraisal and update. Int Dent J. 2000;50:140-161.
- Humphrey SP, Williamson RT. A review of saliva: normal composition, flow and function. J Prosthet Dent. 2001;85:162-169.
- Mandel ID. The functions of saliva. J Dent Res. 1987;66:623-627.
- Johnson DA. Regulation of salivary glands and their secretions by masticatory, nutritional and hormonal factors. In: Scribney LM, ed. The Salivary System. Boca Raton, Fla: CBC Press; 1987:136- 155.
- Roth G, Calmes R, eds. Salivary glands and saliva. In: Oral Biology. St. Louis: Mosby; 1981:196-236.
- Slomiany BL, Murty VL, Poitrowski J, Slomiany A. Salivary mucins in oral mucosal defense. Gen Pharmacol. 1996;27:761-771.
- Gupta A, Epstein JB, Sroussi H. Hyposalivation in elderly patients. J Can Dent Assoc. 2006; 72: 841–846.
- Valdez IH, Fox PC. Interactions of the salivary and gastrointestinal systems. I. The role of saliva in digestion. Dig Dis. 1991;9:125-132.
- Ship JA, Pillemer SR, Baum BJ. Xerostomia and the geriatric patient. J Am Geriatr Soc. 2002;50:535–543.
- Porter SR. Xerostomia: prevalence, assessment, differential diagnosis and implications for quality of life. Oral Dis. 2010;16:501-502.
- Rayman S, Dincer E, Almas K. Xerostomia. Diagnosis and management in dental practice. NY State Dent J. 2010;76:24-27.
- Guggenheimer J, Moore PA. Xerostomia: etiology, recognition and treatment. J Am Dent Assoc. 2003;134:61-69.
- Papas AS, Joshi A, MacDonald SL, Maravelis –Splagounias L, Pretara-Spanedda P, Curro FA. Caries prevalence in xerostomic individuals. J Can Dent Assoc. 1993;59:171-174, 177-179.
- Younger H, Harrison T, Streckfus C. Relationship among stimulated whole, glandular salivary flow rates and root caries prevalence in an elderly population: a preliminary study. Spec Care Dentist. 1998;18:156-163.
- Young WG, Khan F. Sites of dental erosion are saliva-dependent. J Oral Rehab. 2002;29:35-43.
- Navazesh M, Kumar SK. Xerostomia: prevalence, diagnosis, and management. Compend Contin Educ Dent. 2009;30:326-328, 331-332.
- Atkinson JC, Grisius M, Massey W. Salivary hypofunction and xerostomia: diagnosis and treatment. Dent Clin North Am. 2005;49:309-326.
- Diaz-Arnold AM, Marek CA. The impact of saliva on patient care: a literature review. J Prosthet Dent. 2002;88:337-343.
- Edgar WM. Saliva and dental health. Clinical implications of saliva: report of a consensus meeting. Br Dent J. 1990;169: 96-98.
- Ghezzi EM, Lange LA, Ship JA. Determination of variation of stimulated salivary flow rates. J Dent Res. 2000;79:1874-1878.
- Dawes C. How much saliva is enough for avoidance of xerostomia? Caries Res. 2004;38:236-240.
- Vitali C, Bombardieri S, Jonsson R, et al. Classification criteria for Sjögren’s syndrome: a revised version of the European criteria proposed by the American-European Consensus Group. Ann Rheum Dis. 2002;61:554-548.
From Dimensions of Dental Hygiene. October 2010; 8(10): 72-75.



