The Dental Hygienist’s Role in Treating Gingival Recession
Proper prevention, treatment, and maintenance strategies are integral to achieving positive patient outcomes.
This course was published in the May 2022 issue and expires May 2025. The authors have no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Explain site- and patient-related risk factors for mucogingival therapy.
- Discuss the drivers for covering exposed root surfaces or treating deficient bands of attached, keratinized tissue.
- Educate patients on proper self-care practices when there is existing recession.
- Manage dentinal hypersensitivity as part of recare visits.
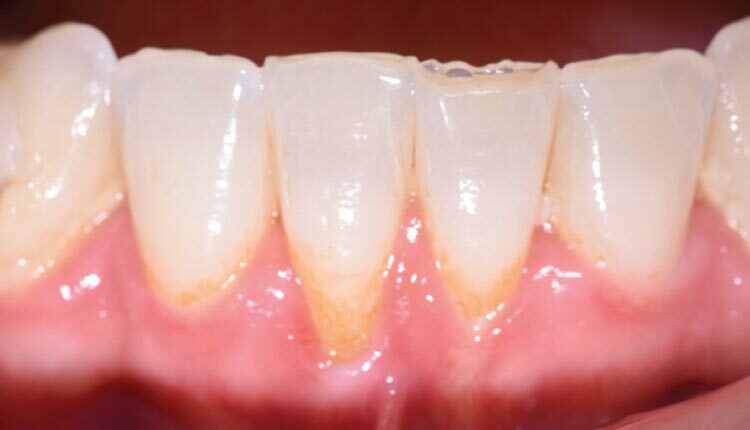
A lack of attached, keratinized tissue and gingival recession are the most common soft tissue conditions encountered by the dental team. In fact, 50% to 80% of adults present with at least one site of recession.1 Exposed root surfaces compromise esthetics and function, leading to an unfortunate sequela of dentinal hypersensitivity, caries, noncarious cervical lesions, and/or impaired plaque control. If these defects are not treated aptly, they can worsen and complicate future surgical, restorative, or orthodontic treatment.2 The surgical approach is unique to each patient and differs by etiology of the condition, oral hygiene compliance, and other risk factors. Dental hygienists need to understand preventive and maintenance practices to prevent and manage gingival recession.
Gingival recession is the apical shift of the gingival margin from the cementoenamel junction.3 Recession can result from bacterial, anatomical, chemical, or mechanical trauma, but the presence of inflammation and bone loss are what truly drive this process. A combination of these factors worsens recession over time (Table 1).3 These factors do not cause recession by themselves, but rather predispose patients to its development.
The influences of a patient’s phenotype, which considers hard and soft tissue thickness, are also considered. Dehiscence and recession more frequently occur in patients with thin phenotypes, especially after orthodontic treatment.4 If a probe is visualized through the facial gingival sulcus, it is classified as thin.5 This should be documented as part of the initial workup, as it may complicate treatment.
Today, a variety of materials and techniques are used to minimize recession. A systematic review of 48 randomized clinical trials with short- and long-term follow-ups found that connective tissue grafts combined with coronally advanced flaps best managed single and multiple recession-type defects with the most significant gain in attachment, keratinized tissue width, and complete root coverage.6 This is often considered the gold standard of root coverage procedures. In this same study, chemical root modification and the type of mechanical debridement did not affect therapeutic outcomes.6 Different chemical agents, including ethylenediaminetetraacetic acid (EDTA), can be used to decontaminate the root surface, remove the smear layer, and expose the dentin tubules for improved connective tissue re-attachment.7 They can, however, be cytotoxic to wound healing cells at specific concentrations, and should be applied in the appropriate indications.7
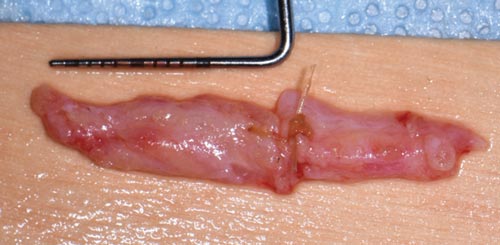
The connective tissue graft is harvested from the palate in a single incision, underneath the overlying epithelium, or de-epithelialized after removal from the donor site. It is then introduced to the recipient site as part of a coronally advanced or rotated flap or tunnel (Figure 1 and Figure 2).6 The use of nonautogenous materials, such as allografts or xenografts, provide viable alternatives to the connective tissue graft to reduce patient comorbidities.6 Table 2 includes a list of available materials and techniques.
Growth factors can also be used to accelerate healing. They stimulate the chemotaxis, mitogenesis, and matrix formation of wound-healing cells.6 Another molecular mediator, enamel matrix derivative, contains different proteins and enzymes from developing pig enamel and enhances fibroblast recruitment and adhesion and angiogenesis.8 These products can hydrate grafts and matrices or directly at the recipient site.
Dental caries, lesions, or cervical restorations may affect root coverage procedures. Noncarious cervical lesions (NCCLs) vary in appearance and result from abrasion, erosion, and corrosion from marginal restorations. The decision to graft or restore NCCLs is often debated. If the lesion extends more than 2 mm into the root surface, it should be restored.9 The lesion’s depth and shape, root convexity, thickness, amount of attached tissue present, and esthetic interests should all be considered.9 Root reshaping with burs or hand instruments shortens the distance cells need to travel for graft vascularization.10 NCCLs, root caries, and defective restorative margins should be diagnosed and treated before surgery.
The Role of Keratinized Tissue
If left untreated, recession can worsen to the depth of the mucogingival junction and compromise the band of attached, keratinized tissue. The 2017 World Workshop on the Classification of Periodontal and Peri-Implant Diseases and Conditions did not numerically specify the amount or thickness of keratinized tissue needed to maintain health. However, tissue that is not inflamed, minimizes further recession, and supports patient comfort, esthetics, and oral hygiene should be considered healthy.11 With this in mind, continued recession is an important driver for treatment.
A wide band of keratinized tissue is protective against future recession, more so in patients with poor oral hygiene who are noncompliant with their recare schedule. This was observed in a split-mouth study, where grafted sites that were not maintained for 5 years were still free of inflammation.12 The amount of keratinized tissue at the initial recare visit was recently considered one of the most important indicators of long-term marginal stability according to a network meta-analysis.13 Keratinized tissue width should therefore be recorded as part of a comprehensive evaluation. This can be manually charted or imported with an intraoral scanner.
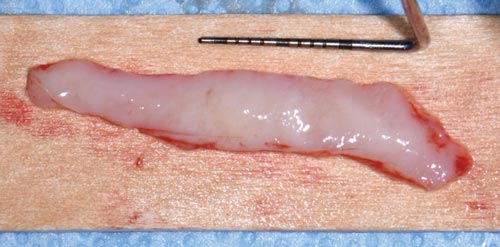
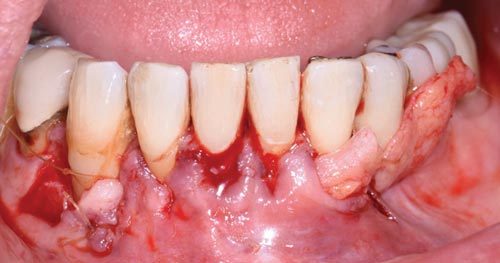
In cases where an increased gain in keratinized tissue width is the desired goal, a free soft tissue autograft or free gingival graft is the preferred treatment.14 A 1-mm to 2-mm thick graft that includes epithelium and connective tissue is harvested from the palate. The donor site is sutured with a hemostatic agent, and a plastic palatal stent is delivered for immediate hemostasis. The graft is then transplanted and secured to a recipient site deficient in attached, keratinized tissue (Figure 3, to Figure 5). Approximately 44% to 58% shrinkage is expected over the grafted area, mainly in the first 30 days.15 This is commonly performed at low esthetic-risk sites. Nonautogenous materials also work well when there is concern for donor site anatomy, post-operative discomfort, or treatment of multiple defects.16
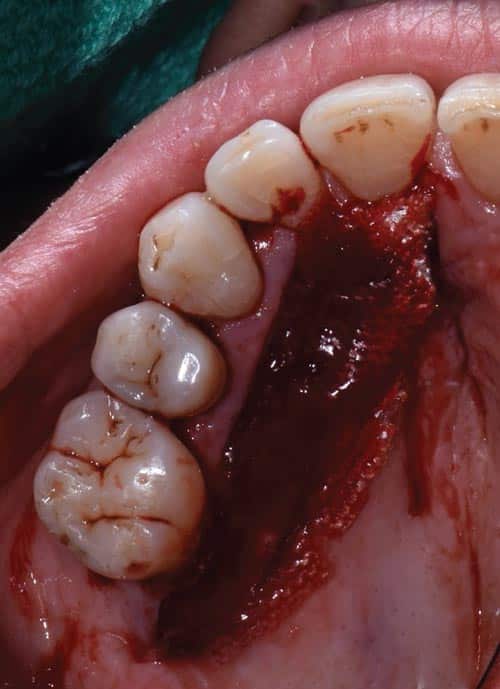
Patients should not initially anticipate root coverage with this procedure. A phenomenon called creeping attachment does however occur from 1 month to 1 year after surgery. The coronal migration of the gingival sulcus is about 0.8 mm on average and can, in turn, cover exposed roots, especially in areas of narrow, shallow recession.17 Setting appropriate clinical expectations is important. Tooth malposition, high frenum attachment, strong muscle pull, shallow vestibule, and other anatomical factors, can predispose patients to mucogingival problems and negatively affect therapeutic outcomes (Figure 6).
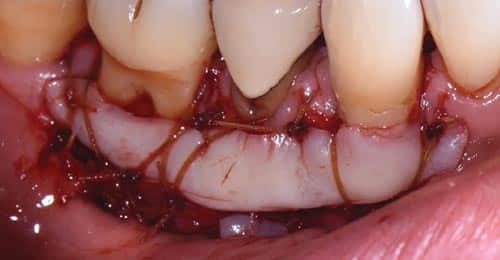
The timing of any orthodontic, restorative, and/or implant treatment should be outlined in the treatment planning phase. For example, gingival augmentation may be needed to minimize recession when teeth are moved beyond their normal alveolar housing to establish proper occlusal relations.4 A best-evidence consensus showed that 20% to 25% of patients develop mucogingival defect(s) within 5 years of orthodontic treatment.4 While recession can at least be maintained with regular prophylaxis and proper self-care, patients must understand the treatment objectives and potential complications of interdisciplinary care. They should also be alerted to the added time and expense required at their first appointment.
Prevention and Maintenance of Recession
As part of the initial recall visit, oral hygiene technique must be reviewed. Studies show that improper toothbrushing can harm gingival tissues with brushing pressure and bristle type serving as the most important determinants.18 In fact, in the vast majority of cases of dentinal hypersensitivity, a hard bristle toothbrush was used.19 Patients should instead use a soft toothbrush or a power toothbrush with a pressure indicator. Evidence suggests these tools help minimize aggressive brushing patterns.2 Technique and angulation affect plaque removal regardless of the toothbrush type. Proper oral hygiene reduces bacterial load and inflammation, which in turn, lessens discomfort and attachment loss.2
Selecting the proper dentifrice is equally important in patients with sensitivity concerns. The relative dentin abrasivity (RDA) value measures the abrasiveness of toothpaste on dentin. RDA is directly related to dentin loss, which contributes to dentinal hypersensitivity. Although this presentation is multifactorial, using a toothpaste with a low RDA can help minimize its symptoms and progression.19 Toothpaste with a RDA value of 0 to 70 is categorized as “low abrasion” and should be recommended to patients with existing sensitivity or exposed root surfaces.19
Patients should also consider a toothpaste with ingredients such as calcium sodium phosphosilicate, arginine and calcium carbonate, strontium, or potassium nitrate. These ingredients depolarize the nerves or block dentinal tubules to prevent fluid movement, thereby decreasing nerve conduction and hypersensitivity.2 Because of the different mechanisms by which sensitivity-relief toothpastes work, patients may need to try several first or use them for weeks at a time before finding one that alleviates their symptoms. For some patients, a high-fluoride concentration toothpaste may also be beneficial.19
When surgery is pursued, patients must establish regular recare appointments, in addition to the recommended post-operative follow-ups with their specialist. Significantly greater root coverage and better long-term marginal stability were achieved in patients who followed up with the treating surgeon compared to the general dentist.20 Furthermore, evidence suggests patients who have undergone surgery benefit from an alternating maintenance schedule between their general dentist and periodontist.20 At each visit, a periodontal exam, prophylaxis, and evaluation by the dentist should take place. The aforementioned tools and habits should be reinforced, and regular maintenance and proper oral hygiene need to be stressed as keys to long-term treatment success.
If patients are not surgical candidates due to medical or financial limitations, their symptoms can still be managed during recare visits. Topical fluoride varnish application provides longer relief than the potassium nitrate found in many sensitivity-relief toothpastes.2 Laser therapy is a more novel approach to relieving hypersensitivity and includes low- and mid-output power lasers, such as diode, Nd:YAG, Er:YAG, and CO2.19 The exact mechanism is unknown. Each laser affects the tissues differently depending on its wavelength, power density, and mode. It is assumed that they provide an analgesic effect by occluding the dentinal tubules when used directly over the tooth surface.21 However, a weak association exists with this technology given its strong placebo effect in which patients feel immediate relief afterward.22 With the proper training and state credentialing, dental hygienists can perform this procedure.
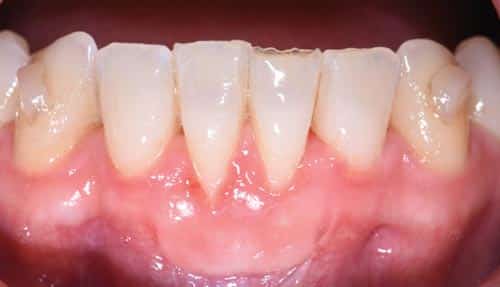
Case Report
This case outlines the steps taken to correct a complete lack of attached tissue thickness and amount by means of a free gingival graft. A 29-year old woman was concerned about worsening recession (Figure 7 to Figure 9). The patient had no significant medical history. After a thorough patient evaluation and review of varying techniques and materials, a surgical treatment plan was developed, and consents were obtained. The treatment plan included a free gingival graft #24, #25 area using an enamel matrix derived growth factor and a coronally repositioned flap to obtain root coverage as needed.23
After initial findings were charted both manually and with an intraoral scan, an increase in keratinized tissue width was determined to be the primary goal. The patient had a shallow vestibule, absent attached tissue around #24 and #25, thin phenotype, labial tooth position #25 due to ongoing orthodontic treatment, and fair oral hygiene. Complete root coverage was not initially guaranteed because of these site-related risk factors.
The recipient site was carefully prepared with partial-thickness dissection from #24-#25 and distal oblique vertical releases. A free gingival graft measuring 8 mm long x 10 mm to 12 mm wide x 1.5 mm thick was harvested from the right palate, trimmed to remove excess adipose tissue, and hydrated in an enamel matrix derived growth factor. EDTA root surface conditioner with a concentration of 24% was used over #24, #25 before applying the growth factor. The graft was secured and stabilized at the #24, #25 area with simple interrupted, nonresorbable sutures. Gauze pressure was applied to the palatal wound and multiple layers of hemostatic gauze were placed and sutured along the donor site for profound hemostasis. A palatal stent was then delivered to further minimize her post-operative bleeding risk, and a periodontal dressing was placed over the recipient site. The patient was discouraged from looking at or manipulating the operated sites for at least 2 weeks.
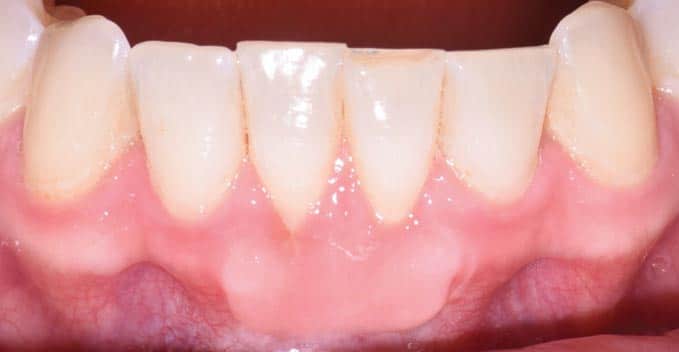
The patient returned for weekly follow-ups and was initially maintained by the specialty practice. Proper oral hygiene techniques were demonstrated at each visit to establish better self-care. A power toothbrush with a pressure indicator was recommended. The recipient site was scaled and polished as needed. Further root coverage can be appreciated during this time with the gradual and coronal reformation of the gingival sulcus. She will finish her orthodontic treatment and alternate prophylaxis every 6 months with her general dentist and periodontist. She denied any esthetic or sensitivity concerns following treatment.
Summary and Conclusions
Procedures to cover exposed roots or increase keratinized tissue width have evolved over time. Site- and patient-related risk factors should be discussed as part of the initial exam, especially if any interdisciplinary treatment is planned. Gingival recession can be treated successfully when identified early on. Esthetics, sensitivity, and worsening recession are important drivers for surgical treatment. Dental hygienists play an important role in the prevention and maintenance of mucogingival defects through patient education and regular prophylaxis.
References
- Kassab MM, Cohen RE. The etiology and prevalence of gingival recession. J Am Dent Assoc. 2003;134:220–225.
- Imber JC, KasaJ A. Treatment of gingival recession: When and how? Int Dent J. 2021;71:178–187.
- Cortellini P, Bissada NF. Mucogingival conditions in the natural dentition: narrative review, case definitions, and diagnostic considerations. J Periodontol. 2018;89(Suppl 1):S204– S213.
- Kao RT, Curtis DA, Kim DM, et al. American Academy of Periodontology best evidence consensus statement on modifying periodontal phenotype in preparation for orthodontic and restorative treatment. J Periodontol. 2020;91:289–298.
- De Rouck T, Eghbali R, Collys K, De Bruyn H, Cosyn J. The gingival biotype revisited: transparency of the periodontal probe through the gingival margin as a method to discriminate thin from thick gingiva. J Clin Periodontol. 2009;36:428–433.
- Chambrone L, Salinas Ortega MA, Sukekava F, et al. Root coverage procedures for treating localised and multiple recession-type defects. Cochrane Database Syst Rev. 2018;10:CD007161.
- Blomlöf JP, Blomlöf LB, Lindskog SF. Smear layer formed by different root planing modalities and its removal by an ethylenediaminetetraacetic acid gel preparation. Int J Periodontics Restorative Dent. 1997;17:242–249.
- Gestrelius S, Lyngstadaas SP, Hammarström L. Emdogain- periodontal regeneration based on biomimicry. Clin Oral Investig. 2000;4:120–125.
- Allen EP, Winter RR. Interdisciplinary treatment of cervical lesions. Compend Contin Educ Dent. 2011;32:16–20.
- Gordon HP, Sullivan HC, Atkins JH. Free autogenous gingival grafts. II. Supplemental findings—histology of the graft site. Periodontics. 1968;6:130–133.
- Cortellini, P, Bissada, NF. Mucogingival conditions in the natural dentition: Narrative review, case definitions, and diagnostic considerations. J Periodontol. 2018;89(Suppl 1):S204–S213.
- Kennedy JE, Bird WC, Palcanis KG, Dorfman HS. A longitudinal evaluation of varying widths of attached gingiva. J Clin Periodontol. 1985;12:667–675.
- Tavelli L, Barootchi S, Cairo F, Rasperini G, Shedden K, Wang HL. The effect of time on root coverage outcomes: a network meta-analysis. J Dent Res. 2019;98:1195–1203.
- Kim DM, Neiva R. Periodontal soft tissue non-root coverage procedures: a systematic review from the AAP regeneration workshop. J Periodontol. 2015;86:S56–S72.
- Silva CO, Ribeiro Edel P, Sallum AW, Tatakis DN. Free gingival grafts: graft shrinkage and donor-site healing in smokers and non-smokers. J Periodontol. 2010;81:692–701.
- Dai A, Huang JP, Ding PH, Chen LL. Long‐term stability of root coverage procedures for single gingival recessions: a systematic review and meta‐analysis. J Clin Periodontol. 2019;46:572– 585.
- Matter J, Cimasoni G. Creeping attachment after free gingival grafts. J Periodontol. 1976;47:574–579.
- Addy M, Hunter ML. Can toothbrushing damage your health? effects on oral and dental tissues. Int Dent J. 2003;53:177–186.
- Clark D, Liran L. Nonsurgical management of tooth hypersensitivity. Int Dent J. 2016;66:249–256.
- McGuire MK, Scheyer ET, Snyder MB. Evaluation of recession defects treated with coronally advanced flaps and either recombinant human platelet-derived growth factor-BB plus β-tricalcium phosphate or connective tissue: comparison of clinical parameters at 5 years. J Periodontol. 2014;85:1361–1370.
- Matsumoto K, Kimura Y. Laser therapy of dentin hypersensitivity. J Oral Laser Application. 2007;7:7–25.
- Sgolastra F, Petrucci A, Gatto R, Monaco A. Effectiveness of laser in dentinal hypersensitivity treatment: a systematic review. J Endod. 2011;37:297–303.
- Bernimoulin JP, Lüscher B, Mühlemann HR. Coronally repositioned periodontal flap. Clinical evaluation after one year. J Clin Periodontol. 1975;2:1–13.
From Dimensions of Dental Hygiene. May 2022;20(5):32-35.



