
An In-depth Look at Air Polishing
Dental hygienists need to understand how air polishing technology works and the proper technique for application in order to produce positive outcomes in patient care.
Air polishing has been available for four decades, however, the concept is based on a technology invented by Dr. Robert Black in 1945. Dr. Black invented the Air Dent, a device that used compressed air, water, and a highly abrasive powder to eliminate pain from cavity preparation, making anesthesia unnecessary. The Air Dent had numerous problems that could not be overcome, but Dr. Black never gave up on his idea. The technology he invented became the basis of air polishing and was first marketed in 1976. By the late 1970s, air polishing was readily available.
Air polishing represents the most significant addition to the polishing armamentarium since the introduction of handpieces and prophylaxis angles.1 Air polishing is accomplished by the propulsion of abrasive particles through a mixture of compressed air and water through a handpiece nozzle.1 Kinetic energy propels the air polishing paste particles against the tooth surface—removing stain and dental plaque.
Air polishing should not be confused with air abrasion. Air abrasion uses greater air pressure and more abrasive particles. It is intended for procedures such as removing decayed enamel and roughening enamel surfaces prior to bonding.2,3 Aluminum oxide is the standard abrasive powder for use in air abrasion. The Mohs Hardness Index ranges from 1-10; aluminum oxide has a Mohs hardness number of 9, which is four to five times more abrasive than air polishing agents.
THE BASICS
Polishing is accomplished by two types of wear—abrasion or erosion. Traditional polishing with a rubber cup and polishing paste uses abrasion. The process creates finer and finer scratches with a series of finer and finer abrasives. Air polishing is accomplished by erosion, which is the recession of surfaces— in this case dental stain and plaque—by suspended abrasive particles within a moving fluid. The most common air polishing abrasive particle is specially processed sodium bicarbonate (SPSB). The SPSB is food grade tribasic and is combined with scant amounts of calcium phosphate and silica to keep it free flowing. The SPSB particles average 74 µm in size. The Mohs hardness number for sodium bicarbonate is 2.5. Comparatively, pumice, the standard particle used in prophylaxis paste, has a Mohs hardness number of 6. The only sodium bicarbonate powder that is safe to use in air polishing is the type specifically designed for air polishing. Over-the-counter sodium bicarbonate can clog air polishing equipment and create operational problems. Of the air polishing abrasive agents developed to date, SPSB has the most extensive body of research available to support its safety and efficacy.
TABLE 1.SEQUELAE THAT CAN DEVELOP AS A RESULT OF COMPRESSED AIR FORCED INTO SOFT TISSUES OF THE HEAD AND NECK.
- Bilateral pneumothorax
- Cerebral air embolism
- Cervicofacial emphysema
- Facial emphysema
- Mediastinal emphysema
- Pneumediastinum
- Pneumothorax
- Retropharyngeal emphysema
Aluminum Trihydroxide Polishing Powder
Aluminum trihydroxide was the first air polishing agent developed as an alternative to SPSB for patients who are sodium intolerant.4 The Mohs hardness number for aluminum trihydroxide is 4 and the particles range in mesh size from 80 µm to 325 µm. Aluminum trihydroxide is indicated for patients who have heavily stained enamel. Contraindications include use on dentin, cementum, amalgam, gold, all composite types, glass ionomers, and implants.4 Aluminum trihydroxide does not cause surface disruption to porcelain. However, the luting agents used for placement of porcelain restorations are removed by aluminum trihydroxide, causing a compromise in the margin integrity that could quickly lead to decay.
DELIVERY SYSTEMS
There are two basic types of air polishing delivery systems: self-contained and those that attach to handpieces. Self-contained air polishing units attach to the compressed air and water lines of the dental unit and require an electric outlet. The alternative type of air polisher attaches to the handpiece connection on the dental unit, obtaining the compressed air and water from the handpiece lines. No electrical connection is required for the handpiece connection unit. In general, self-contained units have a range of water pressure of 10 psi to 50 psi. The inlet air pressure from the dental unit is approximately 60 psi. The outlet air pressure, which is delivered out of the nozzle, is set between 58 psi to 60 psi.5
ASSESSMENT AND PREPARATION
The decision to use air polishing should be made based on a patient’s medical history and patient assessment. Contraindications for air polishing include:6-8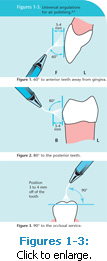
- Patients on a physician-directed sodiumrestricted diet and those who have hypertension. However, research shows that the amount of sodium bicarbonate ingested during air polishing is not sufficient to cause an increase in blood pressure or blood levels of sodium or alkalosis.6,9
- Patients who have respiratory problems such as chronic obstructive pulmonary disease or any condition that interferes with breathing or swallowing. These patients may be bothered by the aerosols created by air polishing and they are also vulnerable to the development of pneumonia.
- Patients with end-stage renal disease or who are otherwise immunocompromised. A physician consult should be initiated before performing air polishing on immunocompromised patients. • Patients with a communicable infection. • Patients who have Addison’s disease or Cushing’s disease.
- Patients taking potassium, anti-diuretics, or steroid therapy.
Standard infection control procedures should be employed when using air polishing, including the use of protective eye wear for the patient and a preprocedural rinse to lower microbial loads in the inherent aerosols. Patients wearing contact lenses should remove them and clinicians should wear a high filtrate face mask.7
TABLE 2.SEQUELAE THAT CAN DEVELOP AS A RESULT OF FACIAL EMPHYSEMAS.
- Bilateral pneumothorax
- Cerebral air embolism
- Embolism
- Pneumediastinum
- Pneumothorax
- Thrombosis
TECHNIQUE
While various manufacturers may have differing instructions for use of their equipment, there is a universal air polishing technique that can be used with all types of air polishing systems.7,8 This technique prevents undue aerosols from deflecting back to the clinician or being directed into the patient’s soft tissues. For the most efficient control of aerosols, high speed evacuation should be used at all times to contain the aerosolized spray.7,10 The handpiece nozzle should be kept in a constant circular motion and the nozzle tip should be kept 3 mm to 4 mm away from the enamel surface. The closer the nozzle is to the enamel, the more spray that will deflect in the direction of the clinician.
Maintaining the proper angulations for polishing the anterior, posterior, and occlusal surfaces of the teeth is essential for removing stain and dental plaque without creating iatrogenic soft tissue trauma, which will occur if the clinician directs the spray into soft tissues directly, such as gingival tissue and buccal mucosa. For the patient, this may result in minor discomfort. If any tissue trauma does result it should heal quickly and usually cannot be identified after 24 hours.1,11 The universal angulations for air polishing are: 60º to anterior teeth away from gingiva (Figure 1), 80º to the posterior teeth (Figure 2), and 90º to occlusal surfaces (Figure 3).7,8
Using correct handpiece angulation is the single best method of controlling excess aerosol production.10 When a clinician directs the handpiece at a 90º angle to any tooth surfaces other than the occlusal, the usual result is an immediate reflux of the aerosolized spray back toward the clinician or patient.
FACIAL EMPHYSEMA

Incorrect handpiece angulation with air polishers must be avoided in order to prevent the occurrence of a very serious medical condition— iatrogenic facial emphysema.7,12,13 Air polishing handpiece nozzles should never be directed subgingivally into periodontal pockets where there is little or no bony support remaining;14 into or near traumatic lacerations or surgical wounds15 where there is disruption of the intraoral barrier (dentoalveolar membrane); or into extraction sites.16
Facial emphysemas can occur even in small areas open to subcutaneous tissues. They have been associated with oral lacerations as small as 4 mm. Iatrogenic facial emphysemas (also known as a tissue emphysemas or subcutaneous emphysemas) occur because of compressed air that becomes trapped in interstitial spaces. Facial emphysemas resulting from the use of compressed air in dental procedures are not rare. The most common causes are the use of high-speed handpieces during procedures associated with third molar extractions,17,18 use of air/water syringes near extraction or surgical sites16,19 or lacerations,20 and air polishing.21,22 Unfortunately, facial emphysemas can be difficult to diagnose and emergency personnel may not be familiar with them.23,24 Facial emphysemas associated with dental procedures exhibit symptoms that result in facial swelling, a “crackling” sensation on the face and neck area, tenderness, and pain.25 If detected early, patients usually require observation, analgesia, and antibiotic therapy. However, much more serious problems, such as thrombosis and fatal embolism, can result if the problem is not diagnosed. Table 1 lists the possible sequelae that can result from using compressed air near areas open to subcutaneous tissues while Table 2 provides the sequelae that can develop from facial emphysemas.
TABLE 3. ADVANTAGES OF AIR POLISHING COMPARED TO TRADITIONAL POLISHING.
- Can remove up to 100% of bacteria and endotoxins
- Can be used on implants
- Creates uniformly smooth root surfaces
- Greater access for stain removal in pits and fissures
- Less abrasive
- Method of choice for plaque removal prior to placement of sealants or bonding procedures
- Method of choice for stain and plaque removal from orthodontically bracketed and banded teeth
- More comfortable for patient
- No heat generated
- No pressure against teeth
- No tooth contact
- Reduced operator fatigue
- Stain and dental plaque removed in less than half the time
- Temporarily relieves dentinal hypersensitivity
RESEARCH
Air polishing has sufficient in vitro and in vivo research available to identify and firmly establish appropriate uses and advantages as well as inappropriate uses and contraindications. Air polishing has many advantages over traditional polishing (see Table 3).
Air polishing creates less discomfort for patients who have dentinal hypersensitivity because the sodium bicarbonate particles embed in the dentinal tubules, lessening dentinal hypersensitivity discomfort almost immediately.26 However, these particles are hydrophilic and dissipate fairly quickly, leaving the dentinal tubules open once again.
Several in vitro research projects have determined that there is little or no disruption of enamel, cementum, and dentin surfaces with air polishing.6,27 Additional research indicates that air polishing can render cementum surfaces uniformly smooth, compared to traditional polishing or the use of curets.28 Virtually 100% of endotoxins and bacteria can be removed with air polishing, which can promote the growth of fibroblasts.29,30 Air polishing can remove subgingival bacteria through the Venturi effect, which occurs when the airwater- powder spray is directed at a 90º angle to the interproximal spaces so that a vacuum is created that extracts tissue fluids, including subgingival bacteria from the subgingival space.10 This advantage can be particularly useful if air polishing is being used for debridement and the goal is to remove as much bacterial load as possible.
Air polishing is the method of choice for preparing teeth for placement of sealants and bonding procedures because the air polishing stream reaches into pits and fissure where rubber cups and brushes cannot.31 Access is not the only reason for using air polishing. Commercially prepared prophylaxis polishing pastes contain glycerin, which can interfere with the chemical aspect of bonding procedures.7 Air polishing has no such ingredient and leaves no residue or dental plaque. Air polishing is also the method of choice for removing dental plaque and stain from orthodontically bracketed and banded teeth.32,33
RESTORATIVE MATERIALS
With the use of the universal technique and SPSB, air polishing is safe for amalgam, gold, porcelain, and orthodontic brackets and bands.32-39 Air polishing should be avoided on all types of composites, glass ionomers, and luting agents (cements).33-39 Air polishing with SPSB does not damage titanium used for implants and is a method of choice for decontamination of implants.40-44 However, the airpowder- water stream should not be directed subgingivally when polishing implants. Table 4 describes the effects of air polishing on various restorative materials and on orthodontic brackets and bands.
EVIDENCE-BASED DECISION MAKING
Numerous investigations have examined the effects of air polishing and most of the outcomes have been fairly uniform. However, enough contradictory results exist to make evidence-based decisions difficult for some. The standards of correct usage of air polishing that have already been supported by evidence should be the foundation of the evaluation of any new research on air polishing. The key area of focus when reviewing air polishing research should be on the methods and materials. See Table 5 for important questions to ask when reviewing new research on air polishing.
One variable that has not received enough attention in air polishing research protocols is the time of exposure. Since 1984, it has been established that a tooth surface receives a 0.5 second exposure to the air-water-powder ratio during a maintenance appointment.29 Some additional points to keep in mind when evaluating research outcomes include: visual changes in restorative materials detected with the eye do not necessarily equate to alteration in the integrity of the material, and statistical significance does not necessarily equate to clinical significance.
Many research articles on air polishing include scanning photomicrographs to illustrate changes or lack of changes in surface characterization. These photomicrographs provide interesting information, but they are limited in the efficacy of their interpretation. Following are some key points for interpreting these photomicrographs:
1. Photomicrographs of particle shape provide no evidence of hardness. It is the hardness of the particle that is the most relevant information because it determines efficacy and safety for tissues and materials. The shape of the particle is irrelevant to abrasiveness as long as the air polishing agent is softer than the surface being polished.
2. If scanning electron photomicrographs of enamel, cementum, or dentin have a cracked, dry creek-bed appearance (Figure 4), the actual tooth was viewed under the scanning scope. For the tooth to be viewed under the SEM scope, it must be placed in a vacuum to remove moisture, therefore, the tooth desiccates and dries out, making the evaluation of the surface characterization all but impossible. When tooth surfaces are replicated and viewed, the image will be free of the cracked appearance, making evaluation of the surface characterization possible.
3. Two dimensional photomicrographs do not show the amount of surface roughness or the amount of tissue or material loss. Surface roughness, gloss, and material loss must be determined using profilometers, (either contact or noncontact) and glossmeters or reflectometers.
References are another area of concern when reviewing air polishing literature. In some cases, references appear to support a particular air polishing agent or brand of equipment but on closer inspection, it is found that the research was conducted on a different type of air polishing equipment or air powder polishing agent. Different types of air polishing equipment have different pressure settings, air-waterpowder ratios can differ, and the composition of powder polishing agents mixtures vary as does the hardness of the polishing agents.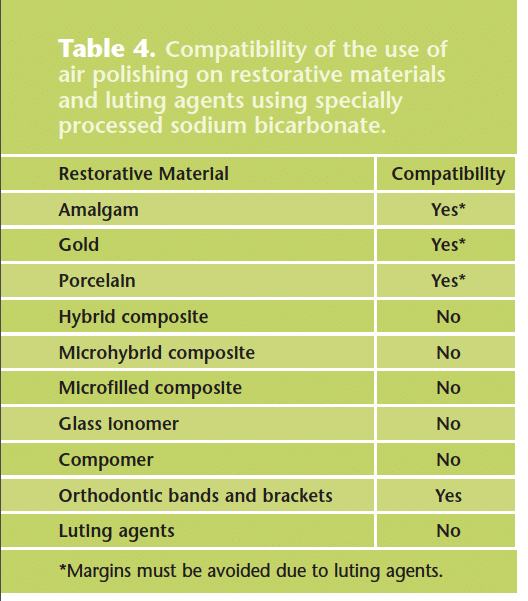
WHAT’S NEW?
Recently several new types of air polishing powders have been introduced that include glycine, calcium carbonate, and calcium sodium phosphosilicate (NovaMin®). However, before using a new powder, clinicians should review the warranty on their air polishing units to see if using a different powder than is recommended for the machine voids the manufacturer’s warranty.
Glycine, an amino acid, is available in two grades: pharmaceutical and technical. Glycine crystals can be grown using a solvent of water and sodium salt and then prepared for use in powder formulations. Glycine particles used for air polishing are 20 µm in size and have a Mohs hardness number of 2. In Europe, glycine powder is unfortunately recommended for use with a device to deliver the air powder polishing agent subgingivally.45,46 However, to ensure that no traumatic event occurs whether it be a facial emphysema or an air embolism, no air powder polishing agent should ever be directed subgingivally with any type of equipment.
Calcium carbonate is a naturally occurring substance that can be found in rocks. It is used as filler for pharmaceutical drugs in pills and tablets and is a main ingredient in antacids. Calcium carbonate is also found in household scouring products, particularly those that claim to not scratch surfaces. It is very soft and has a Mohs hardness number of 3.47 Calcium sodium phosphosilicate or Novamin is a bioactive glass. A significant amount of in vivo research has not been conducted on calcium sodium phosphosilicate yet. In vitro research shows that calcium sodium phosphosilicate interacts with oral fluids and releases sodium, calcium, and phosphate ions. The primary focus of the research conducted on calcium sodium phosphosilicate is on its ability to form hydroxycarbonate apatite when combined with oral fluids for the purpose of remineralization and the treatment of dentinal hypersensitivity by occluding dentinal tubules. Calcium sodium phosphosilicate has a Mohs hardness number of 6, making it the hardest air polishing particle found in air powder polishing agents.
Air polishing has many advantages and it will be exciting to see the research and clinical novelties of these new polishing agents as well as others being investigated for development.
REFERENCES
- Weaks LM, Lescher NB, Barnes CM, Holroyd SV. Clinical evaluation of the Prophy-Jet as an instrument for routine removal of tooth stain and plaque. J Periodontol. 1984;55:486-488.
- Anusavice KJ. Phillip’s Science of Dental Materials. 11 th ed. St. Louis, Mo: Saunders; 2003:73.
- Reyto R. Lasers and air abrasion. New modalities for tooth preparation. Dent Clin North Am. 2001;45:189-206.
- Johnson WW, Barnes CM, Covey DA, Walker MP, Ross JA. The effects of a commercial aluminum air polishing powder on dental restorative materials. J Prosthodont. 2004;13,166-172.
- Horning G. Clinical use of an air-powder abrasive. Compend Contin Ed Dent. 1987;8:652-658.
- Gutmann ME. Air polishing: a comprehensive review of the literature. J Dent Hyg. 1998;73:47-56.
- Wilkins EM. Clinical Practice of the Dental Hygienist. 10th ed. Philadelphia: Lippincott, Williams & Wilkins; 2009:735-738.
- Daniel SJ, Harfst SA, Wilder RS. Mosby’s Dental Hygiene Concepts, Cases and Competencies. 2nd ed. St. Louis, Mo: Mosby; 2008:599-622.
- Snyder JA, McVay JT, Brown FH, et al. The effect of air abrasive polishing on blood pH and electrolyte concentrations in healthy mongrel dogs. J Periodontol. 1990;61:81-86.
- Barnes CM. The management of aerosols with air polishing delivery systems. J Dent Hyg. 1991;65:280-252.
- Agger MS, Hörsted-Bindslev P, Hovgaard O. Abrasiveness of an air-powder polishing system on root surfaces in vitro. Quintessence Intl. 2001;32:407-411.
- Karras SC, Sexton JJ. Cervicofacial and mediastinal emphysema as the result of a dental procedure. J Emerg Med. 1996;14:9-13.
- Fruhauf J, Weinke R, Pilger U, Kerl H, Mullegger RR. Soft tissue cervico facial emphysema after dental treatment. Arch Dermatol. 2005;141:1437-1440.
- Snyder MB, Rosenberg ES. Subcutaneous emphysema during periodontal surgery: report of a case. J Periodontol. 1977;48:790-791.
- McDonnell DG. Surgical emphysema in the subcutaneous tissues of the face following restorative procedures. J Ir Dent Assoc. 1983;29:20-21.
- Tan WK. Sudden facial swelling: subcutaneous facial emphysema secondary to use of air/water syringe during dental extraction. Singapore Dent J. 2000;23(1 Suppl):42-44.
- Arai I, Aoki T, Yamazaki H, Ota Y, Kaneko A. Pneumomediastinum and subcutaneous emphysema after dental extraction detected incidentally by regular medical checkup: a case report. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2009;107:33-38. Epub 2009 Feb 8.
- Sekine J, Irie A, Dotsu H, Inokuchi T. Bilateral pneumothorax with extensive subcutaneous emphysema manifested during third molar surgery. A case report. Int J Oral Maxillofac Surg. 2000;29:355-357.
- Uehara M, Okumura T, Asahina I. Subcutaneous cervical emphysema induced by a dental air syringe: a case report. Int Dent. 2007;57:28628-8.
- Yamada H, Kawaguchi K, Tamura K, Sonoyama T, Iida N, Seto K. Facial emphysema caused by cheek bite. Int J Oral Maxillofac Surg. 2006;35:188-189.
- Finlayson RS, Stevens FD. Subcutaneous facial emphysema secondary to use of the Cavi-Jet. J Periodontol. 1988;59:315-317.
- Liebenberg WH, Crawford BJ. Subcutaneous, orbital, and mediastinal emphysema secondary to the use of an air-abrasive device. Quintessence Int. 1997;28:31-38.
- Frühauf J, Weinke R, Pilger U, Kerl H, Müllegger RR. Soft tissue cervicofacial emphysema after dental treatment: report of 2 cases with emphasis on the differential diagnosis of angioedema. Arch Dermatol. 2005;141;1437-1440.
- Yang SC, Chiu TH, Lin TJ, Chan HM. Subcuteanous emphysema and pneumomediastinum secondary to dental extraction: a case report and literature review. Kaohisiumg K Med Sci. 2006;22:641-645.
- Bavinger JV. Subcutaneous and retropharyngeal emphysema following dental restoration: an uncommon complication. Ann Emerg Med. 1982;11:371-374.
- Bester SP, de Wet FA, Nel JC, Driessen CH. The effect of airborne particle abrasion on the dentin smear layer and dentin: an in vitro investigation. Int J Pros. 1995;8:46-50.
- Gerbo LR, Lacefield WR, Barnes CM, Russell CM. Enamel roughness after air-powder polishing. Am J Dent. 1993;2:696-698.
- Berkstein S, Reiff RL, Mc Kinney JF, Killoy WJ: Supragingival root surface removal during maintenance procedures utilizing an air-powder abrasive system or hand scaling. J Periodontol. 1987;58:327-333.
- Atkinson DR, Cobb CM, Killoy WJ. The effect of an air-powder abrasive system on in vitro root surfaces. J Periodontol. 1984;55:13-18.
- Gilman RS, and Maxey BR. Effect of root detoxification on human gingival fibroblasts. J Periodontol. 1986;57:436-440.
- Brockmann SL, Scott RL, Eick JD. The effect of an air-polishing device on tensile bond strength of a dental sealant. Quintessence Int. 1989;20:211-217.
- Barnes CM, Russell CM, Gerbo LR, Wells B, Barnes DW. Effects of an air-powder polishing system on orthodontically bracketed and banded teeth. Am J Ortho. 1990;97:74-81.
- Gerbo LR, Barnes CM, Leinfelder KF. Applications of the air-powder polisher in clinical orthodontics. Am J Orthod Dentofacial Orthop. 1993;103:71-73.
- Barnes CM, Hayes EF, Leinfelder KF. Effects of an airabrasive polishing system on restored surfaces. Gen Dent. 1987;35:186-189.
- Lubow RM, Cooley RL. Effect of air-powder abrasive instrument on restorative materials. J Prosthet Dent. 1986;55:462-465.
- Cooley RL, Lubow RM, Brown FH. Effect of air-powder abrasive instrument on porcelain. J Prosthet Dent. 1988;60:440-443.
- Carr MP, Mitchell JC, Seghi RR, Vermilyea SG The effect of air polishing on contemporary esthetic restorative materials. Gen Dent. 2002;50:238-41.
- Gutmann MS, Marker VA, Gutmann JL Restoration surface roughness after air-powder polishing. Am J Dent. 1993;6:99-102.
- Yap AU, Wu SS, Chelvan S, Tan ES.Effect of hygiene maintenance procedures on surface roughness of composite restoratives. Oper Dent. 2005;30:99-104.
- Barnes CM, Fleming LS, Mueninghoff LA. SEM evaluation of the in-vitro effects of an air-abrasive system on various implant surfaces. Int J Oral Maxillofac Implants. 1991;6:463-469.
- Augthun M, Tinschert J, Huber A. In vitro studies on the effect of cleaning methods on different implant surfaces. J Periodontol. 1998;69:857-864.
- Mengel R, Buns CE, Mengel C, Flores-de-Jacoby L. Instruments. Int J Oral Maxillofac Implants. 1998;13:91-96.
- Matarasso S, Quaremba G, Coraggio F, Vaia E, Cafiero C, Lang NP.Maintenance of implants: an in vitro study of titanium implant surface modifications subsequent to the application of different prophylaxis procedures. Clin Oral Implants Res. 1996;7:64-72.
- Meschenmoser A, d’Hoedt B, Meyle J, Elssner G, Korn D, Hämmerle H, Schulte W. Effects of various hygiene procedures on the surface characteristics of titanium abutments. J Periodontol. 1996:67:229-235.
- Petersilka G, Faggion CM Jr, Stratmann U, Gerss J, Ehmke B, Haeberlein I, Flemmig TF. Effect of glycine powder air-polishing on the gingiva. J Clin Periodontol. 2008;35:324-332.
- Flemmig TF, Hetzel M, Topoll H, Gerss J, Haeberlein I, Petersilka G.Subgingival debridement efficacy of glycine powder air polishing. J Periodontol. 2007;78:1002-1010.
- Frankenberger R, Lohbauer U, Tay FR, Taschner M, Nikolaenko SA. The effect of different air-polishing powders on dentin bonding. J Adhes Dent. 2007;9:381-389.
From Dimensions of Dental Hygiene. March 2010; 8(3): 32, 34-36, 40.

