
Addressing the Pain of Dentinal Hypersensitivity
Dental hygienists need to understand the dynamics of hypersensitivity in order to best advise their patients.
Dealing with dentin hypersensitivity can be challenging both for the clinician and the patient. Dentin hypersensitivity may seem to clinicians like a minor impediment to providing care. However, the patient’s experience may range from a small twinge to excruciating and persistent pain.
Providing multiple areas of local anesthesia is time consuming and may be unrealistic for the patient when there are several sensitive teeth located throughout the mouth and a full dentition procedure needs to be completed. This procedure often must be planned as two separate half-mouth appointments with local anesthesia, which requires additional time and expense. Neither topical anesthesia nor dispensing anesthetic into the sulcus with a blunt-type needle will provide complete relief from hypersensitivity pain since it is tooth-related rather than soft tissue discomfort. No clinician wants to elicit patient discomfort. Thus, dental hygienists need to be well-versed in dealing with this disorder.
The Patient’s Perspective
Although less critical than a debilitating disease, dentinal hypersensitivity can alter quality of life. The individual with sensitivity may be painfully reminded many times daily to avoid everyday situations such as using a drinking fountain, consuming cool drinks directly from the refrigerator, and talking while walking in the cold. Some people with severe sensitivity have even described not being able to bite into a sandwich made with meat stored in the refrigerator. Cumulatively, these relatively minor annoyances can have a significant impact on a patient. The pain caused by sensitivity can also create a negativity about receiving dental care, which can extend to the clinician who patients may view as being unable to effectively address their sensitivity problem. An easily applied and tolerated product that would allow for immediate comfort during instrumentation of sensitive cervical/root areas would be embraced by both the clinician and the patient. A wide variety of agents and approaches are either currently available or in development that are designed to treat dentin hypersensitivity both in the dental office and at home.
PREVALENCE AND ETIOLOGY
Neither the prevalence statistics nor the accepted theory of etiology have changed in recent years. The generally reported occurrence rate is about one in five American adults, although a prevalence as high as 50% has been found in studies where individuals self-report. This disparity may be caused by patients confusing dentinal hypersensitivity with other forms of dental pain.1
The hydrodynamic theory is still accepted as the explanation of how dentin hypersensitivity occurs. It was first described in 19002 and elaborated on in 1963.3 Still referred to as a “theory” since proof is elusive; no other viable explanation has emerged to replace it. The basic explanation is that fluid movement within the dentinal tubules informs pulpal nerve receptors, resulting in a sensation of pain. Stimuli, such as cold water or evaporative air, readily cause a pressure change in the tubule, which leads to fluid movement. Heat stimulates slower fluid movement than cold. Therefore, heat is not as strongly implicated in causing sensitivity as are cold and evaporative stimuli.
RISK FACTORS
Recession and consequent enamel loss leading to dentin exposure are precursors to the occurrence of dentin hypersensitivity. Recession is typically initiated by damaging oral hygiene practices but can also include sequelae from oral surgery procedures; treatment for periodontal diseases; effects of periodontal diseases, such as necrotizing ulcerative gingivitis; crown preparation procedures; anatomic thin buccal plate of bone or tissue; or narrow zone of attached gingiva.4
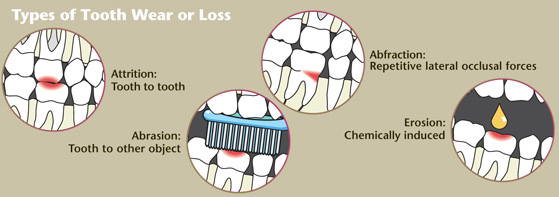
A significant risk factor for the development of hypersensitivity is the loss of enamel or cementum, which results in the underlying dentin becoming exposed. Cementum is thinner and less mineralized, thus, it is removed more easily than enamel. Many different risk factors can lead to the loss of tooth structure. However, the singular or combined effects of abrasion, attrition, erosion, and possibly abfraction are also contributory.
Abrasion can be caused by the contact between the tooth and an object such as a toothbrush, abrasive particles in a dentifrice, prophylaxis paste, or a coarse diet. The texture of the toothbrush bristles in addition to aggressive toothbrushing can lead to abrasion. The recommended use of commercially available dentifrice with an appropriate brushing technique should not lead to loss of tooth structure. However, in combination with aggressive brushing and especially when used in an acidic oral environment favoring erosion, abrasion can be a risk factor for tooth structure loss.
Attrition is the tooth-to-tooth wear that can cause the loss of enamel or cementum— exposing the underlying dentin. Initiated by occlusion or habit, bruxism frequently causes attrition. Another example of attrition is the wear facets created by occlusal forces during mastication.
Erosion is a significant risk factor contributing to loss of tooth structure. Erosion is caused by acids eroding the tooth structure by dissolving the enamel, cementum, and/or dentin. The source of the acid can be acquired extrinsically from the diet or intrinsically, such as from gastroesophageal reflux disease. Carbonated drinks (both regular and diet sodas), sports drinks, fruit juices, wines, and, to a lesser degree, beer, can also contribute to erosion. Patients who consume a significant amount of carbonated beverages and sports drinks should be encouraged to use a straw to decrease the acidic exposure of the teeth.
After the dentin has been exposed, consistent introduction of dietary acid can continue to erode dentin and keep the dentinal tubules open. The hypersensitivity reaction is maintained in the presence of open dentin tubules because a stimulus can move the fluid within the tubules—signaling the pulpal nerves. Bacterial elements can also be transported inward through open tubules, from the oral cavity to the pulp. When this happens, the resulting inflammatory pulpal response can create additional discomfort.
Erosion is most serious when combined with abrasion from toothbrushes and toothpaste.5 The acid in food or beverages can increase susceptibility to abrasion by demineralizing the tooth structure. Dental hygienists can recommend that patients experiencing chronic hypersensitivity change their sequence of oral hygiene if it includes the consumption of acidic beverages, such as orange juice, just before toothbrushing.
Abfraction is a cervical stress-lesion that is caused by occlusal forces that create tooth flexure, which can create a wedge- or v-shaped configuration at the cemento-enamel junction. The relationship between abfraction and dentin hypersensitivity is still under investigation.4
TREATMENT
Two approaches can be used to treat dentin hypersensitivity. First the neural response can be prevented or lessened. Second, the tubules can be blocked to prevent the stimulus from signaling tubular fluid movement.
The commonly used first treatment includes the home use of a dentifrice containing a potassium salt (usually potassium nitrate). Potassium nitrate products work via the potassium- sodium pump, interrupting nerve impulse conduction. Improvement takes about 4 weeks. These dentifrices usually also contain fluoride to maintain an anti-caries effect.
Stannous fluoride has been used in the treatment of sensitive teeth for many years. However, because of issues including taste, stability, and staining, stannous fluoride has not been included in over-the-counter dentifrices until recently, when these issues were resolved. Other fluoride types and products at prescription levels are helpful in treating sensitivity; these can be delivered through home use of dentifrice, gels, or in trays. The most highly concentrated fluoride available is 5% sodium fluoride, which is applied in the professional setting via fluoride varnish. Interestingly, the United States Food and Drug Administration approval for fluoride varnish is actually for the treatment of sensitivity rather than for caries prevention. The mechanism of all fluorides is that they precipitate calcium fluoride at the exposed dentin surface to block open dentin tubules.
Other products work by narrowing the dentinal tubule diameter, causing less fluid flow within the dentinal tubule, which interrupts the signal to the nerve endings. Oxalic salts, such as potassium oxalate and ferric oxalate, can precipitate calcium oxalate crystals within the tubule. Gluteraldehyde products are used to treat sensitivity by decreasing the diameter of the dentinal tubule lumen after coagulation of proteins and amino acids.
The newest products on the market for sensitivity use calcium phosphate technology. Primarily reported to increase remineralization and/or decrease demineralization for caries control, their use has also been applied to the treatment of sensitivity since a more mineralized surface would likely elicit less sensation of sensitivity. There are different types of calcium phosphate technology including calcium sodium phosphosilicate, casein phosphopeptide- amorphous calcium phosphate, and amorphous calcium phosphate. In addition, one of the calcium-containing products that is marketed solely for treatment of sensitivity incorporates arginine, an amino acid.
The theory behind these remineralization products, which are also often recommended for dentinal sensitivity, is that calcium phosphate will diffuse into the tooth, replacing lost mineral. When present, fluoride acts as a catalyst by encouraging this reaction. The usual vehicle for supplying calcium and phosphate is the saliva. Normal saliva is fully saturated with calcium and phosphate for the purpose of remineralization, even when subjected to an acid challenge. When there is a salivary deficiency, these products may play a beneficial role in remineralization and in desensitization.
James S. Wefel, PhD, director of the Dows Institute for Dental Research, administrative director of the Office of Clinical Research, and professor in the Department of Pediatric Dentistry at the University of Iowa, Iowa City, conducts research on tooth remineralization/ demineralization. In a 2009 interview with Dimensions of Dental Hygiene, he provided his opinion on the subject, “The main vehicle for calcium phosphate is saliva. Saliva has enough calcium phosphate in it to keep the oral environment saturated if not super saturated… Most of the products containing calcium phosphate are what I refer to as salivary enhancers. They are designed to provide more calcium phosphate, which normally comes from saliva. These products work best in people with poor salivary flow who don’t maintain the right calcium phosphate levels. This is common in people with some sort of xerostomic condition, whether it’s from head and neck cancer treatment that shuts down the salivary glands, the taking of hypertensive drugs that dry out the mouth, or diseases like Sjogren’s syndrome that decrease salivary flow. If salivary flow is restricted, calcium phosphate products can be quite useful in supplying calcium phosphate that is lacking from the oral environment.”6
RESEARCH
Many of the studies conducted on the calcium phosphate technologies are laboratory-based (in vitro), rather than clinically-based (in vivo). In order to address the need for clinical research the CRA Foundation, an educational nonprofit foundation formerly known as Clinical Research Associates Inc, is currently testing seven commercial products and a control group in a large, randomized, controlled clinical trial with full-banded orthodontic patients as the subjects. The subjects will be followed from banding to debanding day to compare the efficacy of the products in preventing decalcification.
The control will consist of the usual oral hygiene directions within an orthodontic population. This protocol is designed to study decalcification rather than desensitization, although there is some crossover. This type of clinical research will assess the clinical efficacy of products marketed for treatment of hypersensitivity. There are inherent challenges in the objective evaluation of dentinal sensitivity outcomes. However, these can be minimized with welldesigned studies.
The vast array of products that may provide help to patients who experience dentinal hypersensitivity is exciting and dental hygienists should anticipate the results of such clinical research to reveal the efficacy of new additions to the hypersensitivity armamentarium.
REFERENCES
- Murray LE, Roberts AJ. The prevalence of self-reported hypersensitive teeth. Arch Oral Biol. 1994;39(suppl):1295.
- Gysi A. An attempt to explain the sensitiveness of dentine. Br J Dent Res. 1900;43:865.
- Brannstrom ML. Dentin sensitivity and aspiration of odontoblasts. J Am Dent Assoc. 1963;6:366-370.
- Tilliss T. Understanding dentin hypersensitivity. Dimensions of Dental Hygiene. 2003;1(6):22-31.
- Dababneh RH, Khouri AT, Addy M. Dentine hypersensitivity—an enigma: A review of terminology, epidemiology, mechanisms, aetiology, and management. Brit Dent J. l999;187:606-611.
- Chow L, Wefel JS. The dynamics of de- and remineralization. Dimensions of Dental Hygiene. 2009;7:42, 44-46.
From Dimensions of Dental Hygiene. April 2010; 8(4): 46, 48, 50.

