The Inflammatory Response
Periodontal diseases are related to an immunoinflammatory response to various types of oral biofilm.
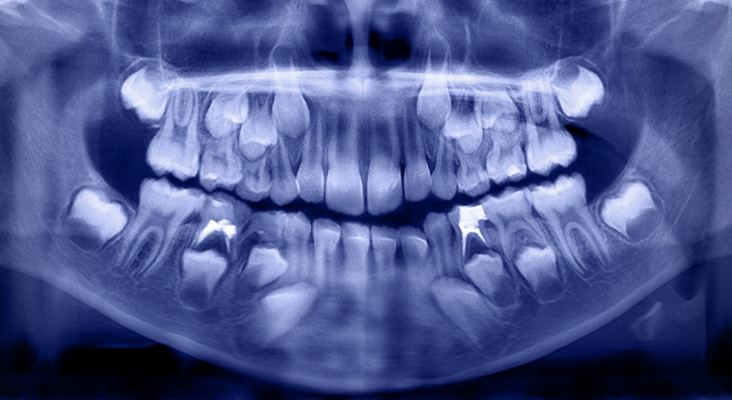
Periodontal diseases are related to an immunoinflammatory response to various types of oral biofilm. They affect approximately 42% of United States adults. Chronic periodontitis is characterized by alveolar bone loss, attachment loss, and apical migration of the junctional epithelium. Periodontal diseases result from dysbiosis: an imbalance between different organisms present in the natural microbiota of the subgingival microenvironment. Periodontal destruction can be attributed to the interaction of two main players: periodontally pathogenic biofilm and the host-mediated immunoinflammatory response to that biofilm. Current treatment guidelines for periodontitis include the physical removal of plaque biofilm and calculus by scaling and root planing. While this treatment aims to eliminate the bacterial insult, scaling and root planing does not address the host response element of periodontal diseases.
Photo Credit: -1001- / iStock / Getty Images Plus
Bacterial Insult
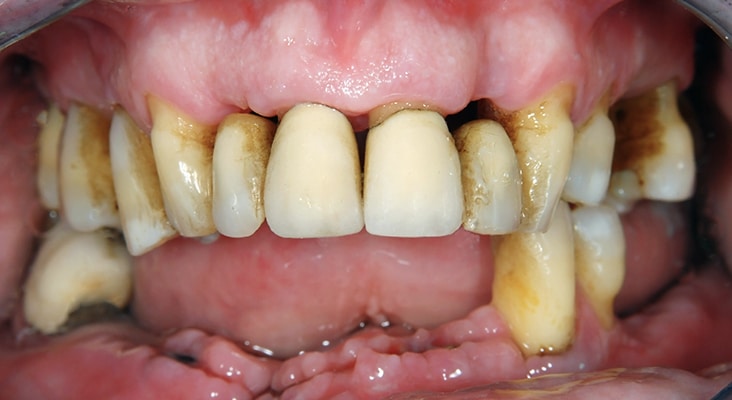
Inflammation is the body’s initial response to various types of injury via physical, bacterial, or chemical means, and is characterized by an increase in temperature, erythema, edema, pain, and loss of function. During an acute inflammatory response, a series of events occurs including increased vessel permeability, emigration of leukocytes to the site of injury, and activation of macrophages, which ingest foreign substances, toxins, and debris. If the acute injury is eliminated, tissue repair begins in an effort to return to its original state. If inflammation persists due to continued insult or an exaggerated immune response, tissue damage continues. The chronic inflammatory response involves various cells of the acquired immune system: macrophages, lymphocytes, and plasma cells and their products. The development of periodontal diseases is initiated by a dysbiotic bacterial plaque insult and tissue destruction is propagated by the host immune response to the biofilm. This response differs among individuals, which may explain why some patients have low levels of bacterial plaque deposits and excellent oral hygiene, but still experience chronic inflammation and bone loss.
Photo Credit: danielzgombic / E+
Host Modulation Therapies
A variety of potential host-modulation therapies are available to treat periodontal diseases, including NSAIDs, subantimicrobial-dose doxycycline (SDD), bisphosphonates, and cytokine inhibitors. However, most of these therapies are not yet proven or approved treatments for periodontitis. NSAIDs regulate the host response by decreasing PGs, pro-inflammatory hormones that demonstrate local upregulation in response to lipopolysaccharides (LPS) present in plaque biofilm. NSAIDs are known to be direct inhibitors of cyclooxygenase, an enzyme involved in tissue breakdown via the production of arachidonic acid metabolites that lead to activation of osteoclasts and MMPs. While NSAIDs are proven to decrease PG synthesis, the side effects of long-term use have prevented it from being incorporated into regular periodontal treatment. A recent systematic review revealed clinical benefits for the use of NSAIDs in conjunction with nonsurgical periodontal treatment in reducing the rate of gingival inflammation and alveolar bone resorption. However, these benefits did not extend to the improvement of probing depth or clinical attachment level. Unwanted side effects may also be reduced by the use of novel NSAID therapies, NSAID dentifrices, and/or lower dose NSAIDs. Geisinger et al concluded that the addition of low-dose aspirin therapy in patients with chronic periodontitis undergoing nonsurgical periodontal therapy was beneficial. Aspirin reduces inflammatory mediators and triggers production of resolvin D3, an inflammation resolving mediator derived from the metabolism of omega-3 fatty acids, by modifying cells’ production of cyclo-oxygenase type 2, another inflammation-inducing PGs. Additional studies on the use of adjunctive aspirin therapy in the treatment of periodontal diseases are needed.
Photo Credit: Fahroni / iStock / Getty Images Plus

Bisphosphonates
Bisphosphonates are bone-sparing agents that modify the host response by disrupting osteoclastic activity, which inhibits bone resorption. Bone sparing drugs are used to treat metabolic bone diseases but are not recommended for the treatment of periodontal diseases. In addition to disrupting osteoclastic activity, bisphosphonates also induce osteoclastic apoptosis (programmed cell death), and may also inhibit MMP activity or provide anti-collagenase properties. Lane et al found that bisphosphonates provided benefits in clinical attachment levels, probing depths, and bleeding on probing. Despite this promising study, bisphosphonates are not without side effects, including changes in white blood cell production and, rarely, medication-related osteonecrosis of the jaw. They are currently only approved for systemic bone loss by the US Food and Drug Administration (FDA). However, future incarnations of bisphosphonates may be used to treat periodontal bone loss.
Photo Credit: Goldfinch4ever / iStock / Getty Images Plus
NF-kB Pathway
The host modulatory system can be targeted via transcription factors activated by the inflammatory response. Transcription factors are proteins that bind to deoxyribonucleic acid- or DNA-promoters, and regulate gene activation. NF-kB is a family of transcription factors activated by pro-inflammatory cytokines and helps to produce additional pro-inflammatory cytokines. This leads to chronic inflammation. Current research has led to phase II clinical trials on several different drugs that target the NF-kB pathway in patients with rheumatoid arthritis (RA). A study of patients with healthy vs periodontally involved tissues showed an upregulation of NF-kB in diseased states, suggesting that therapeutic targeting of NF-kB may help improve the immune-mediated component of periodontal diseases. Several other proteins within the cell signaling pathway may yield future treatment options but research is currently limited.
Photo Credit: Marcin Klapczynski / iStock / Getty Images Plus
Statins
Statins lower cholesterol in the blood by inhibiting the synthesis of mevalonate, the precursor of cholesterol. Statins have also demonstrated anti-inflammatory and immunomodulatory effects (inhibit tissue-degrading enzymes) and interfere with osteoclasts. Currently, statins are used to treat osteoporosis and have been used to enhance bone formation and reduce inflammation in dentistry. Statins seem to increase production of mRNA of osteoprotegerin by osteoblasts, thereby reducing bone resorption. A recent meta-analysis showed that application of locally applied statins as an adjunct to mechanical periodontal treatment produced larger improvements in terms of periodontal depth reduction and gain of clinical attachment. However, little research is available on the use of statins as an adjunct to conventional periodontal therapy and further research is needed.
Photo Credit: Shidlovski / iStock / Getty Images Plus
What the Future Holds
While SDD is currently the only FDA-approved host modulation therapy for periodontitis, the success of using host modulation therapy in the treatment of other chronic inflammatory diseases proves that host modulation may be effective in controlling the immune response responsible for periodontal diseases. Targeting the inflammatory pathways that lead to dysbiosis and chronic inflammation is ideal to reduce tissue destruction related to host immunoinflammatory activity. Cytokine pathways may provide future drug targets that will modify the host’s immune response in periodontal diseases, but thus far have only been studied with other autoimmune diseases. Data regarding outcomes for periodontitis are still largely unknown. More research is needed on the effects of host modulation therapies specifically on periodontal diseases. Currently, the side effects associated with host modulation medications for RA preclude their safe use in patients exclusively with periodontal diseases. In the future, additional research may yield safer drugs, dosages, and/or delivery methods that can also treat periodontitis.

