 APCORTIZASJR/ISTOCK/GETTY IMAGES PLUS
APCORTIZASJR/ISTOCK/GETTY IMAGES PLUS
Safe and Effective Use of Gloves
Gloves provide the most important barrier to disease transmission.
Oral health professionals are at daily risk of disease exposure, and gloves provide the most important barrier to disease transmission. Knowing how to perform proper hand hygiene, choose the right glove, and follow the steps for safe glove use will help keep clinicians and patients safe and healthy.
The federal government has created regulations and recommendations to prevent disease transmission and promote safe health-care procedures. The Occupational Safety and Health Administration (OSHA) was created in 1970 to protect the health and safety of all workers. In 1987, regulations for chemical safety were addressed with the addition of the Hazard Communication Standard and, in 1991, the Bloodborne Pathogens (BBP) Standards designed to protect against bloodborne pathogens were finalized.
The United States Centers for Disease Control and Prevention (CDC) develops guidelines and recommendations to protect clinicians and patients. Infection control in dentistry is largely based on the CDC’s Guidelines for Infection Control in Dental Health-Care Settings—2003. In 2016, a companion document was released that provides a summary of the 2003 guidelines and new recommendations. OSHA and the CDC continue to develop regulations and recommendations to make practice safe for oral health professionals.
EVALUATING THE RISK
In the dental setting, gloves are just one of the types of personal protective equipment (PPE) mandated by OSHA. They are used to protect the wearer and patient from the spread of infection or illness during examinations and other procedures. Gloves provide an essential layer of barrier protection against direct contact with infectious agents that are transmitted during exposure to blood and bloody saliva, contaminated objects, and surfaces.
With more than 700 species of bacteria detected in the mouth,1 and the ability of some pathogens—such as methicillin-resistant Staphylococcus Aureus—to live on a surface for up to 7 months,2 oral health professionals need to follow the basic principles of infection control.
Glove quality and integrity matter. The US Food and Drug Administration (FDA) regulates the manufacturing and labeling of medical devices, including gloves. It also ensures that performance criteria—such as leak resistance, tear resistance, and biocompatibility—are met. While the efficacy of gloves to reduce BBP transmission has been well established,3 they do not provide 100% protection. When manufactured, new gloves will experience a small percentage of allowable defects, such as micro-tears, which may allow exposure to blood or bloody saliva. As such, performing hand hygiene after glove removal is imperative. In addition, glove integrity decreases over time, with the incidence of failure increasing between 30 minutes and 3 hours of use.4 Changing gloves during longer procedures will reduce this risk. Oral health professionals may also want to consult with manufacturers regarding the chemical compatibility of glove material and the dental materials being used.5
In 2016, the FDA banned the use of powdered surgical and patient exam gloves after determining that the powder posed risks to clinicians, including possible airway inflammation, hypersensitivity reactions, granulomas, and scar tissue formation.6 No remaining glove stock with powder should be used.
GLOVE TYPES AND INDICATIONS
Most patient-care gloves are made of natural rubber latex or synthetic materials, such as nitrile or vinyl. These may either be ambidextrous, fitting both the right and left hand, or fitted, which are hand specific. Ambidextrous gloves may contribute to repetitive stress injuries, while hand-specific gloves typically offer better fit, more comfort, and reduce hand and wrist strain.7
Single-Use Disposable Patient Care Gloves. Exam gloves are used for examining patients and performing procedures that involve contact with mucous membranes. They are not intended for surgical procedures. Surgical gloves are sterile gloves that should be used for all oral surgical procedures. A surgical hand wash must be performed before donning these gloves. Individually packaged pairs are often offered in hand-specific designs and sizes.
Nonpatient Care Gloves: Utility Gloves. Heavy duty utility gloves are a vital but often underutilized type of PPE. They are not used for direct patient care. Both OSHA and CDC indicate that chemical- and puncture-resistant utility gloves should be worn when processing contaminated instruments and performing housekeeping duties (eg, cleaning and disinfecting) and tasks involving chemicals.8 Heavy duty utility gloves offer significant protection against both percutaneous injury and chemical exposure. Typically composed of nitrile or neoprene, they should be washed and disinfected after use. Some types may be heat sterilized; this would be indicated in the manufacturer’s instructions for use (IFU). Heavy duty utility gloves are not considered a medical device; therefore, the FDA does not regulate manufacturing standards. Oral health professionals should have their own pair of well-fitting utility gloves.9 Utility overgloves are similar to food-handling gloves. They may be worn over contaminated exam gloves to prevent cross-contamination when clinicians need to handle an item, such as retrieve a hand mirror from a drawer, during patient care.
MAKING THE BEST CHOICE
Choose the best glove based on the following considerations.
- The task at hand. Gloves should be chosen based on the procedure to be performed (eg, patient exam or surgery)
- Material: latex or nonlatex
- Skin sensitivity: consider latex or nitrile allergies
- Size: Offices should have a variety available
- Fit: a snug but comfortable fit is best. If too large, gloves may impede task performance. If too small, they may cause hand discomfort.
- Ambidextrous or hand-specific gloves: based on clinician preference while considering the length of procedure time
- Tactile sensation: sensitivity should not be significantly reduced. Consider grip, glove thickness, and if the material will be slippery when wet.
SAFE GLOVE USE
Hands are the main pathway of disease transmission. Hand hygiene is the most effective way to prevent the spread of infection, thus it must always be done prior to gloving.8 Routine hand washing with antiseptic hand wash, the use of antiseptic hand rub, or surgical hand antisepsis are all types of hand hygiene.10 Oral health professionals involved in patient care must be able to perform this task correctly and at the right time. The use of gloves never replaces the need for cleaning hands.
Safe glove use includes accuracy in technique when donning (putting on) and doffing (removing) gloves (Figure 1 and Figure 2).11 Gloves are the last item of PPE put on prior to patient treatment and their removal follows a specific sequence. If any of these steps are not followed correctly, there is a high risk of skin contamination. Gloves must never be reused.
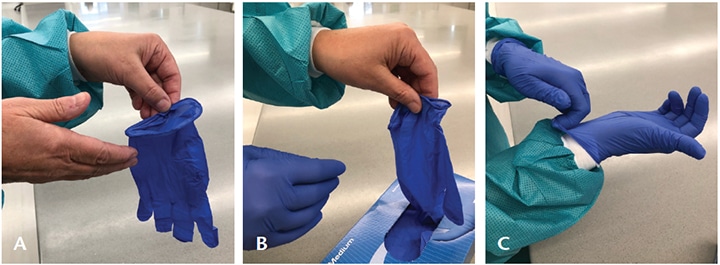
Occasionally during treatment, a clinician must leave a patient to retrieve an instrument or device. If gloves aren’t removed, cross-contamination can occur when touching other surfaces or items. Removing just one glove means that bacteria on that hand could transfer to clean items. The best options are to use a cover glove, or remove both gloves, perform hand hygiene, and then retrieve the item.
Healthy, intact skin is the best defense against pathogen transmission and infection. Selection and use of appropriate medical grade moisturizers and other hand hygiene products are necessary. In an effort to prevent dryness and contact dermatitis often caused by frequent hand hygiene, clinicians often apply lotions. However, lotions with a petroleum-base can hinder the effectiveness of latex gloves.10 Therefore, oral health professionals should consider using only water-based lotions during the workday.
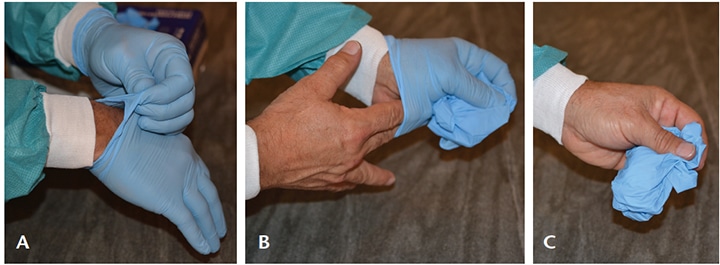
wrist level to remove it (A), without touching the skin of the forearm and peel away from the hand, thus allowing the
glove to turn inside out. Hold the removed glove in the gloved hand and slide the fingers of the ungloved hand inside
between the glove and the wrist (B). Remove the second glove by rolling it down the hand and fold into the first glove.
Discard the removed glove (C).11
Adverse skin conditions can develop due to glove use, chemical exposure, and repeated hand hygiene. Allergies and sensitivities related to gloves should be evaluated by an allergist or dermatologist to pinpoint the cause and direct a course of treatment.
Long nails and hand jewelry can interfere with proper glove selection, cause difficulty when donning, and are likely to create tears and/or punctures. Artificial nails are not recommended as they have been linked to outbreaks of bacterial infections. Natural nail tips should be kept less than a ¼ inch long.12
Gloves stored in areas with considerable moisture, light, or heat are likely to degrade; shelf life varies by type and manufacturer.
CONCLUSION
Gloves are a critical component of PPE necessary for protection against bloodborne pathogens and exposure to other harmful pathogens. Proper hand hygiene along with the proper use of the correct glove will dramatically reduce the likelihood of infection for both oral health professionals and patients. Dental hygienists must have a clear understanding of appropriate glove use in order to be confident they are providing the safest dental visit.
REFERENCES
- Aas JA, Paster BJ, Stokes LN, Olsen I, Dewhirst FE. Defining the normal bacterial flora of the oral cavity. J Clin Microbiol. 2005;43:5721–5732.
- Kramer A, Shwebke I, Kamft G. How long do nosocomial pathogens persist on inanimate surfaces? A systematic review. BMC Infect Dis. 2006;6:130.
- Bardorf MH, Jäger B, Boeckmans E, Kramer A, Assadian O. Influence of material properties on gloves’ bacterial efficacy in the presence of microperforation. Am J Infect Control. 2016;44:1649–1649.
- Partecke LI, Goerdt AM, Langner I, et al. Incidence of micro perforation for surgical gloves depends on duration of wear. Infec Control Hosp Epidemiol. 2009;30:409–414.
- Kohn WG, Collins AS, Cleveland JL, et al. Guidelines for Infection Control in Dental Health-Care Settings—2003. MMWR Recomm Rep. 2003;52(RR-17):1–76. 10.
- United States Food and Drug Administration. Medical Device Bans. Available at: fda.gov/medicaldevices/safety/medicaldevicebans/default.htm Accessed November 20, 2018.
- Organization for Safety, Asepsis and Prevention (OSAP). OSHA and CDC Guidelines: OSAP Interact Training System Workbook. Atlanta: OSAP; 2017.
- Summary of Infection Prevention Practices in Dental Settings: Basic Expectations for Safe Care. Atlanta: Centers for Disease Control and Prevention; 2016.
- Miller CH, Palenik CJ. Infection Control and Management of Hazardous Materials for the Dental Team. 6th ed. St. Louis: Elsevier; 2018.
- United States Centers for Disease Control and Prevention. Hand Hygiene Frequently Asked Questions. Available at: cdc.gov/OralHealth/infectioncontrol/faq/hand.htm. Accessed November 20, 2018.
- World Health Organization. Glove Use Information Leaflet. Available at: who.int/gpsc/5may/Glove_Use_Information_Leaflet.pdf. Accessed November 20, 2018.
- Boyce JM, Pittet D; Healthcare Infection Control Practices Advisory Committee. Society for Healthcare Epidemiology of America. Association for Professionals in Infection Control. Infectious Diseases Society of America. Hand Hygiene Task Force. Guideline for Hand Hygiene in Health-Care Settings: recommendations of the Healthcare Infection Control Practices Advisory Committee and the HICPAC/SHEA/APIC/IDSA Hand Hygiene Task Force. Infect Control Hosp Epidemiol. 2002;23(12 Suppl):S3–S40.
From Dimensions of Dental Hygiene. December 2018;16(12):23–25.


