 OLGA LIS / ISTOCK / THINKSTOCK
OLGA LIS / ISTOCK / THINKSTOCK
Polishing Protocol
There are many factors to consider when choosing the most appropriate polishing agent during prophylaxis.
Dental hygienists have an array of products from which to choose when performing extrinsic stain removal. There are various factors to consider when selecting the correct polishing and/or cleaning agent for each patient. Identifying the types of stain and affected tooth structures is important.1 Dental practices may offer a limited choice of polishing pastes due to clinician preference or cost. Despite this limitation, selecting the appropriate polishing material must be case specific. Abrasive ingredients contained in prophylaxis pastes can remove sound tooth structure. Thus, the abrasiveness of the selected polishing paste needs to be carefully evaluated to ensure that it effectively removes stain without harming tooth structure.2

Social habits and dietary choices promote dental stain accumulation, such as smoking tobacco/marijuana, chewing or dipping tobacco, and consuming red wine, coffee, and tea. Chemical agents such as stannous fluoride and iron also play a role in stain buildup.3 Patients should be advised that toothbrushing with a toothpaste that contains abrasives and chemical agents on a daily basis can help prevent stain accumulation. Table 1 provides a description of common stains and the appropriate technique for their removal.3
While the use of selective polishing—performing prophylaxis only on stained teeth—was once the suggested protocol, it has now been replaced with essential selective polishing. This protocol suggests that polishing the complete dentition is fine as long as the polishing or cleaning agent is chosen based on patients’ individual needs.4
POLISHING RESTORATIONS
When selecting a polishing agent, dental hygienists must consider the tooth structure and restorative materials that may be affected by its use. Clinicians should use polishing agents specially designed for esthetic restorations or mild, nonabrasive toothpastes to maintain their integrity.5 Product descriptions should be read to determine the polishing agent’s indications and compatibility with various dental materials.
Microfilled and nanocomposites, classified according to their filler particle size and shape, offer excellent esthetic characteristics. These biomaterials are often used in anterior regions of the mouth due to their great polishability and lustrous finish. Maintaining this finish is imperative to reducing bacterial and stain adhesion to surfaces that may be roughened by the use of too abrasive polishing agents.6
Zirconia and porcelain materials used for crowns and veneers have a lustrous finish created by the glazing phase during the fabrication process. Such restorations require the use of specially designed polishing agents. Ideally, in-office procedures for amalgam, permanent metal alloy, or gold restorations should use a polishing regimen that incorporates the use of a fine tin oxide slurry to minimize tarnish and reduce bacterial adhesion to the restoration’s surface. These extra steps produce the optimal finish that will improve the longevity of a restoration and promote oral health.7
IMPLANT CARE
There are many factors to consider when caring for implants. According to Humphrey,8 debridement around titanium is limited to plastic, graphite, or gold-plated instruments to prevent damage to the implant. While it is thought that the use of metal scalers and ultrasonic inserts/tips can scratch the surfaces of implants, little in vivo research supports this idea. Plastic instruments are well suited to remove soft plaque deposits, but they are unlikely to displace hard calculus. In fact, some studies show that the metal instrumentation of rough-surface implants results in smoother implant surfaces and less bacterial adherence.9,10
Coarse abrasives can scratch the implant surface. After hard deposits are removed from the implant, the prosthesis can be polished with a rubber cup using a nonabrasive polishing or cleaning paste. Commercial implant pastes or tin oxide are acceptable agents for cleaning implant abutment and prosthesis surfaces.11 Rubber cup polishing with a nonabrasive toothpaste appears to be the least invasive treatment to help retain the smooth surface of the abutment and prosthesis. After polishing, gentle irrigation with water or an antimicrobial solution is recommended to support the health of the peri-implant tissue.12
ABRASIVES
Abrasives used in prophylaxis pastes comprise pumice, zirconium silicate, silicas, and perlite. Manufacturer standards for defining superfine, fine, medium, and coarse grit vary. Factors that affect the abrasiveness of a product include the particle size, particle shape, and hardness.
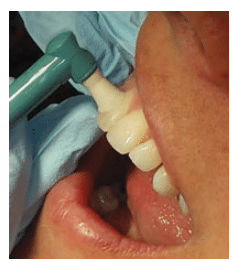
To polish a surface, the particles of the abrasive agent in polishing pastes must be harder than the surface being polished. The abrasiveness of a particle is related to its size and hardness. A very hard abrasive can produce minimal abrasion if its particle size, also known as grit, is very fine to superfine. Particle size affects the surface texture after polishing. Large particles will abrade faster than small particles, while irregularly shaped particles have angular edges that will scratch the tooth surface and accelerate the cleaning process. Hard particles will abrade the surface quickly.13
Small particles elicit the least amount of abrasiveness, while large particles can negatively affect enamel, dentin, cementum, and composites. Although abrasion may not be easily seen, in most situations, the more conservative approach is preferred because coarse grits are more likely to scratch tooth surfaces, thereby enabling stain to easily reaccumulate. Fine grit pastes produce the least amount of surface roughness and also help to maintain the tooth’s natural luster.14 The newest polishing pastes containing perlite incorporate a self-adjusting technology—medium grit transforms to a fine grit during clinical use.
Clinicians should use the least abrasive agent to minimize scratching tooth structures or restorations. When coarse agents are needed, they should be followed by a fine abrasive to create a lustrous finish. When coarse agents are needed to successfully polish areas that are tenaciously stained, they should be followed by a fine grit polishing paste to re-establish the tooth’s natural luster.15
Cleaning agents are another option for tooth and restorative surfaces. These products are nonabrasive and will not scratch restorative surfaces. For patients who request polishing but do not exhibit stain, a cleaning agent may be the best option.16
TECHNIQUE
Appropriate clinical prophylaxis techniques must be employed to minimize alterations to existing smooth surfaces and reduce the retention of biofilm and deposits. Polishing agents should be selected based on material composition and stain accumulation. One study that compared pumice-based prophy pastes with perlite-based prophy paste when applied to tooth-colored biomaterials found that both roughened restorative materials to varying degrees.17 Roughened restorative materials can predispose teeth to recurrent decay, gingivitis, or loss of periodontal attachment. The research indicated that most restorative materials have less abrasive resistance than dentin or enamel. These data emphasize the importance of choosing the correct paste for the task at hand.17
The quantity and method of prophy paste application influence the rate at which a surface is polished. A greater amount of abrasive applied to the tooth surface will hasten the polishing procedure. Dry agents, as opposed to slurry abrasives, will increase the rate of abrasion and are more prone to elicit surface roughness. Conversely, abrasive agents applied to a moist surface will produce a smoother effect. Polishing technique impacts the rate of abrasion. Applying agents at a high speed will create more abrasion to tooth structures and generate heat. High pressure and increased polishing duration will hasten abrasion.
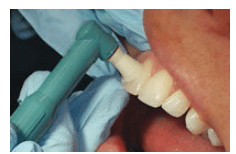
Light, intermittent rubber cup strokes are recommended. Proper rubber cup adaptation is important. Apply the polishing cup with a smooth, continuous motion, flaring the edges of the cup to adapt to proximal areas. Use oblique strokes from cervical to occlusal/incisal surfaces (Figure 1, Figure 2, and Figure 3). Excessive polishing of crown surfaces may remove fluoride-rich enamel layers. Polishing exposed dentin is not recommended, as increased sensitivity and physical damage may result. If the edge of the rubber cup is applied to dentin or root surfaces, it may create scratches or grooves rather than a smooth surface. Resin restorations are also easily damaged.19 Clinicians should avoid iatrogenic damage to tooth surfaces by utilizing proper polishing techniques.19
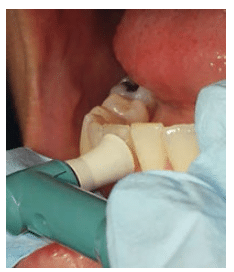
PROPHY ANGLES
Prophy angle quality is another consideration. Most disposable prophy angles offer latex-free rubber cups in various designs. Features may include outer spiral ridges on the cup to improve interproximal cleaning and flared cup designs that reduce splatter and aerosol production. Cups are available in soft or firm textures. Firm cups are less flexible and may adversely affect the pressure applied to tooth structures. Additionally, cup design can impact the amount of agent applied to tooth structure. Webbed prophy cups retain less paste than ribbed cups. In cases where using less polishing paste is indicated, webbed prophy cups may reduce abrasiveness. Recent technology has enabled the creation of prophy angles that clean and polish without the need for polishing paste because the stain removal agent is incorporated into the cup itself. This may improve operator visibility, help save time, and better support infection control efforts by reducing splatter.20
Polishing cup-to-tooth contact times vary significantly. The average contact time reportedly ranges from 5 seconds to 10 seconds with speeds varying from 1,500 revolutions per minute (rpm) to 4,000 rpm.20 The plastic gears of disposable prophy angles operate at an average speed of 3,000 rpm. Variations in prophy angle design may affect overall effectiveness of stain removal. Poorly designed prophy angles may require more time and pressure to polish and necessitate the use of more abrasive pastes. This may also lead to operator fatigue.21
ERGONOMIC CONSIDERATIONS
Dental handpiece design impacts clinician ergonomics. Ideally, handpieces should be lightweight and well balanced, with a hose length as short as possible while still maintaining maneuverability. Handpieces configured with extra hose length add weight and bulkiness. Retractable or coiled hoses should be avoided, as they hinder manipulation of the handpiece when the hose is stretched.22 The tension in the hose can be transferred to the wrist and arm, creating awkward motions that may lead to muscular fatigue. Repetitive movements of the wrist increase stress to the hand and wrist and may lead to inflammation of the tendon.23
Handpiece selection is a personal choice based on performance and operator comfort. A handpiece with a pliable hose and swivel mechanism in the barrel can ease rotation of the device and minimize operator effort. A cordless handpiece may also help reduce mechanical stress to the hand and wrist because there is no cord to restrict the clinician’s wrist rotation.23
CONCLUSION
Dental hygienists are responsible for providing evidence-based treatment, remaining current with innovative equipment selection, and providing input on the purchasing decisions for their dental practices. Dental hygienists can offer optimal care by tailoring polishing options to more precisely meet individual patient needs.
References
- Darby MS, Walsh MM. Dental Hygiene Theory and Practice. 3rd. St. Louis: 2009:512–522.
- Chakravarthy PK, Acharya S. Efficacy of extrinsic stain removal by novel dentifrice containing papain and bromelain extracts. J Young Pharm. 2012;4:245–249.
- Wilkins E. Clinical Practice of the Dental Hygienist. 11th ed. Philadelphia: Lippincott Williams; 2013:306–309.
- Barnes C. Shining a new light on selective polishing. Dimensions of Dental Hygiene. 2012;10(3):42–44.
- Barnes C. The qualitative and qualitative evaluation of the abrasiveness of an abrasive-impregnated rubber prophylaxis polishing cup and two polishing pastes when used on aesthetic restorative materials. Gen Dent. 2011;59:433–473.
- Barnes C. Polishing esthetic restorative materials. Dimensions of Dental Hygiene. 2010;8(1):24–28.
- Dais J. Polishing procedures. Dimensions of Dental Hygiene. 2006;4(6):22–24.
- Humphrey S. Implant maintenance. Den Clin North Am. 2006;50:463–478.
- Duarte PM, Reis AF, de Freitas PM, Ota-Tsuzuki C. Bacterial adhesion on smooth and rough titanium surfaces after treatment with different instruments. J Periodontol. 2009;80:1824–1832.
- Hempton T. Choosing an instrument for implant maintenance. Dimensions of Dental Hygiene. 2016;14(1):62.
- Pattison A, Sumi J. Post-surgical implant care. Dimensions of Dental Hygiene. 2015;13(5);24–29.
- Gulati M, Govila V, Anand V, Anand B. Implant maintenance: a clinical update. International Scholarly Research Notices. 2014;1–8.
- Neme A, Frazier K, Roeder L, Debner T. Effect of prophylactic polishing protocols on the surface roughness of esthetic restorative materials. Oper Dent. 2002;27:50–58.
- Pence S, Chambers D, VanTets I, Wolf R, Pfeiffer D. Repetitive coronal polishing yields minimal enamel loss. J Dent Hyg. 2011;85:348–357.
- Lutz F, Imfeld T. Advances in abrasive technology-prophylaxis pastes. Compend Contin Educ Dent. 2002;23:61–68.
- Barnes C. Follow the evidence. Dimensions of Dental Hygiene. 2015;13(11):36–38.
- Warren D, Colescott T, Henson H, Powers J. Effects of four prophylaxis pastes on surface roughness of a composite, a hybrid ionomer, and a compomer restorative material. J Esthet Restor Dent. 2002;14:245–251.
- Jefferies SR. Abrasive finishing and polishing in restorative dentistry: A state-of-the-art review. Dent Clin N Am. 2007;51:379–397.
- Vitalariu A. Effect of surface polishing and glazing on the roughness of the dental acrylic resins. Annals of International Danube Adria Association for Automation & Manufacturing. 2010:77–78.
- Gladwin M, Bagby M. Clinical Aspects of Dental Materials. 4th. Philadelphia: LWW; 2013:205–220.
- LaCross I. Posing the polishing question. Dimensions of Dental Hygiene. 2007;5(6):20–23.
- Ergonomic and Disability Support Advisory Committee. An Introduction to Ergonomics: Risk Factors, MSDs, Approaches and Interventions. Available at: rgpdental.com/pdfs/topics_ergonomics_paper(2).pdf. Accessed February 11, 2016.
- Cooper M, Wiechmann L. Essentials of Dental Hygiene. Upper Saddle River, New Jersey: Prentice Hall; 2005:281–294.
From Dimensions of Dental Hygiene. March 2016;14(03):38–40,42.

