 ENRIQUE MICAELO SANCHEZ/ISTOCK/GETTY IMAGES PLUS
ENRIQUE MICAELO SANCHEZ/ISTOCK/GETTY IMAGES PLUS
Oral Health Effects of Breast Cancer Therapy
Remaining up to date on breast cancer treatments and their implications is integral to advising this population on maintaining their oral health.
This course was published in the June 2021 issue and expires June 2024. The authors have no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- 1. Describe available breast cancer treatments.
- Identify potential oral complications of breast cancer treatment.
- Discuss oral therapies to aid patients undergoing breast cancer treatment manage their oral side effects.
Breast cancer is caused by uncontrolled division of abnormal cells, resulting in tumor formation. Approximately one in eight women will develop a form of breast cancer, making it the second most common cancer among women in the United States.1 The treatment of breast cancer can cause deleterious side effects, and oral health professionals should be prepared to help patients maintain their oral health and help mitigate negative signs and symptoms.
Breast cancer survival rates vary by stage, with stage 0 to stage I offering a 100% 5-year survival rate; stage II a 93% 5-year survival rate; stage III a 72% 5-year survival rate, and stage IV a 22% 5-year survival rate.2 Treatment options—which include chemotherapy, radiation therapy, hormone therapy, surgical procedures, and pain management—are based on the stage at diagnosis, type of cancer, genetic factors, and cancer progression. Gene therapy is also under investigation as a breast cancer treatment option. Several treatment options are frequently employed to ensure optimal long-term outcomes.
Chemotherapy uses a mixture of drugs to stop the cancer’s spread, slow its growth, kill cancer cells, and relieve symptoms.2 Adjuvant therapy is prescribed post-cancer surgery to kill any remaining cancer cells and lower the risk of the breast cancer returning. Neoadjuvant therapy is given before surgery to shrink the tumor so that a less invasive surgery can be done. Like adjuvant therapy, neoadjuvant therapy can also help prevent breast cancer from returning.3
Drugs used for adjuvant and neoadjuvant therapy are anthracyclines, such as doxorubicin and epirubicin; taxanes, such as paclitaxel and docetaxel; 5-fluorouracil or capecitabine; cyclophosphamide; and carboplatin.3 Chemotherapy drugs used for metastasized breast cancer are taxanes, such as paclitaxel, docetaxel, and albumin-bound paclitaxel; anthracyclines (doxorubicin, pegylated liposomal doxorubicin, and epirubicin); platinum agents (cisplatin and carboplatin); vinorelbine; capecitabine; gemcitabine; ixabepilone; and eribulin.4 Several chemotherapy drugs are often used to enhance effectiveness, which may exacerbate oral side effects.3
Radiation therapy exposes a specific treatment area to high-energy radiation to destroy cancer cells.2 The two main types are external beam radiation therapy (EBRT) and brachytherapy. The most common form of radiation, EBRT focuses the radiation beam on the outside of the body. EBRT can be focused on whole breast radiation, accelerated partial breast radiation, chest wall radiation, or lymph node radiation. Brachytherapy, or internal radiation therapy, places radioactive seeds or pellets into the breast tissues for short periods where the cancer has been surgically removed.5
A form of adjuvant therapy, hormone treatment is another option to slow or stop the growth of cancer cells. It includes oral medications, such as tamoxifen, raloxifene, and toremifene, which are classified as selective estrogen receptor modulators (SERMs), and aromatase inhibitors (AIs), including exemestane, anastrozole, and letrozole.2,6 Depending on the type of medicine used, the mechanism of action is different. SERMs attach to the hormone receptor within the cancer cell and block estrogen from attaching to the receptor. In the case of AIs, the level of estrogen is reduced, preventing cancer cells from growing. While SERMs are used in pre- and post-menopausal women, AIs are used only in post-menopausal women.
Monoclonal antibodies such as trastuzumab; signal transduction inhibitors such as lapatinib; selective estrogen receptor degraders such as fulvestrant; and luteinizing hormone-releasing hormones analogs, such as goserelin and leuprolide, may be used.7 Clinical trials on gene therapy, which replaces the tumor suppressor genes in order to help prevent cancer from developing, are ongoing. Pain medication and management are also important, as 90% of patients with cancer experience pain from the cancer itself and treatment-related side effects.2
In addition, several surgical procedures are used to treat breast cancer. Mastectomy is the surgical removal of the entire breast, which can be disfiguring and debilitating. A lumpectomy is the surgical removal of the breast cancer tumor and surrounding healthy breast tissue, which is the most breast-conserving surgical option available. A lymphadenectomy is the surgical removal of some or all of the surrounding lymph nodes to be tested; this usually follows a mastectomy or lumpectomy to determine if the cancer has spread. Following a mastectomy procedure, women might consider breast augmentation or breast implants as means to reconstruct the post-surgical sites.2
ORAL SIDE EFFECTS OF BREAST CANCER TREATMENT
All cancer treatment options cause side effects, and some may be worsened when a combination of therapies are employed simultaneously. Radiation may cause fatigue, swelling, heaviness in the breast, or a sunburned appearance. These side effects may limit post-operative mobility, hindering oral self-care.2 While breast cancer surgery may not directly cause oral side effects, the post-operative period can impair the ability to properly practice oral self-care.
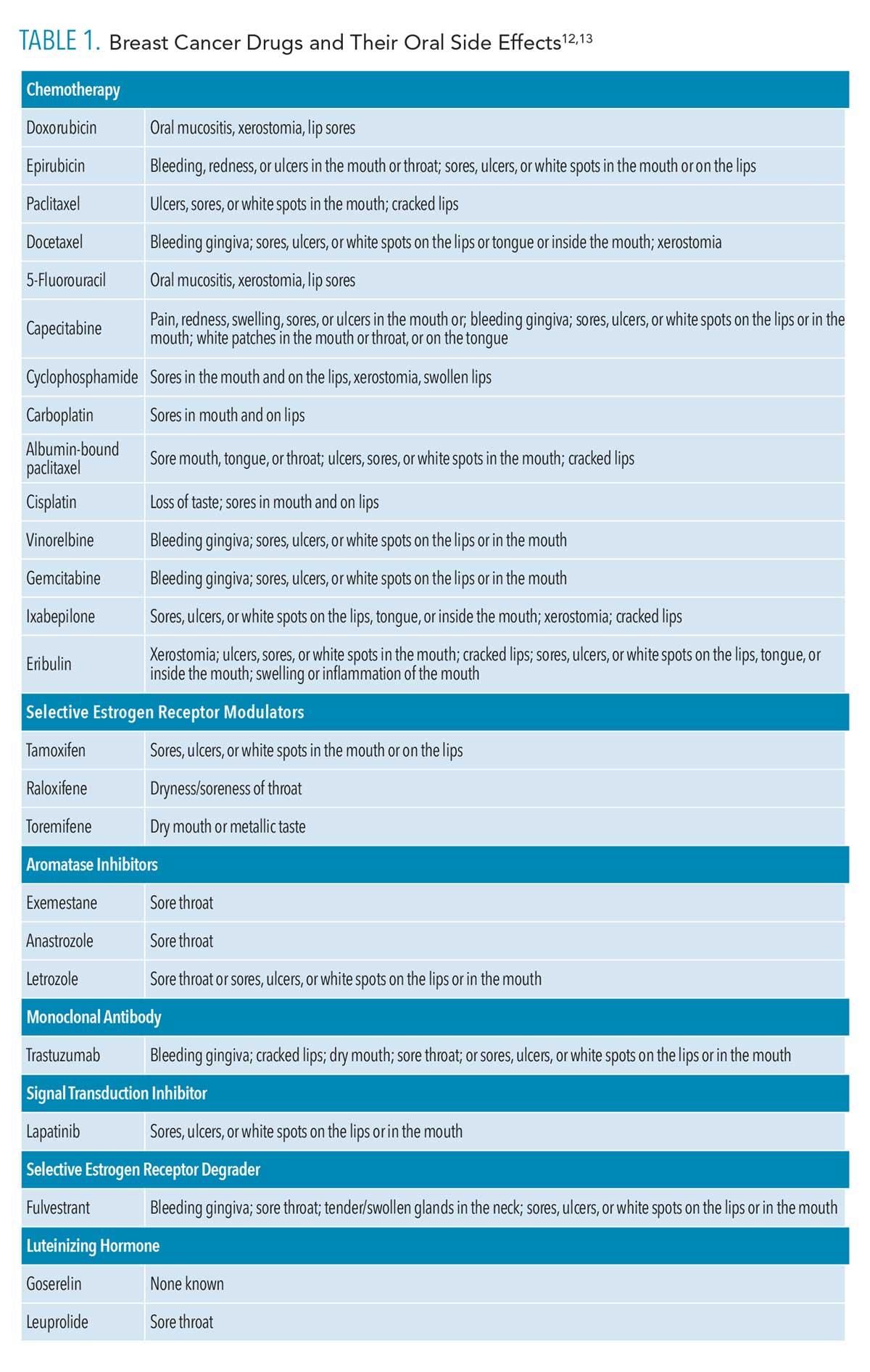
Chemotherapy, hormonal therapy, and other adjuvant breast cancer medicines can cause oral mucositis, stomatitis, dysphagia, xerostomia, pharyngitis, taste alterations, nausea, and vomiting (Table 1, page 42).8–13 Missing teeth, apical lesions, and increased risk of infection and periodontal diseases are also possible.14–16 Oral mucositis lesions are irregular ulcerations with white pseudomembranes that may be large in size and often are accompanied by severe pain, compromising the patient’s nutrition and oral hygiene. A patient’s risk for local and systemic infections may increase because of the treatment-induced immunodeficiency and damaged oral mucosa.17
Xerostomia is a significant oral concern, as a reduction in saliva flow decreases the oral pH and significantly increases the build-up of plaque biofilm, leading to dental caries and/or periodontal diseases. In severe cases, xerostomia may also contribute to oral candidiasis.18 Saliva serves several key functions in the oral cavity, including the provision of antimicrobial properties, control of pH levels, removal of food debris from the oral cavity, lubrication of the oral cavity, remineralization of the enamel, and maintenance of the integrity of the oral mucosa. Xerostomia can affect nutrition, as well as dental, systemic, and psychological health.18 Xerostomia may cause a constant sore throat, burning sensation, difficulty speaking, problems swallowing, hoarseness of voice, dry nasal passages, dysgeusia, glossodynia, parotid gland enlargement, inflammation, cheilitis, inflammation or ulcers of the tongue and buccal mucosa, sialadenitis, cracking and fissuring of the oral mucosa, and oral malodor.10,18
After active treatment is completed, patients may require maintenance therapy, or adjuvant therapy, in which estrogen signaling is halted to suppress secondary tumor formation. A study was conducted to determine the periodontal changes in post-menopausal breast cancer survivors using AIs (eg, anastrozole, exemestane, letrozole) compared to post-menopausal women without breast cancer.19 AIs are a form of adjuvant endocrine therapy to reduce the risk of breast cancer recurrence. The investigators concluded that AI therapy negatively impacted periodontal health by increasing clinical attachment loss, pocket depth, and alveolar bone height loss, among post-menopausal women with breast cancer.19
DENTAL HYGIENE CONSIDERATIONS
Patients undergoing treatment for breast cancer need to maintain a regular recare schedule to support their oral health.11 Oral recare intervals may need to be modified to accommodate different cancer treatment phases. Dental hygienists should be prepared to provide tailored education and recommendations to help patients minimize the long-term effects of cancer treatment.
Oral mucositis is a common oral side effect of chemotherapy, with 56% of patients with breast cancer experiencing it.17 Accurate diagnosis and prompt treatment of oral mucositis are crucial. Patients should be instructed to brush and floss carefully while also incorporating an alcohol-free mouthrinse with saline or sodium bicarbonate.17 Soaking an extra-soft bristle toothbrush in warm water before brushing gently for 2 minutes can help ease sensitivity and irritation to the inflamed tissues. Ingredients such as hydrogen peroxide, iodine, and thyme derivatives may irritate already sensitive oral tissues. Tobacco; alcohol; and spicy, acidic, and hot foods may also cause further irritation. Anesthetic mouthrinses with lidocaine or benzocaine, topically administered gels, and calcium phosphate oral health products may help alleviate oral pain in this patient population.17
As patients undergoing breast cancer treatment may experience decreased salivary flow, they need to be educated on how best to reduce their risk for caries and periodontal diseases.20 Dental hygienists can recommend treatments to address dry mouth symptoms. Barbe et al21 found that a mouthrinse containing glycerin and a mouthrinse containing betaine, taurine, polyvinylpyrrolidone, and sodium hyaluronate significantly reduced the dryness of oral tissues, although relief was temporary. Skrinjar et al22 demonstrated that saliva substitutes improved the quality of life for patients with xerostomia
A variety of options are available for caries prevention. Casein phosphopeptide-amorphous calcium phosphate can be applied in small amounts directly on the tooth surface with a fingertip or cotton swab after brushing, or by trays overnight. Tricalcium phosphate and a high concentration of fluoride can also be helpful, although not recommended for overnight use. Calcium sodium phosphosilicate is recommended as a toothpaste application or for overnight use. Patients could also chew gum or mints containing xylitol up to five times a day for 5 minutes each time.20
The American Dental Association (ADA) Council on Scientific Affairs recommends the at-home use of topical fluoride agents such as 0.5% fluoride gel or paste, and 0.09% fluoride mouthrinses for caries prevention.23 Several prescription-strength fluoride products are available. Kau et al24 compared 1.1% sodium fluoride, 0.21% sodium fluoride, and tricalcium phosphate with fluoride, and concluded that, while all products were effective in reducing demineralization, 1.1% sodium fluoride demonstrated slightly better outcomes.
In patients with breast cancer at high risk for caries, professionally applied fluoride should be administered once every 3 months.20 The ADA Council on Scientific Affairs also recommends professionally applied fluoride treatments, specifically 2.26% fluoride varnish or 1.23% acidulated phosphate fluoride (APF) gel, which can be applied at prophylaxis and periodontal maintenance visits.23 However, given APF gel’s acidic nature, it may further irritate oral tissues in individuals who already experience oral mucositis symptoms and/or xerostomia.
CONCLUSION
Studies show that patients undergoing breast cancer treatments may experience oral side effects. Dental hygienists must be able to adequately educate this patient population on strategies for maintaining their oral health. Evidence supports the use of at-home and professionally applied products to help alleviate possible oral side effects related to breast cancer treatment. Staying abreast of treatment options and their oral side effects is key to providing the best possible patient care.
REFERENCES
- National Institute of Environmental Health Sciences. Breast Cancer. Available at: niehs.nih.gov/health/topics/conditions/breast-cancer/index.cfm. Accessed May 6, 2021.
- Peart O. Breast intervention and breast cancer treatment options. Radiol Technol. 2015;86:535–59M.
- American Cancer Society. Chemotherapy for Breast Cancer. Available at: cancer.org/cancer/breast-cancer/treatment/chemotherapy-for-breast-cancer.html. Accessed May 6, 2021.
- American Society of Clinical Oncology. Breast Cancer: Stages. Available at: cancer.net/cancer-types/breast-cancer/stages. Accessed May 6, 2021.
- American Cancer Society. Radiation for Breast Cancer. Available at: cancer.org/cancer/breast-cancer/treatment/radiation-for-breast-cancer.html. Accessed May 6, 2021.
- Breastcancer.org. Selective Estrogen Receptor Modulators. Available at: breastcancer.org/treatment/hormonal/serms. Accessed May 6, 2021.
- Breastcancer.org. Monoclonal Antibodies and Their Side Effects. Available at: cancer.org/treatment/treatments-and-side-effects/treatment-types/immunotherapy/monoclonal-antibodies.html. Accessed May 6, 2021.
- Watters AL, Epstein JB, Agulnik M. Oral complications of targeted cancer therapies: a narrative literature review. Oral Oncol. 2011;47:441–448.
- Taichman LS, Van Poznak CH, Inglehart MR. Oral health-related concerns, behavior, and communication with health care providers of patients with breast cancer: impact of different treatments. Spec Care Dentist. 2018;38:36–45.
- Taichman LS, Van Poznak CH, Inglehart MR. Self-reported oral health and quality of life of postmenopausal breast cancer survivors on aromatase inhibitors and women without cancer diagnoses: a longitudinal analysis. Support Care Cancer. 2016;24:4815–4824.
- Chemocare. Lapatinib. Available at: chemocare.com/chemotherapy/drug-info/lapitinib.aspx. Accessed May 6, 2021.
- Poulopoulos A, Papadopoulos P, Andreadis D. Chemotherapy: oral side effects and dental interventions a review of the literature. Stomatological Dis Sci. 2017;1:35-49.
- Drugs.com. Available at: drugs.com. Accessed May 6, 2021.
- Willershausen I, Schmidtmann I, Azaripour A, et al. Association between breast cancer chemotherapy, oral health and chronic dental infections: a pilot study. Odontology. 2019;107:401–408.
- Thorpe D, Stringer A, Butler R. Chemotherapy-induced mucositis: the role of mucin secretion and regulation, and the enteric nervous system. Neurotoxicology. 2013;38:101–105.
- Söder B, Yakob M, Meurman JH, et al. Periodontal disease may associate with breast cancer. Breast Cancer Rest Treat. 2011;127:497–502.
- Seiler S, Kosse J, Liobl S, et al. Adverse event management of oral mucositis in patients with breast cancer. Breast Care (Basel). 2014;9:232–237.
- Oral Cancer Foundation. Xerostomia. Available at: oralcancerfoundation.org/complications/xerostomia. Accessed May 6, 2021.
- Eagle I, Benavides E, Eber R, et al. Periodontal health in breast cancer patients on aromatase inhibitors versus postmenopausal controls: a longitudinal analysis. J Clin Periodontal. 2016;43:659–667.
- Su N, Marek C, Ching V, et al. Caries prevention for patients with dry mouth. J Can Dent Assoc. 2011;77:b85.
- Barbe AG, Schmidt-Park Y, Hamacher S, et al. Efficacy of GUM Hydral versus Biotène Oralbalance mouthwashes plus gels on symptoms of medication-induced xerostomia: a randomized, double-blind, crossover study. Clin Oral Invest. 2018;22:169–180.
- Skrinjar I, Boras VV, Bakale I, et al. Comparison between three different saliva substitutes in patients with hyposalivation. Clin Oral Invest. 2015;19:753–757.
- Weyant RJ, Tracy SL, Anselmo T, et al. Topical fluoride for caries prevention: executive summary of the updated clinical recommendations and supporting systematic review. J Am Dent Assoc. 2013;144:1279–1291.
- Kau CH, Wang J, Palombini A, et al. Effect of fluoride dentifrices on white spot lesions during orthodontic treatment: a randomized trial. Angle Orthodontist. 2019;89:365–372.
From Dimensions of Dental Hygiene. June 2021;19(6):40-43.



