 ZLIKOVEC / ISTOCK / THINKSTOCK
ZLIKOVEC / ISTOCK / THINKSTOCK
Improving Periodontal Health
Nonsurgical periodontal therapy—both traditional practices and recent innovations—remains key to improving oral health outcomes.
This course was published in the July 2016 issue and expires July 31, 2019. The authors have no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Describe the importance of self-care in supporting periodontal health.
- Identify the chemotherapeutics and adjunctive therapies used to maintain periodontal health.
- List the factors that influence the effectiveness of scaling and root planing.
- Explain the role of periodontal maintenance in nonsurgical periodontal therapy.
INTRODUCTION
Antimicrobial therapies play key roles in preventing and treating periodontal diseases. Hours of meticulous periodontal therapy alone cannot achieve the oral health goals we set for our patients. Self-care and the use of chemotherapeutics are critical components of maintenance therapy. This article provides a current review of the scientific evidence exploring benefits and limitations of nonsurgical periodontal therapy, the use of chemotherapeutics and adjunctive therapies, and the rationale for periodontal maintenance. The Colgate-Palmolive Company is delighted to have provided an unrestricted educational grant to support “Improving Periodontal Health” in collaboration with the American Academy of Periodontology.
— Matilde Hernandez, DDS, MS, MBA
Scientific Affairs Manager
Professional Oral Care
Colgate Oral Pharmaceuticals
FROM THE AMERICAN ACADEMY OF PERIODONTOLOGY
With professional care and expertise, a patient can regain health, function, and esthetics after a bout with periodontal disease. But the key to maintaining what was once at risk is often out of the specialist’s hands, and hinges on the patient’s daily at-home habits. As such, the periodontist’s and dental hygienist’s job is about more than treating and arresting disease—it’s also about educating patients on what they can do to keep future infection at bay and motivating them to take action. Here, educator Maria L. Geisinger, DDS, MS, offers insights on how anti-infective treatment—when coupled with proper at-home maintenance and regular office visits—is essential to long-term patient health. The American Academy of Periodontology—which advises patients to brush twice a day, floss daily, and receive a comprehensive periodontal evaluation annually— is proud to work with Dimensions of Dental Hygiene and Colgate-Palmolive to bring you information that can aid in keeping your patients’ gums and smiles healthy for a lifetime.
— Wayne Aldredge, DMD
President, American Academy of Periodontology
Periodontal diseases affect approximately 47% of adults in the United States.1 The primary etiology of periodontal diseases is bacterial plaque, or biofilm.2,3 Therefore, the effective and regular removal of biofilm and plaque-retentive factors is critical to achieving optimal periodontal health.4 The success of periodontal therapy depends on removing biofilm from tooth surfaces, periodontal tissues, mucosal surfaces, the tongue, and other niches within the oral cavity and keeping subsequent biofilms under control.5
Nonsurgical periodontal therapy aims to produce root surfaces that are biologically acceptable so that healthy epithelial attachments can be established and maintained.6 It typically includes oral hygiene education, improved self-care, professional debridement of tooth surfaces, and sometimes the use of adjunctive antimicrobial agents. Establishing and maintaining optimal oral hygiene is critical to the long-term success of both nonsurgical and surgical periodontal therapies.
Microbial plaque control is an effective method of preventing and treating gingivitis2 and it plays a critical role in the treatment and prevention of periodontitis.7 Microbial biofilm begins to form almost immediately after cessation of oral hygiene measures and must be removed, on average, at least once every 48 hours to prevent inflammation in periodontally healthy patients.8 The American Dental Association (ADA) recommends that patients brush at least twice per day to effectively remove microbial biofilm and prevent gingivitis.9 While toothbrushing can remove some microbial biofilm, it is often inadequate in interproximal areas. Thus, the use of interdental cleaning aids, such as dental floss and interdental brushes, is often essential to remove plaque effectively in interproximal areas.10,11 Finally, dentifrices and mouthrinses, as well as patient-delivered topical medications may contain chemicals that inhibit plaque microbes for some time after use. Topical medications can also be important adjuncts to manual plaque removal.12
Educating and motivating patients to perform adequate oral hygiene is challenging. Patients have been shown to overestimate their brushing times13 and often to fail to adequately remove microbial biofilm, even following demonstrations of optimal methods.14 Social/esthetic factors, such as fresh breath, an attractive smile, and avoiding disease, are typically the most significant motivators for performing oral hygiene measures.15 Repeated assessment and professionally guided modification of self oral hygiene measures are critical to the successful prevention and treatment of caries and periodontitis.4
Motivational interviewing can be used to emphasize the importance of plaque control in oral health.16 It is a person-centered, goal-directed method that fosters a patient’s intrinsic motivations for positive change.17 Motivational interviewing is based on the use of open-ended questions, affirmations, reflections, and summary (Table 1). Open-ended questions are used to elicit information from patients and typically begin with an interrogative such as “how,” “what,” or requesting a description, such as, “Tell me about…” Affirmations are used to encourage positive health behaviors that patients already practice. Reflections serve to prompt further discussion to better clarify a patient’s intentions. Summary demonstrates that the interviewer is an active listener and sets the stage for change.18 Motivational interviewing may induce change in patients’ health behaviors, including improved oral hygiene. Even a single session of motivational interviewing has been shown to improve gingival bleeding scores and plaque index.19,20
THE USE OF CHEMOTHERAPEUTICS
The use of adjunctive chemotherapeutics delivered in dentifrices may offer additional benefits. For example, stannous fluoride provides antimicrobial properties.21 Dentifrices containing stannous fluoride, however, may cause temporary staining of teeth.22 Dentifrices containing pyrophosphates may interfere with the crystal formation in calculus, thus reducing the formation of supragingival calculus by more than 30%.23 They do not affect subgingival calculus formation.
The anti-gingivitis agent triclosan has antimicrobial properties and may reduce gingivitis when compared with control dentifrice.24 Questions have arisen regarding the safety of triclosan.25 However, many regulatory agencies, including the US Food and Drug Administration, Health Canada, and the Scientific Committee on Consumer Safety of the European Commission, have reviewed the data surrounding triclosan’s safety and effectiveness in products and cosmetics and deemed the ingredient safe for use where a health benefit has been demonstrated.26
Numerous studies have suggested that systemic statin medications may reduce clinical signs of gingival inflammation and periodontal disease progression.27 The mechanism of this action is likely a reduction in the expression of lipopolysaccharide-induced matrix metalloproteinases (MMPs).28 Several randomized controlled trials have demonstrated decreased signs of gingival inflammation following topical application of a simvastatin or atorvastatin gels as compared with placebos.29–31 Other investigations are underway to determine long-term effectiveness.
Dentifrice with edathamil has shown promise in reducing gingivitis levels and decreasing plaque formation.32 Edathamil is thought to help prevent the attraction of bacteria to the dental pellicle. Multicenter trials are now underway to better elucidate edathamil’s mechanism of action and to determine its effect on inflammation and the subgingival microbiome.
Mouthrinses have been shown to be effective adjuncts that reduce plaque and gingival inflammation.33,34 Bisguanide antiseptics (including chlorhexidine gluconate) provide broad spectrum antimicrobial effects while demonstrating high substantivity.35 Long-term use, however, is associated with tooth staining and alterations in taste, which may make compliance challenging. Moreover, beneficial oral microflora may be adversely affected by the long-term use of antiseptic oral mouthrinses.36,37 Other mouthrinses, including those containing propolis, oxygenating agents, amine alcohols, metal ions, triclosan, probiotics, and salicylamide, offer antimicrobial properties.35–38 A propolis-containing mouthrinse demonstrated a reduction in papillary bleeding greater than chlorhexidine mouthrinse or placebo after 28 days of use.36 A salicylamide-containing rinse has demonstrated antimicrobial activity against a wide spectrum of bacterial species.39 Additionally, the use of a probiotic mouthrinse to promote beneficial bacteria and eliminate pathogenic bacteria has also compared favorably with the use of a broad spectrum antimicrobial mouthrinse.40 As technologies permit more detailed evaluations, the development of new mouthrinses with increased effectiveness and decreased side effects may be possible.41
SCALING AND ROOT PLANING
Debridement of tooth surfaces to remove local etiologic factors is the primary goal of mechanical periodontal therapy.42 Scaling and root planing is often the first treatment recommended for periodontitis cases6,43 and has produced beneficial effects for sites with deep probing depths (Table 2).43 The probing depth reduction seen after scaling and root planing is caused by reduced edema, repair of junctional epithelium, and greater tissue resistance to probe penetration at the base of sulci.44 A decrease in subgingival microbes, including a reduction of pathogens, also occurs.43,44 Maintaining these clinical improvements, however, depends on sustained plaque removal by the patient at home.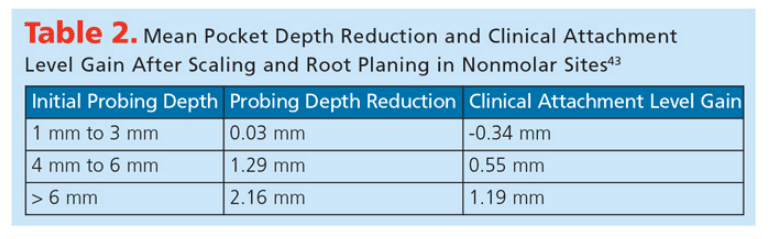
The success of debridement depends on the severity of the initial defect, operator skill, time expended, and the design of instruments.7,45,46 While studies suggest that similar results can be achieved using manual, sonic, or ultrasonic scalers, the time expended during debridement with manual instruments alone can range up to 50% longer when compared with the combined use of manual and powered scalers.47,48
Various protocols for the delivery of mechanical subgingival debridement have been proposed. Two systematic reviews have indicated slightly greater probing depth reduction at sites with deep probing depths (pocket depth > 6 mm) after full-mouth scaling and root planing performed in one appointment or over a brief interval when compared with quadrant scaling and root planing provided over longer intervals.49,50 These protocols may be particularly effective in patients with aggressive periodontitis and/or patients who have significant underlying systemic illnesses that impair healing.49,50
ADJUNCTIVE THERAPIES
Two protocols using laser technologies—laser-assisted new attachment procedure (LANAP) and photodynamic therapy—have been evaluated as adjuncts that may help increase the effectiveness of scaling and root planing. LANAP, which uses an Nd:YAG laser in conjunction with meticulous scaling and root planing and occlusal adjustment, has demonstrated a reduction of pocket depths (PD) and in clinical attachment levels (CAL) and limited evidence of periodontal re-attachment that includes new connective tissue.51,52 No studies have compared the LANAP procedure to a control procedure, such as scaling and root planing and occlusal adjustment, so as to better assess the additive effects, if any, associated with laser use. Nevertheless, the US Food and Drug Administration recently accepted the LANAP protocol as a regenerative approach that may result in the growth of new bone, cementum, and periodontal ligament on a previously diseased root surface.
Photodynamic therapy has demonstrated destruction of putative periodontal pathogens in vivo and improvement in PD and CAL at 3 months, but not 6 months after therapy when compared with scaling and root planing alone.53,54 The long-term benefits of either LANAP or photodynamic therapy are not yet known, but they may be effective in some cases.
A dental endoscope was developed and introduced in 1999 to provide magnified imaging below the marginal gingiva for diagnosis and as an aid in the treatment of periodontitis. Root deposits and caries were identified in situ using the dental endoscope and confirmed with direct visualization after extraction.55 Furthermore, calculus removal on single-rooted teeth was more effective following scaling and root planing with the use of the endoscope than without.56 However, there were no statistically significant outcome differences reported on multirooted teeth that received nonsurgical periodontal therapy with or without the endoscope.57 Clinical results have also varied with regard to probing depth reductions and residual levels of cytokines in gingival crevicular fluid.55,58
Local antimicrobial delivery in the treatment of periodontal diseases has been shown to be effective in the reduction of PD and CAL gain when applied after scaling and root planing.7,59 Patients with chronic periodontitis and isolated recurrent pockets ? 5mm with bleeding on probing are most frequently prescribed this type of therapy.7,60,61 A review of current evidence suggests that the use of chlorhexidine chips, locally delivered doxycycline, and locally delivered minocycline microspheres may provide benefit in patients with chronic periodontitis when used as an adjunct to mechanical debridement.62 Current literature suggests similar adjunctive CAL gains and improvements in clinical parameters between doxycycline and minocycline therapies for initial probing depths of 4 mm to 6 mm.63 The data on the efficacy of the chlorhexidine chip is variable, but those studies that report positive results have similar effect magnitudes as the other adjunctive treatments discussed.64 The use of any of these adjuncts should be tailored to individual patient needs and considered in terms of cost-effectiveness.
 Subantimicrobial dose doxycline (SDD) is prescribed 20 mg twice daily and has been shown to be an effective adjunct in the reduction of PD and CAL gain over 3 months to 9 months.65,66 SDD’s mechanism of action involves the local downregulation of MMPs and the upregulation of tissue inhibitors of matrix metalloproteinases67,68 and may also demonstrate reductions in prostaglandin and pro-inflammatory cytokine levels.67,68 Patients’ overall health may impact the effectiveness of this treatment. A modest benefit has also been reported when systemic antibiotics are used as adjuncts to scaling and root planing in chronic periodontitis patients. Even more robust benefits are reported in patients with aggressive periodontitis.66 A meta-analysis did not report differences in effectiveness based on the types of systemic antibiotics administered.66 Therefore, systemic antibiotics may be considered based on the clinical presentation and overall risk profile.
Subantimicrobial dose doxycline (SDD) is prescribed 20 mg twice daily and has been shown to be an effective adjunct in the reduction of PD and CAL gain over 3 months to 9 months.65,66 SDD’s mechanism of action involves the local downregulation of MMPs and the upregulation of tissue inhibitors of matrix metalloproteinases67,68 and may also demonstrate reductions in prostaglandin and pro-inflammatory cytokine levels.67,68 Patients’ overall health may impact the effectiveness of this treatment. A modest benefit has also been reported when systemic antibiotics are used as adjuncts to scaling and root planing in chronic periodontitis patients. Even more robust benefits are reported in patients with aggressive periodontitis.66 A meta-analysis did not report differences in effectiveness based on the types of systemic antibiotics administered.66 Therefore, systemic antibiotics may be considered based on the clinical presentation and overall risk profile.
Nonsteroidal anti-inflammatory drugs and bisphosphonate medications have demonstrated slowed progression of chronic periodontitis.69 Recent investigations have evaluated the use of low-dose aspirin (81 mg to 325 mg, once daily)70 accompanying polyunsaturated fatty acids.70,71 Polyunsaturated fatty acids have been shown to produce anti-inflammatory effects72 and may also improve CAL gains after nonsurgical periodontal therapy.73,74 The use of statin medications may improve periodontal health in conjunction with nonsurgical periodontal therapy.29 Such results may be more pronounced in patients who have elevated levels of inflammatory mediators due to systemic diseases,75–77 and indicate the need for individualized treatment planning based on each patient’s health status.
Repeated reevaluations of patients’ periodontal therapy and plaque control are essential to establishing and maintaining periodontal health. These reevaluations offer insight into the response to periodontal therapy and the patient’s overall prognosis. Such longitudinal information guides the need for additional interventions, including adjustments in patient’s self-care. Evaluations may occur perpetually, but are particularly useful 4 weeks to 6 weeks after completion of nonsurgical therapy, after surgical therapy if warranted, and at subsequent periodontal maintenance appointments, typically four times per year.78,79 Signs of progressive periodontal disease activity may include progressive attachment loss,80,81 presence of particular bacterial species,82,83 and conversion from nonbleeding to bleeding gingiva.84 While bleeding on probing itself is an inaccurate predictor of attachment loss,85 the absence of bleeding on probing is a reliable predictor of health, making achieving and maintaining nonbleeding sites important therapeutic waypoints.86 The consistent monitoring of periodontal disease activity and re-assessment of etiologic factors is critical to the long-term success of periodontal therapy.
PERIODONTAL MAINTENANCE
Because most periodontal diseases are chronic, following the completion of active periodontal therapy, a periodontal maintenance schedule should be established to maintain periodontal health. Studies have shown that tooth loss in periodontal patients is related to the frequency and quality of their maintenance care.87,88 Patients who are seen at regular intervals experience less attachment loss, lose fewer teeth, and have overall lower oral health care costs than those who are seen more infrequently.89 Intervals for periodontal maintenance should range between 2 months and 6 months depending on the patient’s disease presentation and history, risk factors for progression, treatment performed, new clinical findings, and the initial and changing prognoses for the teeth (based on periodic reevaluations). Shorter maintenance intervals may decrease the likelihood of subsequent disease progression.90 Furthermore, consistent reevaluations that include surveillance of probing depths, bleeding on probing, and subgingival scaling and root planing when indicated result in better maintenance of attachment levels and are more effective than supragingival plaque/calculus control alone.91 Additionally, periodontal maintenance visits enable clinicians to reassess patients’ medical conditions, plaque control, and disease activity while providing the opportunity to motivate and encourage them to be co-practitioners with dental professionals in achieving periodontal health.
CONCLUSION
Anti-infective therapies are the cornerstone of periodontal treatment and critical to the prevention of gingivitis and periodontitis. They are also key to arresting periodontitis and maintaining periodontal health. Anti-infective treatments may include: patient-delivered plaque control; mechanical debridement that may be nonsurgical, surgical, or both; adjunctive antimicrobial/anti-inflammatory therapies; and periodontal maintenance. Understanding the current scientific evidence while incorporating patient needs and health status with clinical expertise are critical to providing excellent patient care.
ACKNOWLEDGEMENT
The author would like to thank Elizabeth Bolton and Maninder Kaur, BDS, MPH, MS, for their assistance with this manuscript.
References
- Eke PI, Dye BA, Wei L, et al. Prevalence of periodontitis in adults in the United States: 2009 and 2010. J Dent Res. 2012;91:914–920.
- Löe H, Theilade E, Jensen SB. Experimental gingivitis in man. J Periodontol. 1965;36:177–187.
- Socransky SS, Haffajee AD, Cugini MA, Smith C, Kent RL Jr. Microbial complexes in subgingival plaque. J Clin Periodontol. 1998;25:134–144.
- Westfelt E, Rylander H, Dahlén G, Lindhe J. The effect of supragingival plaque control on the progression of advanced periodontal disease. J Clin Periodontol. 1998; 25:536–541.
- Bollen CML, Mongardini C, Papaioannou W, van Steenberghe D, Quirynen M. The effect of a one-stage full mouth disinfection on different intraoral niches. Clinical and microbiological observations. J Clin Periodontol. 1998;25:55–66.
- Drisko CH. Nonsurgical periodontal therapy. Periodontol 2000. 2001;25:77–88.
- Drisko CL. Periodontal self-care: evidence based support. Periodontol 2000. 2013;62:243–255.
- Straub AM, Salvi GE, Lang NP. Supragingival plaque formation in the human dentition. In: Lang NP, Attstrom R, Loe H, eds. Proceedings of the European Workshop on Mechanical Plaque Control. Chicago: Quintessence; 1998.
- American Dental Association: ADA Seal Shopping List. Available at: ada.org/ada/seal/shoppinglist.aspx. Accessed June 3, 2016.
- Slot DE, Dorfer CE, van der Weijden GA. The efficacy of interdental brushes on plaque and parameters of periodontal inflammation: a systematic review. Int J Dent Hyg. 2008;6:253–264.
- Berchier CE, Slot DE, Haps S, van der Weijden GA. The efficacy of dental floss in addition to a toothbrush on plaque and parameters of gingival inflammation: a systematic review. Int J Dent Hyg. 2008;6:265–279.
- Featherston JD. The science and practice of caries prevention. J Am Dent Assoc. 2000;131:887–899.
- Macgregor ID, Rugg-Gunn AJ. Toothbrushing duration in 60 uninstructed young adults. Community Dent Oral Epidemiol. 1985;13:121–122.
- Van der Weijden GA, Hioe KP. A systematic review of the effectiveness of self-performed mechanical plaque removal in adults with gingivitis using a manual toothbrush. J Clin Periodontol. 2005;32(Suppl 6): 214–228.
- Geisinger ML, Robinson M, Kaur M, et al. Individualized oral health education improves oral hygiene compliance and clinical outcomes in pregnancy women with gingivitis. Oral Hyg Health. 2013;1:111.
- Curry-Chiu ME, Catley D, Voelker MA, Bray KK. Dental hygienists’ experiences with motivational interviewing: a qualitative study. J Dent Educ. 2015;79:897–906.
- Miller WR, Rollnick S. Motivational Interviewing: Helping People Change. 3rd ed. New York: Gilford Publications; 2013.
- Wilder RS, Bray KS. Improving periodontal outcomes: merging clinical and behavioral science. Periodontol 2000. 2016;71:65–81.
- Stenman J, Lundgren J, Wennstrom JL, et al. A single session of motivational interviewing as an additive means to improve adherence in periodontal infection control: a randomized controlled trial. J Clin Periodontol. 2012;39:947–954.
- Almomani F, Williams K, Catley D, Brown C. Effects of an oral health promotion program on people with mental illness. J Dent Res. 2009;88:648–652.
- Paraskevas S, van der Weijden GA. A review of the effects of stannous fluoride on gingivitis. J Clin Periodontol. 2006;33:1–13.
- Food and Drug Administration. Oral Health Care Drug Products for Over-the-Counter Human Use; Antigingivitis/Antiplaque Drug Products; Establishment of a Monograph; Proposed Rules. Available at: fda.gov/downloads/Drugs/DevelopmentApprovalProcess/DevelopmentResources/Over-theCounterOTCDrugs/StatusofOTCRulemakings/ ucm096081.pdf. Accessed June 3, 2016.
- Kazmierczak M, Mather M, Ciancio S, et al. A clinical evaluation of anticalculus dentifrices. J Clin Prev Dent. 1990;12:13.
- Riley P, Lamont T. Triclosan/copolymer containing toothpastes for oral health. Cochrane Database Syst Rev. 2013;12:CD010514.
- US Centers for Disease Control and Prevention. National Biomonitoring Program. Factsheet: Triclosan. Available at:?cdc.gov/biomonitoring/Triclosan_FactSheet.html. Accessed June 3, 2016.
- United States Food and Drug Administration. Triclosan: What Consumers Should Know. Available at:?fda.gov/ForConsumers/ConsumerUpdates/ucm205999.htm. Accessed June 3, 2016.
- Jordan E, Hsu Y-T, Bashutski J. Do statin medications improve periodontal health and/or outcomes? A systematic review. Clin Adv Periodontol. 2014;4:194–202.
- Thunyakitpisal PD, Chaisuparat R. Simvastatin, a HMG-CoA reductase inhibitor, reduced the expression of matrix metalloproteinase-9 (Gelatinase B) in osteoblastic cells and HT1080 fibrosarcoma cells. J Pharmacol Sci. 2004;94:403–409.
- Pradeep AR, Karvekar S, Nagpal K, et al. Efficacy of locally delivered 1.2% rosuvastatin gel to non-surgical treatment of patients with chronic periodontitis: a randomized, placebo-controlled trial. J Periodontol. 2015;86:738–745.
- Fajardo ME, Rocha ML, Sanchez-Marin FL, Espinosa-Chavez EJ. Effect of atorvastatin on chronic periodontitis: A randomized pilot study. J Clin Periodontol. 2010;37:1016–1022.
- Pradeep AR, Rao NS, Bajaj P, Kumari M. Efficacy of subgingivally delivered simvastatin in the treatment of patients with type 2 diabetes and chronic periodontitis: A randomized double-masked controlled clinical trial. J Perioodontol. 2013;84:24–31.
- Ralston D, Carrasco R, Jacobsen PL, Wink C. Comparison of plaque removal capabilities between two dentifrices. Oral Hyg Health. 2014;2:157.
- Haps S, Slot DE, Berchier CE, van der Weijden GA. The effect of cetylpyridinium chloride-containing mouth rinses as adjuncts to toothbrushing on plaque parameters of gingival inflammation: a systematic review. Int J Dent Hyg. 2008;6:290–303.
- Stoeken JE, Paraskevas S, van der Weijden GA. The long-term effect of a mouthrinse containing essential oils on dental plaque and gingivitis: a systematic review. J Periodontol. 2007;78:1218–1228.
- Eley BM. Antibacterial agents in the control of supragingival plaque—a review. Br Dent J. 1999;186:286–296.
- Anauate-Netto C, Anido-Anido A, Leegoy HR, et al. Randomized, double-blind, placebo-controlled clinical trial on the effects of propolis and chlorhexidine mouthrinses on gingivitis. Braz Dent Sci. 2014;17:11–15.
- Ercan N, Erdemir EO, Ozkan SY, Hendek MK. The comparative effect of propolis in two different vehicles; mouthwash and chewing gum on plaque accumulation and gingival inflammation. Eur J Dent. 2015;9:272–276.
- Nadkerny PV, Ravishankar PL, Pramod V, Agarwal LA, Bhandari S. A comparative evaluation of the efficacy of probiotic and chlorhexidine mouthrinses on clinical inflammatory parameters of gingivitis: A randomized controlled study. J Indian Soc Periodontol. 2015;19:633–639.
- Pauk K, Zadrazilova I, Imramovsky A, et al. New derivatives of salicylamides: Preparation and antimicrobial activity against various bacterial species. Bioorg Med Chem. 2013;21:6574–6581.
- Noordin K, Kamin S. The effect of probiotic mouth rinse on plaque and gingival inflammation. Ann Dent Univ Malaya. 2007;14:19–25.
- Harini PM, Anegundi RT. Efficacy of a probiotic and chlorhexidine mouth rinses: A short-term clinical study. J Indian Soc Pedod Prev Dent. 2010; 28:179–182.
- Hallmon WW, Rees TD. Local anti-infective therapy: mechanical and physical approaches. A systematic review. Ann Periodontol. 2003;8:99–114.
- Cobb CM, Nonsurgical pocket therapy: mechanical. Ann Periodontol. 1996;1:443–490.
- Cugini MA, Haffajee AD, Smith C, Kent RL Jr. Socransky SS. The effect of scaling and root planing on the clinical and microbiological parameters of periodontal diseases: 12-month results. J Clin Periodontol. 2000;27:30–36.
- Stambaugh RV, Dragoo M, Smith DM, Carasali L. The limits of subgingival scaling. Int J Periodont Res Dent. 1981;1:30–41.
- Tunkel J, Heinecke A, Flemmig TF. A systematic review of efficacy of machine-driven and manual subgingival debridement in the treatment of chronic periodontitis. J Clin Periodontol. 2002;29(Suppl 3):72–81.
- Badersten A, Nilveus R, Egelberg J. Effect of nonsurgical periodontal therapy. I. Moderately advanced periodontitis. J Clin Periodontol. 1981;8:57–72.
- Badersten A, Nilveus R, Egelberg J. Effect of nonsurgical periodontal therapy. II. Severely advanced periodotntitis. J Clin Periodontol. 1984;11:63–76.
- Eberhard J, Jervoe-Storm PM, Needleman I, Worthington H, Jepsen S. Full-mouth treatment concepts for chronic periodontitis: a systematic review. J Clin Periodontol. 2008;35:591–604.
- Fang H, Han M, Li Q-L, et al. Comparison of full-mouth disinfection and quadrant-wise scaling in the treatment of adult chronic periodontitis: a systematic review and meta-analysis. J Periodont Res. October 19, 2015. Epub ahead of print.
- Nevins M, Kim SW, Camelo M, Martin IS, Kim D, Nevins M. A prospective 9-month human clinical evaluation of laser-assisted new attachment procedure (LANAP) therapy. Int J Periodont Res Dent. 2014;34:20–27.
- Nevins M, Camelo M, Schupbach P, Kim SW, Kim DM, Nevins M. Human clinical and histological evaluation of laser-assisted new attachment procedure. Int J Periodont Res Dent. 2012;32:496–507.
- Akram Z, Al-Shareef SAA, Daood U, et al. Bactericidal efficacy of photodynamic therapy against periodontal pathogens in periodontal disease: A systematic review. Photomed Laser Surg. 2016;34:137–149.
- Theodoro LH, Silva SP, Pires JR, et al. Clinical and microbiological effects of photodynamic therapy associated with nonsurgical periodontal treatment: a 6-month follow up. Lasers Med Sci. 2012;27:687–693.
- Stambaugh RV, Myers GC, Watanabe J, et al. Endoscopic instrumentation of the subgingival root surface in periodontal therapy. J Dent Res. 2000;79;489.
- Geisinger ML, Mealey BL, Schoolfield J, Mellonig JT. The effectiveness of subgingival scaling and root planing: An evaluation of therapy with and without the use of the periodontal endoscope. J Periodontol. 2007;78:22–28.
- Michaud RM, Schoolfield J, Mellonig JP, Mealey BL. The efficacy of subgingival calculus removal with endoscopy-aided scaling and root planing: a study on multirooted teeth. J Periodontol. 2007;78:2238–2245.
- Avradopoulso VA, Wilder RS, Chichester S, Offenbacher S. Clincial and inflammatory evlaution of perioscopy on patients with chronic periodontitis. J Dent Hyg. 2004;78:30–38.
- Smiley CJ, Tracy SL, Abt E, et al. Evidence-based clinical practice guideline on the nonsurgical treatment of chronic periodontitis by means of scaling and root planing with or without adjuncts. J Am Dent Assoc. 2015;146:525–535.
- Bonito AJ, Lux L, Lohr KN. Impact of local adjuncts to scaling and root planing in periodontal disease therapy: a systematic review. J Periodontol. 2005;76:1227–1236.
- Paquette D, Oringer R, Lessem J, et al. Locally delivered minocycline microspheres for the treatment of periodontitis in smokers. J Clin Periodontol. 2003;30:787–794.
- Hanes PJ, Purvis JP. Local anti-infective therapy: pharmacological agents. A systematic review. Ann Perioodontol. 2003;8:79–98.
- Breault LG, Spadaro SE. Local chemotherapeutics as an adjunct to scaling and root planing. Dent Assist. 2004;73:20–25.
- Carvalho J, Novak MJ, Mota LF. Evaluation of the effect of subgingival placement of chlorhexidine chip as an adjunct to scaling and root planing. J Periodontol. 2007;78:997–1001.
- Caton J, Ryan ME. Clinical studies on the management of periodontal diseases utilizing subantimicrobial dose doxycycline (SDD). Pharmacol Res. 2011;63:114–120.
- Haffajee AD, Socransky SS, Gunsolley JC. Systemic anti-infective periodontal therapy. A systematic review. Ann Periodontol. 2003;8:115–181.
- Golub LM, Wolff M, Lee HM, et al. Doxycycline inhibits neutrophil (PMN)-type matrix metalloproteinases in human adult periodontitis gingiva J Clin Periodontol. 1995;22:100–109.
- Golub LM, McNamara TF, Ryan ME, et al. Adjunctive treatment with subantimicrobial doses of doxycycline effects on gingival fluid collagenase activity and attachment loss in adult periodontitis. J Clin Periodontol. 2001;28:146–156.
- Reddy MS, Geurs NC, Gunsolley JC. Periodontal host modulation with antiproteinase, anti-inflammatory, and bone-sparing agents. A systematic review. Ann Periodontol. 2003;8:12–37.
- Flemmig TF, Rumetsch M, Klaiber B. Efficacy of systemically administered acetylsalicylic acid plus scaling on periodontal health and elastase-alpha-1-proteinase inhibitor in gingival crevicular fluid. J Clin Periodontol. 1996;23:153–159.
- Geisinger ML, Holmes CM, Vassilopoulos PJ, et al. Adjunctive treatment of chronic periodontitis with systemic low-dose aspirin therapy. Clinical Advances in Periodontics. 2012;2(3):195–199.
- Martinez GL, Koury JC, Martins MA, et al. Serum changes of long chain-polyunsaturated fatty acids in patients undergoing periodontal therapy combined with one year of omega-3 supplementation: a pliot randomized controlled trial. J Perioodontal Implant Sci. 2014;44:169–177.
- Rosenstein ED, Kushner LJ, Kramer N, Kazandjian G. Pilot study of dietary fatty acid supplementation in the treatment of adult periodontitis. Prostaglandins Leukot Essent Fatty Acids. 2003;68:213–218.
- Naqvi AZ, Hasturk H, Mu L, et al. Docosahexaenoic Acid and Periodontitis in Adults: A randomized controlled trial. J Dent Res. 2014;93:767–773.
- Simpson TC, Weldon JC, Worthington HV, et al. Treatment of periodontal disease for glycaemic control in people with diabetes mellitus. Cochrane Database Syst Rev. 2015;6:11:CD004714
- Chistiakov DA, Orekhov AN, Bobryshev YV. Links between atherosclerotic and periodontal disease. Exp Mol Pathol. 2016;100:220–235.
- Gregor MF, Hotamisligil GS. Inflammatory mechanisms in obesity. Annu Rev Immuol. 2011;29:415–445.
- Greenstein G and Caton J. Periodontal Disease Activity: A critical assessment. J Periodontol. 1990;61:543–552.
- Claffey N. Decision making in periodontal therapy—the reevaluation. J Clin Periodontol. 1991;18:384–389.
- Jeffcoat MK, Reddy MS. Progression of probing attachment loss in adult periodontitis. J Periodontol. 1991;62:185–189.
- Lindhe J, Okamoto H, Yoneyama T, Haffajee A, Socransky SS. Periodontal loser sites in untreated adult subjects. J Clin Periodontol. 1989;16:671–678.
- Armitage G, Dickinson WR, Jenderseck RS, Levine SM, Chambers DW. Relationship between the percentage of subgingival spirochetes and the severity of periodontal disease. J Periodontol. 1982;53:550–556.
- Slots J, Emrich LJ, Genco RJ, Rosling BG. Relationship between some subgingival bacteria and periodontal pocket depth and gain or loss of periodontal attachment after treatment of adult periodontitis. J Clin Periodontol. 1985;12:5540–552.
- Caton J, Thilo B, Polson A, Espeland M. Cell populations associated with conversion from bleeding to nonbleeding gingiva. J Periodontol. 1988;59:7–11.
- Lang NP, Joss A, Orsanic T, Gusberti FA, Siegrist BE. Bleeding on probing. A predictor for the progression of periodontal disease? J Clin Periodontol. 1986;13:590–596.
- Lang NP, Adler R, Joss A, Nyman S. Absence of bleeding on probing. An indicator of periodontal stability. J Clin Periodontol. 1990;17:714–721.
- Axelsson P, Nystrom B, Lindhe J. The long-term effect of a plaque control program on tooth mortality, caries, and periodontal disease in adults. Results after 30 years of maintenance. J Clin Periodontol. 2004;31:749–757.
- Wislon TG, Glover ME, Malik AK, Schoen JA, Dorsett D. Tooth loss in maintenance patients in a private practice. J Periodontol. 1987;58:231–235.
- Gaunt F, Devine M, Pennington M, et al. The cost-effectiveness of supportive periodontal care for patients with chronic periodontitis. J Clin Periodontol. 2008;35:67–82.
- Cohen RE; Research, Science, and Therapy Committee, American Academy of Periodontology. Position paper: periodontal maintenance.
- Van der Weijden GA, Timmerman MF. A systematic review on theclinical efficacy of subgingival debridement in the treatment of chronicperiodontitis. J Clin Periodontol. 2002;29(Suppl 3):55–71.
From Dimensions of Dental Hygiene. July 2016;14(07):53–58.



