 POVARESHKA/ISTOCK/GETTY IMAGES PLUS
POVARESHKA/ISTOCK/GETTY IMAGES PLUS
Improving Oral Health With Polyphenols
These antioxidants provide anti-inflammatory, anticariogenic, and anticarcinogenic effects.
This course was published in the May 2018 issue and expires May 30, 2021. The authors have no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Describe polyphenols and their therapeutic properties that aid disease prevention.
- Explain the actions by which polyphenols promote oral health, and some of the conditions for which they appear clinically useful.
- Discuss the extent to which common oral diseases affect the general population.
- List common pathogenic oral microbes, including species that polyphenols may help inhibit.
Oral health is paramount for patients’ overall well-being and quality of life. Multiple therapies are available to prevent oral disease, yet caries, periodontal conditions, oral cancer, and oral infectious diseases remain prevalent worldwide.1 For some patients, natural remedies are a main source of health care treatment.1,2 Current evidence supports the role that nutrition plays in combating diseases, specifically through the anti-inflammatory, anticariogenic, and anticarcinogenic actions of specific antioxidants known as polyphenols.3–6 A multitude of in vitro and in vivo studies have been conducted to ascertain how polyphenols aid in reducing oral disease.
Polyphenols belong to a class of plant-based chemical compounds known as phytochemicals. The generic term “polyphenol” encompasses more than 8,000 types of phenolic structures, the most common being phenolic acids, flavonoids, lignans, and stilbenes.7 These phytochemicals possess important therapeutic antioxidant properties that may promote oral health by reducing the risk of inflammation and infections, and lowering the risk of tumor production and oxidative stress (the body’s inability to remove free radicals).7–9 Polyphenols are found in many dietary sources, including a wide array of herbs, spices, fruits, vegetables, whole grains, cereals, nuts, seeds, dry legumes, honey, olive oils, chocolate and soy, as well as plant-based beverages, such as teas, coffee, wine (mainly red), and beer (Table 1, page 52).4,7,9–11
CARIES RISK REDUCTION
The World Health Organization reports that nearly 100% of adults globally present with caries, as well as 60% to 90% of school-age children.1 Streptococcus mutans plays a major role in the etiology of caries.12 Therefore, a reduction in S. mutans levels may help lower the risk for caries. Multiple studies support the use of polyphenols as a possible caries risk reduction intervention.13–16
A 2011 clinical study involving subjects who chewed laboratory-created gum enriched with the polyphenol quercetin (a flavanoid contained in fruits, vegetables, and beverages) found that quercetin was effectively released into saliva within minutes, and by day 7, a marked reduction in overall levels of S. mutans was evident.13 In one clinical study, S. mutans was significantly reduced after subjects rinsed daily for 6 weeks with mouthrinse supplemented with cranberry extract.14
An in vitro study using polyphenols contained in hops demonstrated reduced levels of lactic acid production by S. mutans, while bakuchiol (similar to the polyphenol resveratrol found in red wine) and macelignan polyphenols (found in nutmeg) displayed anti-biofilm activity by inhibiting growth of S. mutans, S. sanguinis, and Actinomyces viscosus.15 Additional in vitro studies demonstrated that certain polyphenolic components found in cranberries prevented the production of organic acids by cariogenic bacteria, restricted the biofilm formation of S. mutans and S. sobrinus, and inhibited the attachment and coaggregation of numerous additional Streptococcus oral species, including S. mitis, S. criceti, and S. oralis.15 In a laboratory study investigating orange peel extract as a method of caries reduction, researchers noted inhibited microbial growth of S. mutans, suggesting the need for additional research to ascertain the effects of these polyphenols when incorporated into mouthrinses and dentifrices.16
PERIODONTAL DISEASES
According to the United States Centers for Disease Control and Prevention, almost half of Americans age 30 and older have some type of periodontal disease—that’s approximately 65 million people. More than 70% of individuals age 65 and older are affected.17Several key organisms in periodontal destruction are the red complex bacteria; these subgingival colonizers consist of Porphyromonas gingivalis, Treponema denticola, and Tannerella forsythia.18 Numerous studies support polyphenol’s role in the prevention of periodontal disease.18–23
In an in vitro study, Bodet el al18 reported strong evidence that cranberry polyphenols reduced the inflammatory actions of all three red complex species by partially deactivating their inflammatory enzymes, thus potentially preventing degradation of the collagen in the periodontal sulcus. Catechins (class of flavonoids found in green tea) demonstrated the same inhibitory effect in P. gingivalis.19Resveratrol (stilbene found in red wine and red-skinned grapes) proved to markedly inhibit in vitro growth of Aggregatibacter actinomycetemcomitans and P. gingivalis within 24 hours and reduce key pro-inflammatory markers produced by both species.7,20,21Notably, black tea extract potentiated a highly susceptible antibacterial effect against P. gingivalis and significantly reduced inflammatory factors, preventing gingival cellular damage.22
In 2015, Priya et al23 conducted a clinical study using human subjects to compare the efficacy of mouthrinse containing green tea to that of chlorhexidine gluconate (CHX) in managing plaque-induced gingivitis. Over 30 days, one group rinsed with 5% green tea mouthrinse, and the other rinsed with 0.12% CHX. Both groups demonstrated a reduction in plaque and bleeding scores on day 15 and day 30; however, those who used green tea rinse exhibited a significantly marked reduction in bleeding scores on day 30. The researchers concluded that green tea mouthrinse was just as effective in inhibiting biofilm aggregation—and was even more effective at reducing gingival bleeding—than CHX. No significant changes in staining were reported in either group.
ORAL CANCER
According to the National Cancer Institute, approximately 346,902 individuals in the United States are living with oral cancer, a condition that accounts for 2.9% of all new cancer cases.24 A variety of polyphenolic oral cancer studies involving dietary agents have been conducted to ascertain their chemopreventive roles.25–30
During an in vitro experiment, Ghashm et al25 demonstrated that polyphenols found in Malaysian honey were successful at treating cell lines with oral squamous cell carcinoma. Varying concentrations (2% to 10%) of Malaysian honey were directly applied to the malignant cells and microscopically examined over a 24-, 48- and 72-hour period. Results showed that programmed cell death was induced at all three time intervals; the researchers also noted a marked reduction in the number of cancerous cells.
Other studies concluded that many polyphenols found in green and black tea aided in reversing, suppressing, and preventing premalignant cells from transitioning into malignant cells.26,27 Letchoumy et al26 studied the effects of black tea in hamsters infected with oral squamous cell carcinoma. They found that after 18 weeks of ingesting prepared straight black tea (equivalent to 30 mg to 40 mg of tea polyphenols per kg body weight in humans), there was a reduction in the incidence of carcinomas and preneoplastic lesions. This was possibly due to interference with cancer signaling pathways.
Wang et al27 examined whether green tea and black tea had an effect on esophageal cancer growth in rats. Subjects drank a solution of tea infused in water; the concentrations were 0.6% decaffeinated green tea, 0.6% decaffeinated black tea, 0.9% regular green tea, or 0.9% decaffeinated green tea. All rats consumed only one tea, which was their sole source of liquid for 5 weeks. Upon conclusion, all rats showed a > 50% reduction in tumor growth.
A human study by Schwartz et al28 reported that heavy smokers who consumed five cups of green tea per day (400 mg to 500 mg green tea extract per cup) for 4 weeks showed a decrease in mucosal DNA damage. In addition, randomized, placebo-controlled trials in humans presenting with precancerous oral lesions demonstrated that green tea extract has a protective role against the progression of premalignant lesions in the oral cavity.29,30
ORAL MALODOR
Oral malodor is a common complaint of patients, and is initiated chiefly by volatile sulfur compounds (VSCs) prevalent in the oral cavity. The particular VSCs are methyl mercaptan and hydrogen sulfide, which are produced by certain strains of bacteria, specifically S. moorei.31,32 According to Aylıkcı et al,31 more than 50% of the general population presents with oral malodor. Although it can originate from oral and nonoral sources, approximately 90% of cases are associated directly with the oral cavity. These are predominantly attributed to poor self care, periodontal disease/deep periodontal pockets, biofilm retention on the tongue, food impaction or material alba, unclean dentures, faulty/failing restorations, deep caries lesions, oral cancers, and/or throat infections.31 A study by Morin et al32 demonstrated that green tea extract exhibited antibacterial activity against S. moorei. Additionally, studies that incorporated green tea polyphenols into mouth spray and chewing gum demonstrated irreversible damage to the bacterial membrane and successful removal of the VSCs responsible for oral malodor.32,33
ORAL CANDIDIASIS
Candidiasis is a fungal infection caused by Candida yeasts, with the most common being Candida albicans. There are more than 20 species that may cause infection in humans.34 Although such yeasts are part of the normal microflora of the skin, mucous membranes and intestinal tract, infection may arise when overgrowth occurs, especially for immunocompromised patients—such as those undergoing chemotherapy, individuals afflicted with the human immunodeficiency virus, or immunosuppressed organ transplant patients.34,35
Shahzad et al36 explored the antifungal potential of 14 polyphenols for inhibition and adhesion properties of C. albicans. This study used reference strains and isolates collected from the oral cavity, dentures and the bloodstream. Seven of the examined polyphenols (curcumin, pyrocatechol, pyrogallol, quercetin, gallic acid, caffeic acid, and naringenin) proved effective against growth and exhibited dose-dependability. Curcumin (found mainly in curry powder and turmeric) and pyrogallol (common in cocoa powder, coffee and beer) were the most effective polyphenols in reducing biofilm. The team’s results showed that both curcumin and pyrogallol significantly suppressed genes that coded for adhesion molecules in biofilm development by 50% to 70%.
Evenson and Braun37 determined that green tea extract caused metabolic instability in the cell walls of C. albicans, and also retarded and inhibited its growth. Results were dose-dependent and, as green tea extract concentrations increased (a maximum of 5 mg/ml experimental concentration), fungal growth rate patterns steadily declined by as much as 43% within 24 hours. Furthermore, it disrupted established biofilm communities.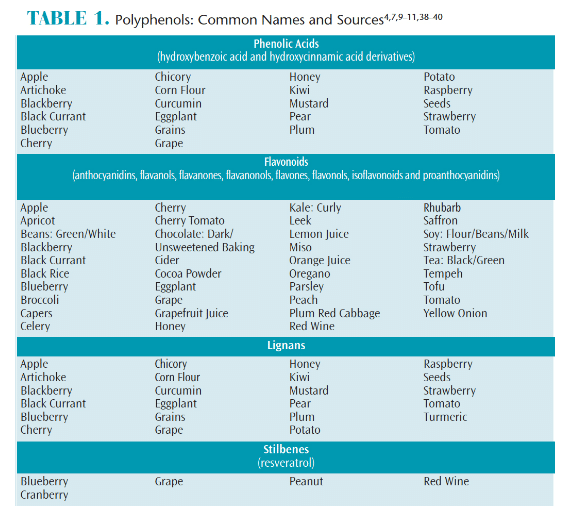
CONCLUSION
Although numerous in vitro and in vivo research studies demonstrate that various polyphenols have beneficial therapeutic effects on oral health, many investigations are still in preliminary phases. Although polyphenols are found in a multitude of dietary sources, it is recommended that all foods be eaten in moderation, and no single food be eaten in high quantities. Until further studies are conducted, no more than two cups of caffeinated green tea should be consumed per day.3 Pregnant or nursing women, and individuals with systemic conditions, should consult with their physician prior to incorporating any supplements into their diet.3
In order to truly comprehend the role of polyphenols and oral health, additional human studies are needed. Moreover, the inclusion of these compounds in toothpaste, mouthrinses, mouth sprays, chewing gums—and possibly oral and systemic medications—may prove helpful for managing an array of oral conditions in the future.
REFERENCES
- World Health Organization. Oral health Information Sheet April 2012. Available at: who.int/oral_health/publications/factsheet/en/. Accessed February 8, 2018.
- World Health Organization. WHO traditional medicine strategy: 2014–2023. Geneva, Switzerland: WHO Press; 2013.
- Singhal K, Raj N, Gupta K, Singh S. Probable benefits of green tea with genetic implications. J Oral Maxillofac Pathol. 2017;21:107–114.
- Varela-López A, Bullón P, Giampieri F, Quiles JL. Non-Nutrient, naturally occurring phenolic compounds with antioxidant activity for the prevention and treatment of periodontal diseases. Antioxidants (Basel). 2015;4:447–481.
- Iriti M, Varoni EM. Chemopreventive potential of flavonoids in oral squamous cell carcinoma in human studies. Nutrients.2013;5:2564–2576.
- Yoo S, Murato RM, Duarte S. Antimicrobial traits of tea- and cranberry-derived polyphenols against Streptococcus mutans. Caries Res. 2011;45:327–335.
- Ding Y, Yao H, Yao Y, Fai LY, Zhang Z. Protection of dietary polyphenols against oral cancer. Nutrients. 2013;5:2173–2191.
- Liu RH. Health-promoting components of fruits and vegetables in the diet. Adv Nutr. 2013;4:384S–392S.
- Scalbert A, Johnson IT, Saltmarsh M. Polyphenols: antioxidants and beyond. Am J Clin Nutr. 2005,81(suppl 1):215S–217S.
- Kumar S, Pandey AK. Chemistry and biological activities of flavonoids: an overview. Sci World J. 2013;2013:167250.
- Samarghandian S, Farkhondeh T, Samini F. Honey and health: a review of recent clinical research. Pharmacognosy Res. 2017;9:121–127.
- Darby M, Walsh M. Dental Hygiene Theory and Practice. 2nd ed. St. Louis, Mo: Saunders;2003:238.
- Ferrazzano GF, Cantile T, Coda M, et al. In vivo release kinetics and antibacterial activity of novel polyphenols-enriched chewing gums. Molecules. 2016;21:1008.
- Ferrazzano GF, Amato I, Ingenito A, Zarrelli A, Pinto G, Pollio A. Plant polyphenols and their anti-cariogenic properties: a review. Molecules. 2011;16:1486–1507.
- Bonifait L, Grenier D. Cranberry polyphenols: potential benefits for dental caries and periodontal disease. J Can Dent Assoc. 2010;76:a130.
- Shetty SB, Mahin-Syed-Ismail P, Varghese S, et al. Antimicrobial effects of Citrus sinensis peel extracts against dental caries bacteria: an in vitro study. J Clin Exp Dent. 2016;8:e71–e77.
- U.S. Centers for Disease Control and Prevention. Periodontal disease. Available at: cdc.gov/oralhealth/ periodontal_disease/. Accessed February 8, 2018.
- Bodet C, Piché M, Chandad F, Grenier D. Inhibition of periodontopathogen-derived proteolytic enzymes by a high-molecular-weight fraction isolated from cranberry. J Antimicrob Chemother. 2006;57:685–690.
- Okamoto M, Sugimoto A, Leung KP, Nakayama K, Kamaguchi A, Maeda N. Inhibitory effect of green tea catechins on cysteine proteinases in Porphyromonas gingivalis. Oral Microbiol Immunol. 2004;19:118–120.
- O’Connor DJ, Wong RW, Rabie AB. Resveratrol inhibits periodontal pathogens in vitro. Phytother Res. 2011;25:1727–1731.
- Núñez MJ, Novío S, Balboa J, Seoane J, Suárez JA, Freire-Garabal M. Effects of resveratrol on expression of vascular endothelial growth factor in human gingival fibroblasts stimulated by periodontal pathogens. Acta Odontologica Scandinavica. 2010;68:239–247.
- Bedran TBL, Morin M-P, Palomari Spolidorio D, Grenier D. Black tea extract and its theaflavin derivatives inhibit the growth of periodontopathogens and modulate interleukin-8 and β-defensin secretion in oral epithelial cells. PLoS One. 2015;10:e0143158.
- Priya PB, Anitha V, Shanmugam M, Ashwath B, Sylva SD, Vigneshwari SK. Efficacy of chlorhexidine and green tea mouthwashes in the management of dental plaque-induced gingivitis: a comparative clinical study. Contemp Clin Dent.2015;6:505–509.
- National Cancer Institute. Cancer stat facts: Oral cavity and pharynx cancer. Available at: https://seer.cancer.gov/statfacts/html/oralcav.html. Accessed February 9, 2018.
- Ghashm AA, Othman NH, Khattak MN, Ismail NM, Saini R. Antiproliferative effect of Tualang honey on oral squamous cell carcinoma and osteosarcoma cell lines. BMC Complement Altern Med. 2010;10:49.
- Letchoumy PV, Mohan KV, Prathiba D, Hara Y, Nagini S. Comparative evaluation of antiproliferative, antiangiogenic and apoptosis inducing potential of black tea polyphenols in the hamster buccal pouch carcinogenesis model. J Carcinog. 2007;6:19.
- Wang ZY, Wang LD, Lee MJ, et al. Inhibition of N-nitrosomethylbenzylamine-induced esophageal tumorigenesis in rats by green and black tea. Carcinogenesis.1995;16:2143–2148.
- Schwartz JL, Baker V, Larios E, Chung FL. Molecular and cellular effects of green tea on oral cells of smokers: a pilot study. Mol Nutr Food Res. 2005; 49:43–51.
- Li N, Sun Z, Han C, Chen J. The chemopreventive effects of tea on human oral precancerous mucosa lesions. Proc Soc Exp.Biol Med. 1999;220:218–224.
- Tsao AS, Liu D, Martin J, et al. Phase II randomized, placebo-controlled trial of green tea extract in patients with high-risk oral premalignant lesions. Cancer Prev Res (Phila). 2009;2:931–941.
- Aylıkcı BU, Çolak H. Halitosis: from diagnosis to management. J Nat Sci Biol Med. 2013;4:14–23.
- Morin MP, Bedran TB, Fournier-Larente J, Haas B, Azelmat J, Grenier D. Green tea extract and its major constituent epigallocatechin-3-gallate inhibit growth and halitosis-related properties of Solobacterium moorei. BMC Complement Altern Med. 2015;15:48.
- Narotzki B, Reznick AZ, Aizenbud D, Levy Y. Green tea: a promising natural product in oral health. Arch Oral Biol. 2012;57:429–435.
- U.S. Centers for Disease Control and Prevention. Candidiasis. Available at: cdc.gov/fungal/diseases/candidiasis/index.html. Accessed February 9, 2018.
- Wang Y, Bandara HM, Mikkelsen D, Samaranayake L. Effects of tea extracts on the colonization behaviour of Candida species: attachment inhibition and biofilm enhancement. J Med Microbiol. 2017;66:1244–1252.
- Shahzad M, Sherry L, Rajendran R, Edwards CA, Combet E, Ramage G. Utilising polyphenols for the clinical management of Candida albicans biofilms. Int J Antimicrob Agents. 2014;44:269–273.
- Evensen NA, Braun PC. The effects of tea polyphenols on Candida albicans: inhibition of biofilm formation and proteasome inactivation. Can J Microbiol. 2009;55:1033–1039.
- Milder IE, Arts IC, van de Putte B, Venema DP, Hollman PC. Lignan contents of Dutch plant foods: a database including lariciresinol, pinoresinol, secoisolariciresinol and matairesinol. Br J Nutr. 2005;93:393–402.
- Meagher LP, Beecher GR. Assessment of data on the lignan content of foods. J Food Comp Anal. 2000;13:935–947.
- Khalil MI, Sulaiman SA. The potential role of honey and its polyphenols in preventing heart diseases: a review. Afr J Tradit Complement Altern Med. 2010;7:315–321.
From Dimensions of Dental Hygiene. May 2018;16(5):50-53.




This information is amazing! I love having more different natural options to offer my patients for those opposed to the approach of the use of medicine. It directly correlates to our diets and may be more encouraging to patients to work on there diets to help improve oral health.