 DANIIL DUBOV / ISTOCK / GETTY IMAGES PLUS
DANIIL DUBOV / ISTOCK / GETTY IMAGES PLUS
Implementing Buffered Articaine in Dentistry
The use of buffered articaine offers several advantages in dental pain management.
This course was published in the July 2022 issue and expires July 2025. The authors have no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Describe the properties and pharmacokinetics that make articaine a viable local anesthetic in dental settings.
- Explain the concept and benefits of using buffered local anesthetics—and buffered articaine, in particular.
- Describe clinical modalities and best practices when using buffered anesthetic agents.
Comfort is a cornerstone of compassionate care, and, toward this goal, proactive drug selection is a key factor in providing profound local anesthesia (LA) in the dental setting. Advances in the pharmacology of LA drugs—along with adjunctive technologies, such as buffering—enhance the efficacy of these agents.
As the first hybrid amino amide LA with both ester and amide properties, articaine’s pharmacology addresses dentistry’s requirement for profound and relatively safe anesthesia. According to systematic reviews, 4% articaine demonstrates a greater ability to provide profound anesthesia in a majority of situations. A 2020 review on the effectiveness of LA drugs in the presence of pulpitis, for example, demonstrated that of the five LA drugs available in cartridge form, 4% articaine provided superior efficacy. In fact, it was reported to be ~1.3 to six times more effective.1 When compared to 2% lidocaine, 4% articaine was more than six times more effective in pulpitis, and it diffused to both palatal and lingual tissues in significantly higher concentrations following buccal infiltration—frequently high enough to preclude the need for separate palatal and lingual injections.1–3
Articaine’s unique pharmacology includes a thiophene ring that increases liposolubility, which facilitates diffusion through nerve membranes, resulting in enhanced potency and overall effectiveness (see sidebar on page 40).4–6 Efficacy is also attributed to articaine’s 94% protein binding rate, which is similar to bupivacaine’s (96%).7–9 This is important because stronger protein binding enhances anesthetic duration.9 Table 1 presents protein binding information for dental LA cartridges.8,10
Yet despite articaine’s advantages in various clinical situations, challenges remain. For example, while 4% articaine was found to have the highest success rate in pulpitis after administration of an inferior alveolar nerve block, this agent’s 27% failure rate means three out of 10 attempts fail to provide profound anesthesia.11 In addition, like inferior alveolar blocks in general, nonsupplemental buccal infiltrations of the mandible offer similar success rates.4
Fortunately, more successful anesthesia outcomes are not only possible, but fairly easy to achieve. Recent research, for example, presents strong evidence in favor of buffering LA drugs.12,13 Current adjunctive technologies make buffering relatively simple. By increasing the pH of LA solutions via buffering prior to administration, multiple advantages can be realized with each of the LA agents. These include decreased pain and onset time, as well as enhanced efficacy.12,13 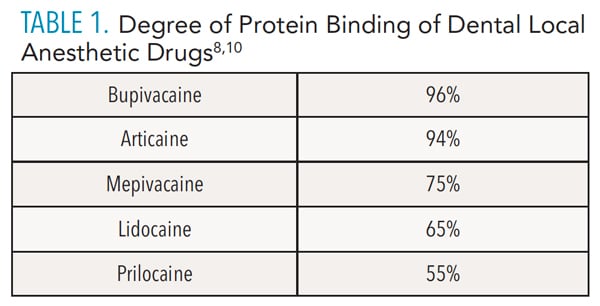
Fundamentals of Buffering
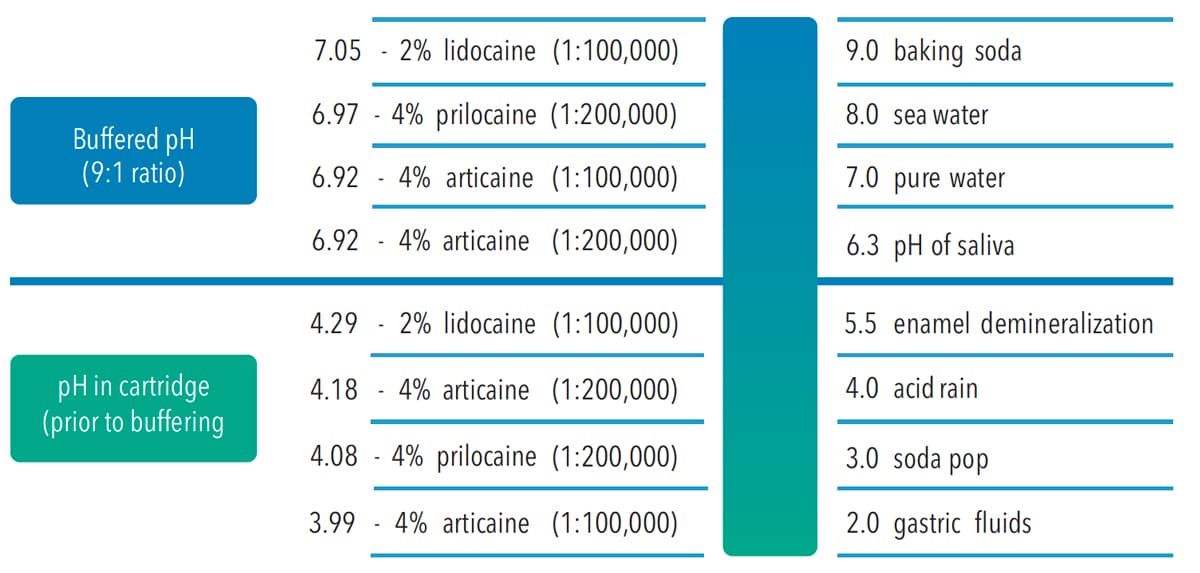
Manufacturers acidify LA drug solutions to enhance solubility and increase stability during sterilization and storage.4 In addition to varying degrees of pH adjustment during the manufacturing process, solutions with vasoconstrictors are more acidic than nonsupplemented LA solutions because they include sulfite antioxidants to improve vasoconstrictor shelf life—which also decreases pH levels. Throughout a vasoconstrictor-formulated LA drug’s useful life span, these preservatives continue to lower pH levels. Figure 1 and Table 2 present pH and buffering information for LA drugs.14
Low pH levels increase the percentage of charged molecules that are initially deposited into tissues surrounding nerves, which can delay onset and increase burning and stinging sensations.4 Carbonate buffering in human tissues quickly increases the pH of LA drugs after injection, offsetting initially low levels. An estimate of the pH of 2% lidocaine cartridges in clinical practice ranges from 2.9 to 4.4.15–19 At this pH range, the percentage of effective nonpolar molecules is significantly less than it is at a more typical extracellular tissue pH of 7.2 to 7.4.
On the other hand, for a drug with an initially low pH and a dissociation constant (pKa) of 7.2, if the pH is increased to 7.2, there is a predicted equal distribution of charged and uncharged LA molecules in solution.4 The percentage of charged molecules would decrease to 50% after buffering to 7.2, while the percentage of nonpolar molecules would increase to 50% in this example.4
Buffering prior to injection alters the balance of the two forms of LA molecules in solution, cations and neutral base or nonpolar molecules by increasing the nerve membrane-penetrating nonpolar concentration. In addition, it has been confirmed that CO2—also a nonpolar molecule that easily passes through nerve cell membranes—is generated when LA agents are buffered with 8.4% sodium bicarbonate.13 Catchlove20 describes three mechanisms by which CO2 can enhance LA efficacy: via a direct depressant effect; by decreasing axoplasmic pH; and by concentrating charged LA molecules inside the axoplasm, referred to as ion trapping.19,20
Additionally, CO2 is known to provide independent anesthetic activity. And when combined with water, it forms carbonic acid in the axoplasm, thus reducing pH levels.13,20 Lower axoplasmic pH increases the percentage of charged molecules within the axoplasm by converting uncharged molecules that enter the membrane into charged molecules.13,20 Outgoing movement is restricted, concentrating cations in the axoplasm.13,20 Since cationic LA molecules close or gate sodium ion channels (inhibiting impulse generation), these rapid actions of CO2 have been suggested as an explanation for buffering’s enhanced anesthetic efficacy.13,20
Considerations When Discussing Lidocaine and Articaine
While systematic reviews demonstrate that 4% articaine with epinephrine is more potent than 2% lidocaine with epinephrine, milligram to milligram, lidocaine is more potent than articaine.1–3,5 The approved concentrations of lidocaine and articaine solutions with epinephrine balance effectiveness with risk. In one study, 5% lidocaine with epinephrine yielded a 93% effectiveness in pulpitis.6 This contrasts with a recent systematic review that demonstrated an effectiveness of only 12% for 2% lidocaine with epinephrine in pulpitis vs 73% for 4% articaine with epinephrine.1
When considering these results, it is important to note that concentrations of lidocaine greater than 2%—despite the inclusion of epinephrine to slow systemic uptake—significantly increase toxic potential compared to 4% solutions of articaine. These higher concentrations of lidocaine are not available in dental cartridges.
Clinical Benefits
Although research has confirmed the benefits of buffering with sodium bicarbonate, not all are in consensus. A 2019 systematic review reported a 2.29x greater likelihood of achieving successful maxillary and mandibular anesthesia using buffered anesthetics rather than their nonbuffered counterparts.19 However, in a systematic review examining comfort and onset with buffered anesthetics, Auliesta-Viera et al21 recommended against buffering. Based on their review, routine alkalinization resulted in significantly faster onset in inferior alveolar blocks and in the presence of inflammation, yet they concluded the time it took to buffer (mixing times averaged ~1.3 minutes) negated any improvements in onset.
This may be less relevant when using modern chairside buffering methods that are much faster than earlier technologies.18,21 Using current buffering devices—where buffered LA is available in a matter of seconds—the reduction in onset times of inferior alveolar blocks and inflamed tissue anesthesia appears to be clinically significant.21–23
Analysis of the results of buffered injections suggests improvements in pain scores when injecting buffered anesthetics into skin and other nonmucosal tissues do not necessarily apply to intraoral injections.21 The premise is that much of the oral mucosa is easily penetrated compared to skin; therefore, reports of significant pain reduction using buffered LAs in dermatology, for example, may not apply to dentistry. However, other studies comparing nonbuffered to buffered intraoral LA techniques have noted significant improvements in comfort using buffered solutions.13,22,23
Compared to conventional LA solutions, reduced volumes of buffered LA have been recommended when performing infiltrations and nerve blocks.13,23 If one cartridge of a particular nonbuffered anesthetic is typically administered for inferior alveolar blocks, one-half to two-thirds may be sufficient with buffered delivery. Another consideration is that nonbuffered solutions are diluted when sodium bicarbonate buffering agents are added. For example, a recommended dilution of 2% lidocaine, 1:100,000 epinephrine with 8.4% sodium bicarbonate results in a concentration of 1.8% lidocaine.13,14,18 The epinephrine concentration decreases proportionately, as well.13,14,18 Compared to traditional LA delivery, the resulting injection volumes and concentration of buffered anesthetic enhance safety.
It should also be noted that buffered solutions, even if only marginally less concentrated, provide enhanced benefits. Evidence confirms that diluted, buffered anesthetics have faster onsets and are more effective than their nonbuffered, more concentrated counterparts.12,18
Enhanced time management is an indirect benefit of buffering. Anesthesia develops rapidly, which reduces wait time before initiating treatment—and if adequate anesthesia fails to develop in a relatively short period, additional anesthetic can be administered sooner. According to Malamed et al,23 it takes 1:51 minutes on average for pulpal analgesia to develop with alkalinized anesthetic, compared to 6:37 minutes for nonalkalinized agents. It was further reported that 71% of study participants achieved pulpal analgesia in 2 minutes or less with buffered anesthetic, whereas only 12% achieved pulpal analgesia in that time frame when nonbuffered anesthetic was used.23
Delivery Methods
Historically, it has been relatively easy to buffer LA drugs in medicine by drawing from separate vials and mixing drug solutions with buffering agents (ie, hand mixing). This method can also be used in dentistry and, when appropriately performed, is a viable alternative—although it has not been widely adopted.18 Two automated options are also available for buffering LA agents.16,17
One system extracts a precise volume of LA cartridge solution, and transfers the same volume of sodium bicarbonate solution into the cartridge immediately before delivery. The other system mixes buffering solution with LA from separate vials, after which the mixture is drawn into a proprietary aspirating syringe for immediate use. As with direct manipulation of the solutions, mixing just prior to delivery is recommended to optimize the enhanced potency and rapid anesthetic effect produced by CO2.
Future technologies are aimed at providing prepackaged, prebuffered cartridges. Bifurcated cartridges that would work with all current cartridge-loading syringes—for example, with separate storage chambers for anesthetic and buffering solution—are possible. These or similarly creative approaches will hopefully be introduced in the near future. In addition, the removal of sodium chloride from cartridges could further increase patient comfort.13 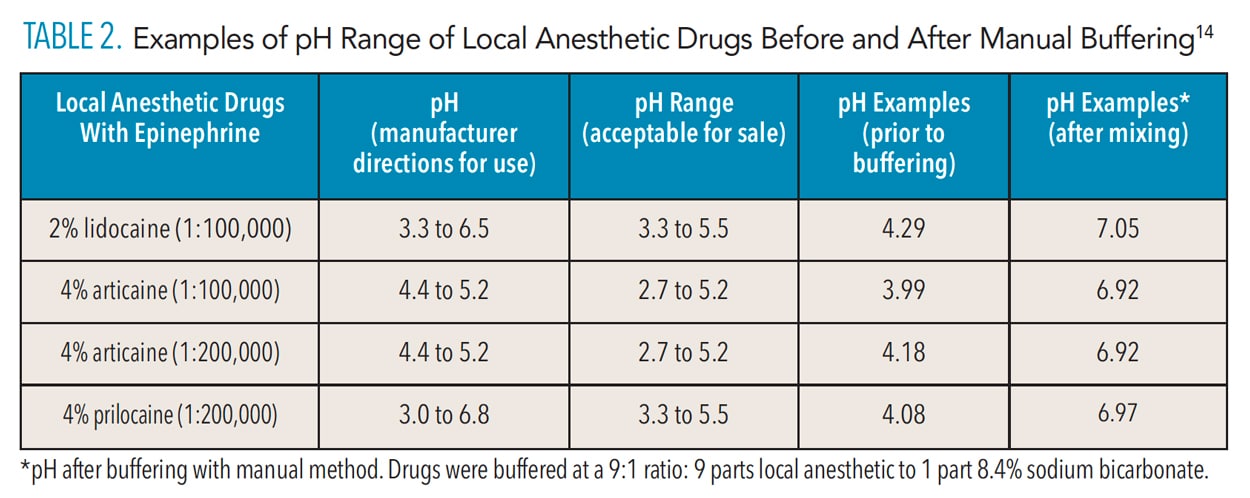
Additional Considerations
When weighing the potential benefits of buffered articaine, it may be helpful to consider patients who do not anesthetize adequately following LA administration. This occurs with some frequency. For example, alcohol use disorder induces changes to pathophysiology, such as cardiomyopathy, central nervous system dysfunction, metabolic acidosis, and liver disease.24 These changes increase the risk of toxicity due to impaired metabolism. An acidic environment at the site of injection reduces the ability of LA drugs to cross nerve membranes. It has been theorized that alcohol-induced physiological changes may be responsible for decreasing the diffusibility of LA agents through these membranes.24
In dental settings, the unique characteristics of 4% solutions of articaine with epinephrine include greater potency and diffusibility, shorter half-life, and reduced potential for liver toxicity. Additionally, buffered articaine helps enhance diffusion across nerve cell membranes, thus boosting anesthetic efficacy.
Known benefits of buffered articaine (especially for patients with alcohol use disorder) include reduced doses of a somewhat lower concentration solution. This decreases articaine’s already lessened impact on the liver since articaine is 90% to 95% metabolized by plasma carboxyesterase. That noted, caution is recommended in patients with severe hepatic disease, which is likely to be encountered with greater frequency in this patient population.16,17
Conclusion
Offering both ester and amide properties, articaine’s pharmacology addresses dentistry’s need for effective and safe anesthesia. Compared to other LA drugs, systematic reviews have shown that 4% articaine demonstrates a greater ability to provide profound anesthesia in a majority of situations. In addition, recent research presents strong evidence in favor of buffering LA agents, as this approach reduces onset times and pain associated with injections, while enhancing the anesthetic effect.12,13
Considering the well documented benefits of articaine for achieving profound anesthesia, buffering articaine prior to injection further enhances what is already one of dentistry’s most effective options for pain management.
References
- de Geus JL, Nogueira da Costa JK, Maran BM, Loguercio AD, Reis A. Different anesthetics on the efficacy of inferior alveolar nerve block in patients with irreversible pulpitis. J Am Dent Assoc. 2020;151:87–97.
- Rathi NV, Khati AA, Agrawal AG, Baliga M, Thosar NR, Deolia SG. Anesthetic efficacy of buccal infiltration articaine versus lidocaine for extraction of primary molar teeth. Anesth Prog. 2019;66:3–7.
- Nagendrababu V, Duncan HF, Whitworth J, et al. Is articaine more effective than lidocaine in patients with irreversible pulpitis? An umbrella review. Int Endod J. 2020;53:200–213.
- Bassett KB, DiMarco AC, Naughton DK. Dental local anesthetic drugs, articaine. In: Local Anesthesia for Dental Professionals. 2nd ed. Upper Saddle River, NJ: Pearson Education; 2015:55–58.
- Ali SG, Mulay S. Articaine vs lidocaine: A review. IOSR J Dent Med Sci. 2014;33:417–435.
- Quinn CL. Injection techniques to anesthetize the difficult tooth. J Calif Dent Assoc. 1998;26:665–667.
- Isen DA. Pharmacology and clinical use of a recently approved local anesthetic. Dent Today. 2000;19:72–77.
- Snoeck M. Articaine: a review of its use for local and regional anesthesia. Local Reg Anesth. 2012;5:23–33.
- Becker DE, Reed KL. Essentials of local anesthetic pharmacology. Anesth Prog. 2006;53:98–109.
- Malamed SF. Handbook of Local Anesthesia. 7th ed. St. Louis: Elsevier Mosby; 2020.
- Martin E, Nimmo A, Lee A, Jennings E. Articaine in dentistry: an overview of the evidence and meta-analysis of the latest randomized controlled trials on articaine safety and efficacy compared to lidocaine for routine dental treatment. BDJ Open. 2021;7:1–13.
- Malamed SF. Articaine 20 Years Later. Available at: dentallearning.net/articaine-20-years-later. Accessed June 3, 2022.
- Donaldson M, Goodchild JH. Taking local anesthesia to the next level: Four strategies clinicians may consider. Compend Contin Educ Dent. 2022:43:20–24.
- Goodchild JH, Donaldson M. Comparing the pH change of local anesthetic solutions using two chairside buffering techniques. Compend Contin Educ Dent. 2016;37:e6–e12.
- Frank SG, Lalonde DH. How acidic is the lidocaine we are injecting, and how much bicarbonate should we add? Can J Plast Surg. 2012;20:71–73.
- Onpharma Co. Onset Buffering System. Available at: onpharma.com. Accessed June, 2022.
- Anutra Medical. Anutra Local Anesthetic Delivery System. Available at: anutramedical.com. Accessed June 3, 2022.
- Goodchild JH, Donaldson M. Novel direct injection chairside buffering technique for local anesthetic use in dentistry. Compend Contin Educ Dent. 2019:40:1–10.
- Kattan S, Lee S-M, Hersh EV, Karabucak B. Do buffered local anesthetics provide more successful anesthesia than nonbuffered solutions in patients with pulpally involved teeth requiring dental therapy? A systematic review. J Am Dent Assoc. 2019;150:165–177.
- Catchlove RF. The influence of CO2 and pH on local anesthetic action. J Pharmacol Exp Ther. 1972;181:298–309.
- Aulestia-Viera PV, Braga MM, Borsate MA. The effect of adjusting the pH of local anaesthetics in dentistry: a systematic review and meta-analysis. Int Endod J. 2018;51:862–876.
- Malamed SF. Buffering local anesthetics in dentistry. Pulse. 2011;44:8–9.
- Malamed SF, Tavana S, Falkel M. Faster onset and more comfortable injection with alkalinized 2% lidocaine with epinephrine 1:100,000. Compend Contin Educ Dent. 2013;34:10–20.
- Tsuchiya H, Mizogami M. Drinking-related tetrahydroharmine counteract the membrane effects of local anesthetic lidocaine. J Drug Alcohol Res. 2014;3:1–6.
From Dimensions of Dental Hygiene. July 2022; 20(7)40-43.



