 DR P. MARAZZI/SCIENCE PHOTO LIBRARY
DR P. MARAZZI/SCIENCE PHOTO LIBRARY
Current Concepts in Caries Risk Assessment
Staying abreast of the changing landscape of caries risk assessment, management, and prevention will help oral health professionals provide the highest quality patient care.
This course was published in the January 2017 issue and expires January 2020. The author has no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Describe the caries disease process.
- Identify the components of caries management by risk assessment.
- Discuss strategies for preventing and managing caries.
The body combats this acidic environment with the help of saliva, which has a natural buffering/neutralizing capacity.2 The introduction of saliva results in a more basic pH level where cariogenic bacteria do not thrive. If an acidic pH is sustained, however, a metabolic shift occurs, resulting in caries lesion formation and progression.2 This shift can occur due to the frequency, quantity, and type of sugars consumed, from lack of regular or effective biofilm removal, and from decreased saliva secretion.2 When simple sugars and fermentable carbohydrates are regularly consumed, the oral environment never returns to a neutral state, where remineralization of enamel can take place.
Biofilm is defined as a matrix of bacteria, their byproducts, and sugars from the diet. It can be disrupted and or removed by mechanical intervention (eg, brushing/flossing). If left undisturbed, the biofilm becomes home to acid-producing bacteria that proliferate. Soon, enamel demineralization predominates, with the consequence being active caries. Poor biofilm removal may be why 42% of children in the United States between the ages of 2 and 11 have dental caries in their primary dentition.3 However, even in a fairly clean mouth, minerals of the enamel are continuously lost and gained. In fact, even in the absence of a clinically visible lesion, demineralization still may occur.2
Saliva’s role in the remineralization process is best seen in patients with hyposalivation, otherwise known as xerostomia. Xerostomia can be the result of medication use, medical treatment, illness, or medical conditions that slow down or arrest saliva production. Because saliva plays a main role in the buffering process after an acid attack, a lack of saliva can lead to an altered oral microflora. The results of this altered environment often lead to dental caries on both enamel and cementum surfaces.2
Modern caries management should focus on the following objectives: decrease the amount and frequency of fermentable carbohydrates in the diet; encourage regular and effective removal of biofilm from the tooth surface; implement caries management by risk assessment (CAMBRA), including the assessment of saliva production; increase the application of professional and over-the-counter sources of topical fluoride in both adults and children; and encourage the placement of dental sealants.
CLASSIFYING CARIES LESIONS
Caries can be primary, with lesions that have developed on intact tooth surfaces, or secondary, with lesions that have developed adjacent to existing restorations.2,4 In addition to primary and secondary lesions, residual lesions are classified by any amount of outstanding demineralized dentin.2 More important, caries lesions are classified by whether cavitation exists.1,2 Visual examination, both clinically and radiographically, should be the primary method for caries detection. The use of a sharp-tipped instrument, such as an explorer, can create an opening on demineralized surfaces, causing inadvertent cavitation.1
A noncavitated lesion is typically limited to demineralization of the subsurface of the tooth where the outer layer of tooth structure is without fracture. These lesions are commonly referred to as white spot lesions, but may appear yellow or brown as the lesion picks up stains from the diet. When the tooth is dried, a brighter more opalescent lesion may appear. These lesions are a more advanced form of noncavitated lesions. They can still be arrested with the use of topical fluorides. Both of the above-mentioned lesions do not exhibit breaks in the tooth structure and are considered noncavitated. However, either one can be in a state of activity or rest. These types of lesions require attentiveness, careful judgment, and increased preventive measures to eliminate the chance of progression.
Cavitated lesions display breaks in the surface of enamel or cementum, perhaps exposing the dentin.1 In many cases, demineralization is so significant, the lesion is visible on a radiograph. At this stage, mechanical intervention may be necessary. The management of dental caries should be based on performing the least invasive treatment required to help restore a balanced oral environment.1 The decision to restore lesions vs remineralize lesions should be based on the patient’s risk factors through CAMBRA.1
UNDERSTANDING CARIES RISK
CAMBRA is an essential part of caries management.5,6 Clinical or radiographic lesions are a historical representation of disease activity. This means current disease activity is difficult to determine, just as future disease activity is challenging to predict. However, extensive evidence has identified key determinants that can predict caries risk. These risk factors are poor or ineffective oral hygiene, existing restorations and/or caries lesions, xerostomia, smoking, and frequency of carbohydrate consumption. Caries management and prevention methods for high-risk patients should be individualized to meet their unique needs. Using a standard way of identifying caries risk is important so that all team members are aware of the management and prevention strategies.4
Many CAMBRA forms are available. The American Dental Association (ADA), American Academy of Pediatric Dentistry, and California Dental Association all have forms for office use. They encourage the management of caries before the lesion exists and agree that the best predictor of future disease is based on patients’ 3-year caries history and current dietary habits. Each group bases its preventive guidelines on a destructive vs protective factor balancing act, meaning the advancement or reversal of dental caries is highly dependent on the balance or imbalance between protective factors and destructive factors.5
New technologies are available that can support standardization of caries risk assessment. For example, an app developed by researchers at the University of California, San Francisco, brings caries risk assessment, planning, and management online.7
MODERN PREVENTIVE STRATEGIES
Patients at high caries risk should undergo a diet analysis as part of a caries management and prevention program.2 A diet analysis is typically a two-part procedure involving the patient and the clinician. The patient journals the foods he or she consumes over 3 days to 4 days, along with day and time of intake. At a subsequent appointment, the dental hygienist and the patient review the journal and investigate some reasons for dental decay. Once a decay source is identified, the patient and dental hygienist can formulate a plan of action on the best way to manage and prevent future caries. This plan may include restorations, dental sealants, increased oral hygiene procedures, saliva substitutes, or topical fluorides. The plan should be revisited at each recare appointment and adjusted according to the patients’ risk and needs.2
Patients contribute to the daily removal of biofilm through oral hygiene measures. Dental biofilm is extremely diverse in its structure and composition. If the biofilm advances in structure due to ineffective removal, many varieties of prokaryotes will grow and populate, excreting an acidic exotoxin. This acidic exotoxin demineralizes the tooth structure.
Saliva is a naturally occurring protective feature of the oral environment and increased caries incidence is often seen in adults with hyposalivation. Patients with dry mouth should avoid consuming sugary foods and drinks. Patients should be instructed on optimal oral hygiene, sip water frequently, and receive a professional fluoride varnish every 3 months.2
Normal salivary flow rates are 0.3 prokaryotes to 0.5 ml/min unstimulated and 1 prokaryotes to 3ml/min when stimulated. A simple salivary flow assessment can be performed by the dental hygienist. Patients should be asked to avoid eating or drinking 30 minutes prior to saliva collection. In an upright seated position, the patient should spit into a disposable cup for 1 minute. Then the saliva is collected into a disposable graduated syringe for measurement. Saliva flow should be documented into the record. It may be difficult to collect saliva samples from patients with xerostomia, as their saliva may be foamy. If this is the case, saliva flow may need to be stimulated, such as having the patient chew on a piece of wax for 5 minutes before expectorating.
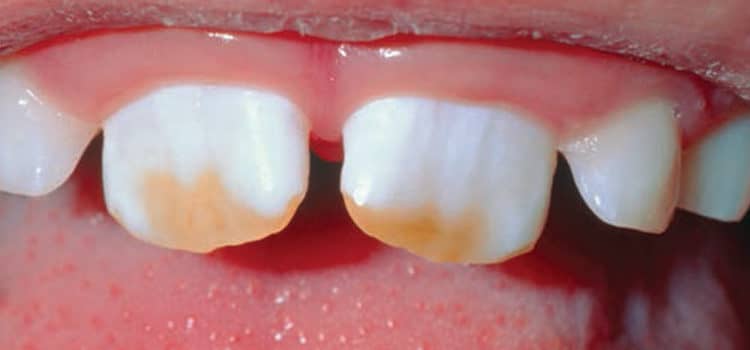
For many years, fluoride was thought to be most effective when ingested systemically. It was theorized that the fluoride ion was most effective at the site of amelogenesis, when the enamel and dentin were still in formation.1,8 Although effective, too much fluoride during tooth development can lead to dental fluorosis (Figure 1). Fluoride maintains a presence in the saliva during the demineralizationremineralization processes.2 In fact, the quantity and efficacy of the actual fluoride ion that is available before, during, and after brushing and rinsing are key.8Researchers and manufacturers are currently working on a new family of toothpastes and mouthrinses that use chemical release strategies to maximize the bioavailability of fluoride ions in the saliva.8 As such, patients at high caries risk should be given customized oral hygiene instruction focusing on effective brushing techniques with a dentifrice that contains 1,000 ppm to 5,000 ppm fluoride for daily at-home use.2,9
Fluoride toothpaste should be used with the eruption of the first tooth.8,10–12 For children age 0 to 2, the US Department of Health and Human Services and ADA Council on Scientific Affairs recommend using a “smear” (approximately 0.1 mg of fluoride or 0.1 g toothpaste) of fluoride toothpaste containing no less than 500 ppm of fluoride.8,11,12 Patients age 2 to 6 should use a pea-sized amount of toothpaste that contains 0.25 mg fluoride two times per day.8,11,12 A metaanalysis of the literature, however, determined that regardless of age, children using fluoride toothpaste should be supervised by a parent in order to prevent overconsumption.8,9,12,13
Recent systematic reviews of topical fluoride use for the management and prevention of dental caries report children and adolescents age 6 to 18 should use a weekly mouthrinse of 0.09% fluoride (900 ppm).8,14 Additionally, patients at high risk for enamel caries or root caries, such as those with existing restorations and/or active caries lesions, should receive professionally applied fluoride varnish at regular 3-month to 6-month intervals.8,10,14,15 Individuals at high caries risk need close monitoring of oral hygiene habits and increased topical application of fluoride varnishes (50,000 ppm NaF/22,600 ppm F).8,14 In addition to managing caries in permanent teeth, the US Preventive Services Task Force recommends that primary care clinicians apply professional fluoride varnish on all erupted primary teeth of infants and children.15
In the early years of community water fluoridation, notable decreases (about 50%) in decayed, missing, and filled teeth were seen in areas with water fluoridation. When fluoridated water is consumed, a low amount of fluoride is absorbed by the biofilm that adheres to enamel surfaces. Nearly 75% of the systemic intake of fluoride in the US comes from the water and processed foods.16 Since 2011, the federal guidelines on community water fluoridation recommend 0.7 ppm in all communities. Currently, approximately 10 million people live in areas that have naturally occurring fluoride in their drinking water.17 Years of water fluoridation have lead to an increase in the fluoride found in processed foods and beverages, including baby formula. Therefore, fluoride supplements should be limited to children who live in areas where they consume water containing less than 0.6 ppm. In addition, fluoride supplements should be adjusted based on age.17
In the fall of 2014, the US Food and Drug Administration (FDA) approved the use of silver diamine fluoride (SDF) to treat dentinal hypersensitivity. Until December 2016, when the FDA noted SDF 38% as a “Breakthrough Therapy Designation” for the arrest of dental caries, SDF was used off-label to prevent cavitation. It is available as a 38% solution that contains 24.4% to 28.8% (w/v) silver and 5.0% to 5.9% fluoride (44,800 ppm) in a basic form (pH of 10).18 Clinical studies demonstrate that SDF is a safe, effective, and economical preventive product. SDF not only aids in the prevention of caries, but it also helps arrest active cavitated lesions. SDF is effective in low but frequent doses, and does not require that all demineralized dentin be fully removed.19–21 In addition, silver ions are bactericidal and interfere with new biofilm formation.
SDF is recommended to treat patients at extremely high caries risk; caries lesions that are difficult to treat (distal of the third molars); patients whose behaviors or medical conditions make dental treatment difficult; patients with multiple active caries lesions that cannot be treated in a single visit; and individuals living in areas with few dental providers.18 SDF is contraindicated in patients with a silver allergy or ulcerative gingivitis/stomatitis. Adverse side effects of SDF include darkening of the demineralized enamel as the SDF forms a tough, impervious layer on the tooth.19 For best results, the areas of active cavitated lesions should be dried then swabbed with a microbrush containing 38% SDF once per week for 3 weeks.19–21
The success of SDF in caries arrest is dependent on repeat applications over multiple years and dryness of the lesion during application. Reviews have indicated that longer application time does not improve the outcome, but it does decrease the need for a post-treatment rinse due to the increased amount of absorption.19 Patients need to understand that multiple applications are necessary and that permanent enamel staining is likely.18
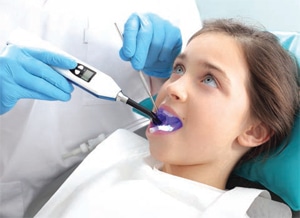
Not all areas on the tooth can be maintained by brushing or flossing alone. The occlusal surfaces of teeth contain deep pits and fissures that are too small in width for a toothbrush to effectively clean. For patients with noncavitated lesions, the use of resin-based dental sealants or glass-ionomer based sealants can prevent and arrest caries in the pits and fissures (Figure 2).22–24
Dental sealants are recommended by the ADA and the Centers for Disease Control and Prevention for the prevention of dental caries in sound teeth. Up to 80% of caries can be prevented through the use of dental sealants.25 Sealants are effective in reducing lesion progression from noncavitated to cavitated.26,27 In 2001, an independent task force reviewed several school-based sealant programs and found that children with sealants had a 60% decrease in overall caries development.26 In addition, literature reviews have concluded that teeth with partial or complete loss of sealants are not at any higher risk for future caries development as compared to teeth that had never received sealants at all.28 Therefore, dental sealants should be a form of caries prevention for children and adolescents who have permanent molars erupting.28
Dental hygienists should stay abreast of the changing landscape of caries risk assessment, management, and prevention measures. From educating patients on effective oral hygiene and healthy nutrition, to knowing how to balance the use of systemic and topical fluorides, it is possible to provide the most up-to-date and effective methods of dental caries management and prevention.
Acknowledgement
The author would like to thank Sheree Duff, RDH, MS, for her help with this manuscript.
REFERENCES
- Fontana M, Cabezas CG, Fitzgerald M. Cariology for the 21st century. J Mich Dent Assoc. 2013;95:32–40.
- Kidd E, Fejerskov O. Essentials of Dental Caries. Oxford, United Kingdom: Oxford University Press; 2016.
- National Institute of Dental and Craniofacial Research. Dental caries in children. Available at: nidcr.nih.gov/DataStatistics/FindDataByTopic/DentalCaries/DentalCariesChildren2to11.htm. Accessed December 26, 2016.
- Pitts NB, Ekstrand KR, ICDAS Foundation. International Caries Detection and Assessment System (ICDAS) and its International Caries Classification and Management System (ICCMS)–methods for staging of the caries process and enabling dentists to manage caries. Community Dent Oral Epidemiology. 2013;41:41–52.
- Young DA, Featherstone JD, Roth J, et al. Caries management by risk assessment: implementation guidelines. J Calif Dent Assoc. 2007;35:799–805.
- Hurlbutt M, Young DA. Best practices approach to caries management. J Evid Based Dent Pract. 2014;14:77–86.
- Ellington B. MyCAMBRA Mobile App streamlines caries risk assessment. J Calif Dent Assoc. 2013;41:905.
- Carey CM. Focus on fluorides: update on the use of fluoride for the prevention of dental caries. J Evid Based Dent Pract.2014;14:95–102.
- Mattana DJ, Relich EE. Fluorides. In Wilkins EM. Clinical Practice of the Dental Hygienist. 12th ed. Philadelphia:Wolters Kluwer; 2017:594–618.
- Clark MB, Slayton RL, Section on Oral Health. Fluoride use in caries prevention in the primary care setting. Pediatrics. 2016;134:626–633.
- American Dental Association Council on Scientific Affairs. Fluoride toothpaste use for young children. J Am Dent Assoc. 2014;145:190–191.
- Wright JT, Hanson N, Ristic H, Whall CW, Estrich CG, Zentz RR. Fluoride toothpaste efficacy and safety in children younger than 6 years. J Am Dent Assoc. 2014;145:182–190.
- Scottish Intercollegiate Guidelines Network. Prevention and Management of Dental Decay in the Preschool Child. Available at: ign.ac.uk/guidelines/fulltext/138/index.html. Accessed December 26, 2016.
- Weyant RJ. Topical fluoride for caries prevention: executive summary of the updated clinical recommendations and supporting systematic review. J Am Dent Assoc. 2013;144:1279–1291.
- US Preventive Services Task Force. Prevention of Dental Caries in Children From Birth Through Age 5 Years. Available at: uspreventiveservicestaskforce.org/Page/Document/UpdateSummaryFinal/dental-caries-in-children-from-birth-through-age-5-years-screening. Accessed December 26, 2016.
- US Department of Health and Human Services Federal Panel on Community Water Fluoridation. US Public Health Service recommendation for fluoride concentration in drinking water for the prevention of dental caries. Public Health Rep. 2015;130:318–331.
- American Academy on Pediatric Dentistry Liaison with Other Groups Committee.; American Academy on Pediatric Dentistry Council on Clinical Affairs. Guideline on fluoride therapy. Pediatr Dent. 2008-2009;30(7 Suppl):121–124.
- Horst JA, Ellenikiotis H, Milgrom PL. UCSF protocol for caries arrest using silver diamine fluoride: rationale, indications and consent. J Calif Dent Assoc. 2016;44:16–28.
- Mei ML, Lo EC, Chun-Hung Chu BD. Clinical use of silver diamine fluoride in dental treatment. Compend Contin Educ Dent. 2016;37:93–98.
- Rosenblatt A, Stamford TC, Niederman R. Silver diamine fluoride: a caries “silver-fluoride bullet.” J Dent Res. 2009;88:116–125.
- Quock R. Silver diamine fluoride: an operative dentistry perspective. J Oper Esthet Dent. 2016;1:6–9.
- Ahovuo-Saloranta A, Hiiri A, Nordblad A, Mäkelä M, Worthington HV. Pit and fissure sealants for preventing dental decay in the permanent teeth of children and adolescents. Cochrane Database Syst Rev. 2008;4:CD001830.
- Griffin SO, Oong E, Kohn W, et al. The effectiveness of sealants in managing caries lesions. J Dent Res. 2008;87:169–174.
- Oong EM, Griffin SO, Kohn WG, Gooch BF, Caufield PW. The effect of dental sealants on bacteria levels in caries lesions: a review of the evidence. J Am Dent Assoc. 2008;139:271–278.
- Centers for Disease Control. Dental sealants prevent cavities. Available at: cdc.gov/vitalsigns/dental-sealants/index.html. Accessed December 26, 2016.
- Gooch BF, Griffin SO, Gray SK, et al. Preventing dental caries through school-based sealant programs: updated recommendations and reviews of evidence. J Am Dent Assoc. 2009;140:1356–1365.
- Beauchamp J, Caufield PW, Crall JJ, et al. Evidence-based clinical recommendations for the use of pit-and-fissure sealants: a report of the american dental association council on scientific Affairs. J Am Dent Assoc. 2008;139:257–268.
- Griffin SO, Gray SK, Malvitz DM, Gooch BF. Caries risk in formerly sealed teeth. J Am Dent Assoc. 2009;140:415–423.
From Dimensions of Dental Hygiene. January 2017;15(1):42-44,47.



