
Causality or Coincidence?
A look at the emerging evidence surrounding the association between periodontal diseases and cardiovascular diseases.
This course was published in the September 2014 issue and September 30, 2017. The author has no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- List the proposed mechanisms of action by which periodontal diseases may cause cardiovascular diseases (CVD).
- Identify the role of infection in CVD.
- Explain the components necessary to determine causality.
- Discuss the evidence that demonstrates an association between periodontal diseases and CVD.
INTRODUCTION
The joint workshop of the American Academy of Periodontology (AAP) and the European Federation of Periodontology on periodontitis and systemic diseases, which took place in November 2012 in Segovia, Spain, discussed at length the question of causality vs coincidence. The workshop was sponsored by Colgate and included more than 90 international experts who reviewed the evidence relating to periodontal diseases and diabetes mellitus, cardiovascular diseases (CVD), adverse pregnancy outcomes, and other systemic illnesses. The objective of the workshop was to help practicing dental professionals make evidence-based decisions with their patients. The proceedings were published in April 2013 in a joint publication of the American Academy of Periodontology and the European Federation of Periodontology in both the Journal of Clinical Periodontology and the Journal of Periodontology.
The Colgate-Palmolive Company is committed to ensuring that practicing dental professionals have access to the latest educational resources and is delighted to have provided an unrestricted educational grant to support a series of articles on topics discussed during the workshop, in collaboration with the AAP. The first article of this series examined the evidence related to diabetes. “The Bidirectional Link” can be found in the March 2014 edition of Dimensions of Dental Hygiene. This second article “Causality or Coincidence?” provides a very thought-provoking review of the proposed mechanisms of causality and the factors needed to establish a causal relationship. I hope you find the article a valuable resource to help manage the patients at risk of CVD in your practice.
—Barbara Shearer, BDS, MDS, PhD
Director of Scientific Affairs
Colgate Oral Pharmaceuticals
FROM THE AMERICAN ACADEMY OF PERIODONTOLOGY
The relationship between periodontal diseases and cardiovascular diseases (CVD) has been well established. Research indicates that periodontal diseases may increase the risk for CVD, similar to smoking, elevated cholesterol levels, and obesity. Despite the fact that research has been unable to conclude that periodontal diseases cause CVD, dental professionals need to understand that this relationship is more than a coincidence. Both periodontal diseases and CVD are complex, multifactorial diseases that are likely mediated by numerous biologic and inflammatory mechanisms. As a result, dental and medical professionals must be armed with the most up-to-date information on this association in an effort to provide the best possible care to patients.
The American Academy of Periodontology (AAP) is committed to supporting the science behind the relationship between periodontal diseases and CVD. Our joint workshop with the European Federation of Periodontology in November 2012 reviewed a variety of studies examining this link and resulted in clinical recommendations that enable dental professionals to determine the most optimal treatment options. On behalf of the AAP, I would like to thank the Colgate-Palmolive Company for its support of this important workshop. Additionally, Colgate-Palmolive has generously provided funding for this ongoing article series. The second installment “Causality or Coincidence?” authored by AAP member Nabil F. Bissada, DDS, MSD, provides an overview of the evidence behind the link between periodontal diseases and CVD, and describes what research is needed to confirm causality. This article dispels the notion that the relationship between periodontal diseases and CVD is simply a coincidence, and serves as a helpful resource in patient care.
—Stuart J. Froum, DDS
President, American Academy of Periodontology
Clinical Professor and Director of Clinical Research, Department of Periodontics
and Implant Dentistry, New York University College of Dentistry
 The fact that oral diseases can affect overall health has been long established. Over the past several years, numerous epidemiologic and clinical studies have reported associations between periodontal diseases and cardiovascular diseases (CVD). Some studies have reported no relationships, which suggest that if such associations are genuine, they may be subtle. Epidemiologic studies imply that the relationships are multifactorial, but such studies are limited by design insofar as they cannot clearly establish causation. Causation is difficult to establish in epidemiology. The implications of establishing a causal relationship between periodontal diseases and CVD are profound for oral health professionals.1–10 If a causal relationship exists, control of periodontal diseases will be as important as smoking, diet, and exercise in both the prevention and management of CVD. 11,12
The fact that oral diseases can affect overall health has been long established. Over the past several years, numerous epidemiologic and clinical studies have reported associations between periodontal diseases and cardiovascular diseases (CVD). Some studies have reported no relationships, which suggest that if such associations are genuine, they may be subtle. Epidemiologic studies imply that the relationships are multifactorial, but such studies are limited by design insofar as they cannot clearly establish causation. Causation is difficult to establish in epidemiology. The implications of establishing a causal relationship between periodontal diseases and CVD are profound for oral health professionals.1–10 If a causal relationship exists, control of periodontal diseases will be as important as smoking, diet, and exercise in both the prevention and management of CVD. 11,12
The proposed mechanisms by which periodontal diseases may cause or aggravate CVD include: 13–18
- The host inflammatory response to periodontitis
- Oral microorganisms that invade endothelial coronary cells
- The role periopathogens may play in acute thromboembolic events during bacteremia
THE ROLE OF INFECTIONS AND PERIODONTAL PATHOGENS IN CARDIOVASCULAR DISEASES

Infections, such as periodontal diseases, may increase blood viscosity while increasing its coagulability. Individuals with high levels of plaque and gingival inflammation show elevated numbers of leukocytes and higher levels of fibrinogen.13 Cytokines, growth factors, and other proinflammatory mediators can also be atherogenic.6,7,16,18–22 Some strains of Streptococcus sanguis, as well as other Gram-negative periodontal pathogens, selectively bind to blood platelets.14 These pathogens result in larger aggregation on heart valves, greater leukocytosis, more myocardial ischemia, and higher mortality rates.1,4,13
Periodontal pathogens also produce a systemic immunological challenge via the release of bacterial endotoxins. Nguyen et al23 demonstrated the presence of Porphyromonas gingivalis lipopolysaccharide in atheromatous plaque of coronary arteries using immunohistochemical and polymerase chain reaction in autopsy studies of patients with a history of CVD. Their work showed that 27% (four out of 15 specimens) of the coronary vessels harbored P. gingivalis lipopolysaccharide in the tunica media of the involved blood vessel (Figure 1), and not in other examined vessels (renal, abdominal aorta, and hepatic arteries). The authors concluded that localization of P. gingivalis lipopolysaccharide, specifically in coronary arteries, indicates a possible tissue attraction toward this blood vessel. This finding further supports the existence of a link between the periodontal pathogen P. gingivalis and CVD.
THE INFLUENCE OF THE INFLAMMATORY PROCESS ON DISEASE DEVELOPMENT
Individuals who ignore cardiac risk factors, such as smoking, diabetes, and elevated blood pressure, may also neglect their oral health.24 The possibility that oral infectious agents might directly or indirectly trigger the cascade of biological and biochemical reactions leading to inflammation, atherosclerosis, and vascular thrombotic events has been reported.25–27
Bazile et al28 noted an association between gingival inflammation and CVD—specifically acute myocardial infarction. The fact that both gingivitis or early periodontitis and CVD are preceded or accompanied by inflammatory processes suggests a relationship. Both conditions are chronic with acute episodic occurrences. Table 1 lists the questions that must be answered in order to establish causality.
EVIDENCE BASE SUPPORTING THE ASSOCIATION BETWEEN PERIODONTAL DISEASES AND CARDIOVASCULAR DISEASES
To date, there is substantial evidence supporting the association between periodontal diseases and CVD. This relationship could be due to the similarity of risk factors between the two diseases,12 including:
- Sociodemographic characteristics—age, race, gender, and socioeconomic status16
- Systemic factors—diabetes and genetics29–30
- Health behaviors—smoking, stress, diet, physical activity, and obesity1
These common risk factors may be either coincidental (confounding variables) or common sources of disease.31
Several studies have examined the hypothesis that there is a causal relationship between periodontal diseases and CVD. For example, a series of clinical, epidemiologic, and microbiologic investigations in live patients and in cadavers was conducted in the Department of Periodontics at Case Western Reserve University (CWRU) in Cleveland. In one cross-sectional study, patients with CVD were found to have fewer remaining teeth than those without CVD.29 In another cross-sectional study, a significant positive relationship between signs of clinical gingival inflammation and CVD also was reported.28 Furthermore, a higher C-reactive protein (CRP) concentration was found in patients with unstable angina or myocardial infection and severe periodontitis than in patients with stable angina and mild periodontitis.22
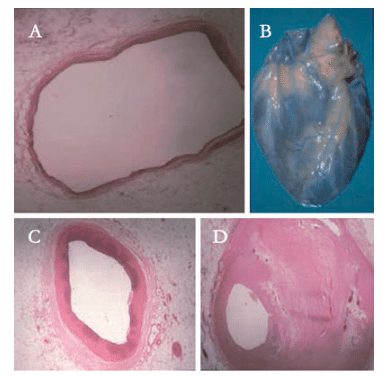
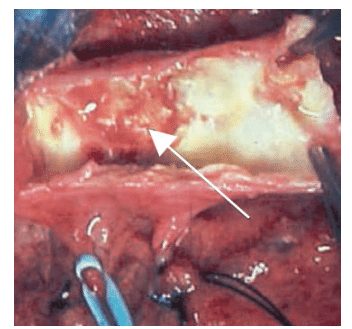
The presence of bacterial DNA in coronary arteriosclerotic blood vessels also was demonstrated in an autopsy study at CWRU.32 Of the 41 examined coronary atheromas, 95% (39 out of 41 specimens) contained bacterial DNA. Of these, 49% (20 out of 41 specimens) were infected with at least one periodontal pathogen and 50% (10 out of 20 specimens) of the infected atheromas were associated with the presence of more than one periodontal pathogen. As seen in Figure 2A through Figure 2D, the narrower the involved atherosclerotic blood vessel, the higher the prevalence of periodontal pathogens.32 Furthermore, in a follow-up in vivo study, a positive trend was seen in specific bacterial DNA in coronary atheroma that exactly matched subgingival bacterial DNA in dental plaque of the same individual.33 In an autopsy study of patients with a history of CVD, P. gingivalis DNA was not only found within the atheroma lesion, but also in parts of the coronary artery slightly removed from the atheroma lesion (Figure 3). The authors concluded that the in vivo presence of P. gingivalis in nonatheromatous sites of the coronary artery suggests that this periopathogen can actively invade arterial intima rather than be coincidentally trapped by the existing atheroma.34 This indicates that P. gingivalis may play a role in infection, culminating in the development of an atheromatous lesion. Interestingly, not only bacterial DNA, but also P. gingivalis lipopolyaccarides and interleukin-1B were detected in the atheromatous plaques.34
The association between periodontitis and atherosclerosis has also recently been evaluated by several indirect measures of atherosclerosis.4,25–27 For example, the intima-media thickness (IMT) of the carotid arteries in relation to periodontitis has been assessed in 19 studies, 11 of which found a higher IMT in patients with periodontitis. Interestingly, nonsurgical periodontal treatment has been shown to improve arterial function and/or reduce severity of atherosclerosis 6 months to 12 months after intervention,5,6,9 demonstrating that the regeneration rate in the IMT is relatively slow. Findings of a recent study also indicate that a single session of nonsurgical treatment for periodontal diseases significantly reduced the thickness of artery walls, a risk factor for CVD.13
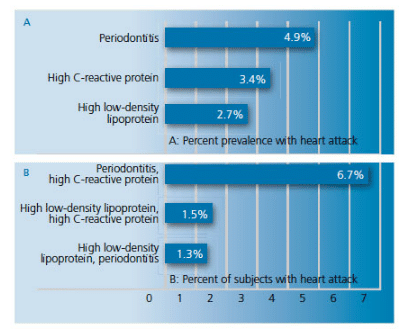
A report on the link between periodontal diseases and CVD in the National Health and Nutrition Examination Survey (NHANES) III showed that the likelihood that individuals had a history of heart attack increased with the severity of periodontal diseases.35 Tehrani et al36 compared the prevalence of heart attack among patients in NHANES III with periodontal diseases and two known systemic risk factors of CVD—high CRP (? 3 mg/dl) and elevated low-density lipoprotein levels ?( 3 mg/dl), either separately or in combination. The authors found that patients with periodontal diseases and increased levels of CRP had the highest prevalence (6.7%) of past history of heart attack compared to patients with only periodontitis, elevated CRP, or increased low-density lipoprotein levels (4.9%, 3.4%, 2.7%, respectively, Figure 4). The authors suggest that chronic infection/inflammation may play a role in atherogenesis and myocardial infarction.36
CONSIDERATIONS FOR FURTHER UNDERSTANDING THE RELATIONSHIP BETWEEN PERIODONTAL DISEASES AND CARDIOVASCULAR DISEASES
 Substantial evidence documents an association between periodontal diseases and CVD, although a causal relationship has yet to be shown. The association may be due to an underlying inflammatory response trait, which places an individual at high risk for developing both periodontal diseases and atherosclerosis.
Substantial evidence documents an association between periodontal diseases and CVD, although a causal relationship has yet to be shown. The association may be due to an underlying inflammatory response trait, which places an individual at high risk for developing both periodontal diseases and atherosclerosis.
Periodontitis, once established, engenders a biological burden of endotoxin (lipopolysaccharide) and inflammatory cytokines, which serve to initiate and exacerbate atherogenesis and thromboembolic events. Although periopathogens have been detected in carotid and coronary atheromas, their exact role needs to be further investigated.
It seems that interventional periodontal care is invaluable not only for oral health, but for systemic health as well. The development of a wide-ranging system of prevention that incorporates multiple behavioral risk factors related to both periodontal diseases and CVD, instead of addressing risk factors related to only one disease may prove beneficial for the health of the general public. Involving oral health professionals in a broad effort to promote healthy behaviors, in addition to their role in treating and managing periodontal and oral infections, is warranted.
To confirm the causality relationship between periodontal diseases and CVD, additional studies should be conducted to establish a precise dose-response relationship (ie, increased severity of periodontal diseases means more frequent or more severe CVD). Also, the development of a time-sequence relationship (ie, periodontal diseases occur before CVD) needs to be determined. Future research should also focus on definitive diagnosis of existing systemic disease(s); identification of specific periodontal pathogens; and the role of other confounding factors, such as genetics, blood cholesterol level, stress, and the effect of different medications on host response.
REFERENCES
- Morrison HI, Ellison LF. Periodontal disease and risk of fatal coronary heart and cerebrovascular diseases. J Cardiovasc Risk. 1996;6:7–11.
- Mercanoglu F, Oflaz H, Oz O, et al. Endothelial dysfunction in patients with chronic periodontitis and its improvement after initial periodontal therapy. J Periodontol. 2004;75:1694–1700.
- Meurman JH, Sanz M, Janket SJ. Oral health, atherosclerosis, and cardiovascular disease. Crit Rev Oral Biol Med. 2004;15:403–413.
- Desvarieux M, Demmer RT, Rundek T, et al. Periodontal microbiota and carotid intima-media thickness: the Oral Infections and Vascular Disease Epidemiology Study (INVEST). Circulation. 2005;111:576–582.
- Seinost G, Wimmer G, Skerget M, et al. Periodontal treatment improves endothelial dysfunction in patients with severe periodontitis. Am Heart J. 2005;149:1050–1054.
- Elter JR, Hinderliter AL, Offenbacher S, et al. The effects of periodontal therapy on vascular endothelial function: a pilot trial. Am Heart J. 2006;151:47.
- Blum A, Front E, Peleg A. Periodontal care may improve systemic inflammation. Clin Invest Med. 2007;30:E114–E117.
- Spahr A, Klein E, Khuseyinova N, et al. Periodontal infections and coronary heart disease: role of periodontal bacteria and importance of total pathogen burden in the Coronary Event and Periodontal Disease (CORODONT) study. Arch Intern Med. 2006;166:554–559.
- Tonetti MS, D’Aiuto F, Nibali L, et al. Treatment of periodontitis and endothelial function. N Engl J Med. 2007;356:911–920.
- Piconi S, Trabattoni D, Luraghi C, et al. Treatment of periodontal disease results in improvements in endothelial dysfunction and reduction of the carotid intima-media thickness. FASEB J. 2009;23:1196–1204.
- Al-Zahrani MS, Borawski EA, Bissada NF. Periodontitis and three health-enhancing behaviors: normal weight, recommended level of exercise and high-quality diet. J Periodontol. 2005;7:1362–1366.
- Al-Zahrani M, Kayal R, Bissada NF. Periodontitis and cardiovascular disease: a review of shared risk factors and new findings supporting a causality hypothesis. Quintessence Int. 2006;37:41–48.
- Kapellas K, Maple-Brown LJ, Jamieson LM, et al. Effect of periodontal therapy on arterial structure and function among aboriginal Australians: a randomized, controlled trial. Hypertension. 2014 Jun 23. Epub ahead of print.
- Herzberg MC, Meyer MW. Effects of oral flora on platelets: possible consequences in cardiovascular disease. J Periodontol. 1996;67(Suppl 10):1138–1142.
- Kinane DF. Periodontal diseases’ contributions to cardiovascular disease: an overview of potential mechanisms. Ann Periodontol. 1998;3:142–150.
- Genco R, Offenbacher S, Beck J. Periodontal disease and cardiovascular disease: epidemiology and possible mechanisms. J Am Dent Assoc. 2002;133:14S–22S.
- Alzahrani AS, Bissada NF, Jurevic RJ, Narendran S, Nouneh IE, Alzahrani MS. Reduced systemic inflammatory mediators after treatment of chronic gingivitis. Saudi Med J. 2013;34:415–419.
- Schenkein HA, Loos BG. Inflammatory mechanisms linking periodontal diseases to cardiovascular diseases. J Clin Periodontol. 2013;40(Suppl 14):51S–69S.
- Han YW, Koucken W, Loos BG, Schenkein HA, Tezal M. Periodontal disease, atherosclerosis, adverse pregnancy outcomes, and head-and-neck cancer. Adv Dent Res. 2014;26:47–55.
- Ridker PM, Rifai N, Stampfer MJ, Hennekens CH. Plasma concentration of interleukin-6 and the risk of future myocardial infarction among apparently healthy men. Circulation. 2000;101:1767–1772.
- Noack B, Genco RJ, Trevisan M, et al. Relation between periodontal infection and C-reactive protein. J Periodontol. 2001;72:1221–1227.
- Davis R, Bissada N, DeLuca D, Weinberg A, Nair R. C-reactive protein levels and bacterial DNA presence in patients with coronary artery and periodontal diseases. J Dent Res. 2003;82[abstract]:174.
- Nguyen S, Bissada N, Weinberg A, DeLuca D, McCormick T. Identification of P. gingivalis LPS and IL-1ß in atheromatous plaques of coronary arteries. J Dent Res. 2003;82[abstract]:882.
- DeStefano F, Anda RF, Kahn HS, Williamson DF, Russell CM. Dental disease and risk of Coronary heart disease and mortality. Br Med J. 1993;206:669–691.
- Beck JD, Elter JR, Heiss G, et al. Relationship of periodontal disease to carotid artery intima-media wall thickness: the Atherosclerosis Risk In Communities (ARIC) study. Arterioscler Thromb Vasc Biol. 2001;21:1816–1822.
- Beck JD, Eke P, Lin D, et al. Associations between IgG antibody to oral organisms and carotid intimamedial thickness in community-dwelling adults. Atherosclerosis. 2005;183:342–348.
- Higashi Y, Goto C, Hidaka T, et al. Oral infection-inflammatory pathway, periodontitis, is a risk factor for endothelial dysfunction in patients with coronary artery disease. Atherosclerosis. 2009;206:604-610.
- Bazile A, Bissada N, Nair R, Siegel B. Periodontal assessment of patients undergoingangioplasty for the treatment of coronary artery disease. J Periodontol. 2002;73:631–636.
- Mirmadjlessi N, Bissada N, Nelson S, Wotman S. Systemic factors and graded periodontal disease. J Dent Res. 1999;78[abstract]:1299.
- Schaefer AS, Richter GM, Groessner-Schreiber B, et al. Identification of a shared genetic susceptibility locus for coronary heart disease and periodontitis. PLoS Genet. 2009;5:e1000378.
- Lockhart PB, Bolger AF, Papapanou PN, et al. Periodontal disease and atherosclerotic vascular disease: does the evidence support an independent association?: a scientific statement from the American Heart Association. Circulation. 2012;125:2520–2544.
- Meyers G, Bissada N, DeLuca D, et al. Periodontal pathogens in the atheromas of coronary arteries. J Dent Res. 2001;80[abstract]:1605.
- Goel S, Bissada N, DeLuca D, et al. Presence of periodontal pathogens on coronary balloon catheters following angioplasty. J Dent Res. 2002;81[abstract]:828.
- Cataldo J, De Luca DJ, Bissada N, et al. Pg DNA identified in non-atheromatous sites of Human Coronary arteries. J Dent Res. 2002;81[abstract]:831.
- Arbes SJ, Jr, Slade GD, Beck JD. Association between extent of periodontal attachment loss and selfreported history of heart attack: An analysis of NHANES III data. J Dent Res. 1999;78:1777–1782.
- Tehrani E, Bissada N, Jasinevicius T, Al-Zahrani M, Nair R. Mycocardial infarction in patients with periodontitis, high CRP and LDL [abstract 2320]. J Dent Res. March 23, 2007.
From Dimensions of Dental Hygiene. September 2014;12(9):63–68.




