 MICROSCAPE / SCIENCE SOURCE
MICROSCAPE / SCIENCE SOURCE
Beyond Bacteria
Bacteria may not be the only culprits behind periodontitis. New research suggests a viral component may also play a role in specific cases.
This course was published in the December 2015 issue and expires December 21, 2018 2021. The authors have no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Discuss the incidence and prevalence of periodontal diseases.
- Identify the etiology of periodontal diseases.
- Explain the role that viruses may play in the development of chronic periodontitis.
- List possible treatment options for viral-related chronic periodontitis.
Dental hygienists encounter a variety of oral health beliefs and self-care behaviors, as well as different levels of periodontitis in their patient populations. Sometimes, despite thorough treatment planning and the provision of nonsurgical periodontal therapies, patients with chronic periodontitis fail to demonstrate the desired treatment outcomes. Researchers have been studying the factors that may contribute to unique periodontal cases in which bacteria alone cannot explain periodontal destruction. These cases include periodontal breakdown with minimal biofilm, periods of latency, site-specific breakdown, and advanced breakdown in only a fraction of the population.1–8 In an effort to explain the etiological factors that might work with pathogenic bacteria to aggravate periodontitis, research has led some dental professionals to consider that certain viruses may be involved. The herpes viruses, specifically Epstein-barr virus and cytomegalovirus, have been the focus of many of these studies. Their prevalence among the United States population, especially those of certain cultures, may provide the dental community with a new outlook on the etiology of periodontal diseases.
According to the US Centers for Disease Control and Prevention, periodontitis affects 47.2% of adults older than 30.9 As individuals age, specifically those age 65 and older, the prevalence of periodontitis increases to 70.1%. Table 1 includes more data about periodontitis in various groups.9 Considering the fact that almost half of adults have some level of periodontitis, dental hygienists need to be aware of the etiology of chronic periodontitis and emerging evidence regarding additional pathological factors associated with this prevalent disease.
ETIOLOGICAL FACTORS
Periodontal disease is characterized as an “inflammatory disease determined by the presence of microorganisms in the subgingival crevice, able to induce an inflammatory response, eventually leading to resorption of alveolar bone and periodontal ligament.”10 An astounding number of microorganisms inhabit the oral cavity and gingival sulcus. Studies have specifically identified more than 1,200 bacterial species in the oral cavity and 400 bacterial species in the gingival sulcus.8,11 While numerous bacteria reside in the gingival sulcus, the bacteria most closely associated with periodontitis are Porphyromonas gingivalis, Tannerella forsythia, Prevotella Intermedia, Fusobacterium nucleatum, Treponema denticola, Campylobacter rectus, and Aggregatibacter actinomycetemcomitans.3,5,7,11 Bacterial inhabitants associated with periodontitis are anaerobic, proteolytic, and Gram negative.7,8,11,12 In contrast, a healthy periodontium consists of bacteria that are facultative, fermentive, and Gram positive.7,8
Chronic periodontitis (previously known as adult periodontitis) is often associated with age, genetics, environment, demographics, immunogenic effects, and/or systemic disease.8 Slots13 stated, “the current consensus is that the etiopathogeny of periodontitis entails a multifaceted dynamic interaction of periodontopathic bacteria and viruses, innate and adaptive immune responses, adverse environmental events, and genetic susceptibility factors.” If viruses play a part in periodontitis, it is important to understand their roles in periodontal breakdown.
HUMAN HERPESVIRIDAE
There are eight members of the herpesviridae family.1 After a primary infection caused by human herpesvirus (HHV), the virus lies latent in various cells for the life of the host with periodic cycles of reactivation.1,12 Active episodes of HHV have been linked to stress and/or immune suppression.1,12 Combined, the HHV family infects 80% to 90% of the adult population.3 The most commonly discussed members of this family are herpes simplex 1 and herpes simplex 2. This article will focus on Epstein-Barr virus (EBV, HHV 4) and cytomegalovirus (CMV, HHV 5). Table 2 lists the characteristics of EBV and CMV including transmission, symptoms, diagnosis, and treatment.
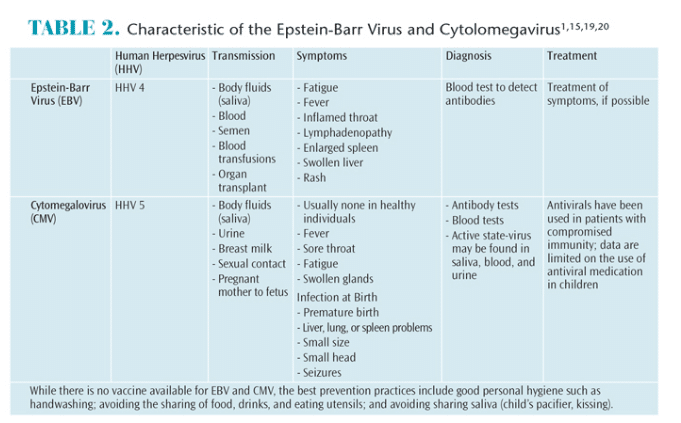
EBV is an enveloped virus consisting of double-stranded DNA.5,14 This virus affects 90% of the population worldwide, and 25% of those infected develop infectious mononucleosis.2,5,15–17 EBV can also cause conditions such as viral meningitis, encephalitis, Guillain-Barre syndrome, and, in more severe cases, Burkitt lymphoma and Hodgkins disease.5,15 EBV cell replication takes place in the epithelial cells of the oropharynx and in B lymphocytes.14 During periods of latency, EBV is harbored in B lymphocytes, allowing viral secretion into the saliva, which provides an outlet for virions to enter the oral cavity.12,15–18 Typically, B lymphocytes target inflammation. As periodontitis is an inflammatory disease, it is thought that these cells may be directed to periodontal tissues and contribute to periodontal destruction.4
CMV is also an enveloped virus consisting of double-stranded DNA.14 Approximately, 50% to 80% of adults in the US are infected with CMV by age 40.2,19 Congenital CMV occurs when a mother passes the infection to her fetus.19 During pregnancy, 1% to 4% of women develop a primary CMV infection. Of these women, 33% pass the virus to the fetus as the mother’s blood crosses the placenta. One in 150 newborns is infected with CMV.19 While CMV is usually harmless, one in five develop permanent conditions such as hearing and/or vision loss, developmental disabilities, and lack of coordination. In rare cases, CMV can cause death.19,20
Viral replication occurs mainly in the salivary glands and kidneys, from which virions are shed into the saliva and urine.14 Unlike EBV, CMV is harbored in the T lymphocytes.4 But like B lymphocytes, T lymphocytes can also spur inflammation, making these cells active contributors to periodontitis.4
ORAL HEALTH CARE
Researchers have proposed that the herpes virus can infect the cells of the periodontium and reduce the ability of periodontal tissues to manage bacterial insults.3 Because EBV and CMV lie dormant in B lymphocytes and T lymphocytes, activation of these viruses, due to immunosuppression of the host, might facilitate the overgrowth of periodontopathic bacteria, aiding in disease progression.4,16 Additionally, HHV virions might accommodate oral bacteria by suppressing host resistance to bacterial colonization of periodontal pathogens.1
Slots et al16 proposed the following model mapping the progression of periodontitis associated with EBV:
- EBV-infected B lymphocytes are released into the periodontium after initiation of the host immune response as a result of bacteria-induced gingivitis.
- Impairment of the host immune defense due to infection, drugs, tissue trauma, stress, etc, allows for the activation of latent EBV in the periodontium. Spontaneous activation of latent EBV might also be to blame.
- EBV-restricted cellular cytotoxicity is rendered insufficient, allowing an outgrowth of EBV-infected B lymphocytes and the release of tissue-damaging mediators.
- Periodontal neutrophils and other host responses critical in controlling periodontitis-causing bacteria might also be restricted.
- Disease remission occurs after herpesvirus is controlled.
Numerous studies have been conducted in an attempt to associate these viruses with periodontitis and its progression. Bilder et al1 conducted a study of HHV in the saliva of patients with chronic periodontitis in comparison to healthy control groups. In this study, EBV was found in approximately 30% of all subjects, and 15% of the participants with chronic periodontitis had CMV in their saliva.1 Dawson et al4 found EBV virions in the saliva of patients with chronic periodontitis, while CMV was rarely found. These researchers concluded that EBV virions found in the saliva meant that the virions were present in subgingival plaque but not necessarily related to periodontal breakdown.4
In contrast, Vincent-Bugnas et al18 conducted a study in which EBV was shown to inhabit gingival epithelial cells and was present in increased amounts among patients with chronic periodontitis. EBV was also found to correlate to disease severity by producing larger amounts of CCL20, an inflammatory chemokine that controls the infiltration of immune cells.18 Bilichodmath et al2 conducted a study on South Indian patients with chronic periodontitis. HSV 1 was detected in 100% of the samples; HSV 2 was found in 15.7% of the samples; EBV was detected in 78.9% of the samples; and CMV was noted in 26.31% of the samples.2 In patients with aggressive periodontitis, detection statistics were: herpes virus 1: 57.14%; herpes virus 2: 0; EBV: 28.57%; and CMV: 7.14%, respectively.2
A study conducted in Iran found higher levels of EBV-1 and CMV in deeper periodontal sites.3 Other studies found that patients with chronic periodontitis exhibited statistically significant levels of EBV in deeper periodontal sites.5,6 Saygun et al7 conducted a study involving 15 periodontally involved patients in which CMV was detected in eight periodontal lesions and in one normal periodontal site, while EBV was detected in nine periodontal lesions.
While the previously mentioned studies suggest there may be a missing link related to the etiology of chronic periodontitis, other researchers have questioned the outcomes of the research. Stein et al21 and Nibali et al10 note that most of the studies showing a positive correlation of viral involvement utilized small sample sizes, limiting the ability to generalize results. Another potential problem is that the studies used polymerase chain reaction assays to detect EBV and CMV. This ultrasensitive molecular technique can result in false-positives because of sample contamination. Further, these studies were performed in ethnically mixed populations in which the viruses are more prevalent. The weaknesses in the research suggest that the prevalence of viruses in periodontal disease is probably not universal and may be more dependent on the population characteristics of the sample.10
Dawson et al12 conducted a study of 450 periodontal sites, a much larger sample than previous studies. Of the sites tested for the presence of CMV and EBV DNA, there was no statistical significance showing the viruses were more likely to be found in subgingival plaque at chronic periodontitis sites.12 Nibali et al10 conducted a study with patients of diverse ethnic backgrounds; however, the majority were of European descent. Findings showed that viruses were not statistically significant in subgingival areas. Stein et al21 conducted a study of a central European Caucasian population and found that herpes virus 1, EBV, and CMV were rarely present in subgingival plaque, even though the patients were infected with the virus.
TREATMENT CONSIDERATIONS
Periodontal treatment typically consists of two phases: stabilization (may include regenerative procedures) and maintenance.8 In the stabilization phase, dental hygienists often perform nonsurgical periodontal therapy that includes periodontal debridement to remove biofilm, calculus deposits, and endotoxins from the root surfaces.8,11 They may also incorporate antimicrobial agents such as oral antibiotics, subgingival irrigation with chlorhexidine or povidone iodine, sustained slow-release subgingival antimicrobials, or laser therapy.8 In cases that do not respond to nonsurgical periodontal therapy, periodontal surgery may be indicated.8 The maintenance phase consists of motivating patients to perform adequate self-care to achieve biofilm control.8 These patients may require more frequent recare intervals.8 If patients continue to present with chronic periodontal problems, additional protocols may be indicated.
Sunde et al17 presented a case in which a 63-year-old man with recurrent periodontitis had a viral load of EBV 100 times greater than the minimal detectable limit. He was treated with antiviral therapy to arrest periodontal tissue breakdown. The patient exhibited periodontal pockets ?6 mm after nonsurgical periodontal therapy. Upon consultation with a virologist, the patient was given 500 mg of valacyclovir twice a day for 10 days. At subsequent evaluations, the patient’s viral load decreased to the detectable limit and remained consistent. In addition, there was an improvement in pocket depths and reduced bleeding. Periodontal stability was noted 1 year post-treatment.
Slots11 also endorses the use of antiviral therapy after microbial sampling is completed. Table 3 demonstrates his recommended sequence of periodontal therapy when considering the use of antivirals. Viral therapy should be considered when a patient’s viral load is much higher than normal or there are other signs of viral activity.17
Currently, there are no easily accessible tests to screen periodontal patients for EBV and CMV. In order to establish the viral load, the patient would need to visit his or her physician for a blood test. The physician would confirm that viral loads were significantly elevated and that antiviral therapy was indicated. Although research continues to evolve in this area, dental hygienists should consider a virology evaluation for patients who appear to be nonresponsive to traditional periodontal therapy. If the patient tests positive for EBV, CMV, or other viral pathology, antiviral therapy, in conjunction with nonsurgical periodontal therapy, might be helpful in arresting the patient’s periodontitis and preventing further periodontal destruction. Current research, however, is limited to case studies and lower level evidence. High-level evidence, such as randomized controlled clinical trials, would strengthen support for the use of antiviral therapy in the treatment of some forms of periodontitis.
CONCLUSION
While controversy exists over the presence and involvement of EBV and CMV in periodontal breakdown, further research investigating the relationship of these viruses to the progression of periodontitis and the usefulness of antivirals is warranted. Cautious researchers make many valid points when evaluating the ability to generalize the results of studies establishing viral involvement of periodontal breakdown. Small sample sizes of ethnically diverse populations, however, are transferrable to today’s dental clientele. Cultural diversity is part of the modern landscape, and dental hygienists need to consider diseases and conditions that may be more prevalent in specific races/ethnicities. Treating patients as individuals enables dental hygienists to consider unique and nontraditional approaches to improving periodontal health. This is especially true in patients whose self-care is good and conventional professional approaches are well-executed, but success remains elusive. Viral testing and antiviral therapy may be considered for patients whose response to conventional periodontal procedures and good self-care is suboptimal.
REFERENCES
- Bilder L, Elimelech R, Szwarcwort-Cohen M, Kra-Oz Z, Machtei EE. The prevalence of human herpes viruses in the saliva of chronic periodontitis patients compared to oral health providers and healthy controls. Arch Virol. 2013;158:1221–1226.
- Bilichodmath S, Mangalekar SB, Sharma DC, et al. Herpesviruses in chronic and aggressive periodontitis patients in an Indian population. J Oral Sci. 2009;51:79–86.
- Chalabi M, Rezaie F, Moghim S, Mogharehabed A, Rezaei M, Mehraban B. Periodontopathic bacteria and herpesvirus in chronic periodontitis. Mol Oral Microbiol. 2010;25:236–240.
- Dawson DR, Wang C, Danaher RJ, et al. Salivary levels of Epstein-Barr virus DNA correlate with subgingival levels, not severity of periodontitis. Oral Diseases. 2009;15:554–559.
- Kato A, Imai K, Ochiai K, Ogata Y. Higher prevalence of Epstein-Barr virus DNA in deeper periodontal pockets of chronic periodontitis in Japanese patients. PLoS One. 2013;8:e71990.
- Rotola A, Cassai E, Farina R, et al. Human herpesvirus 7, Epstein-barr virus and human cytomegalovirus in periodontally diseased and healthy subjects. J Clin Periodontol. 2008;35:831–837.
- Saygun I, Kubar A, Sahin S, Sener K, Slots J. Quantitative analysis of association between herpesvirus and bacterial pathogens in periodontitis. J Periodontal Res. 2008;43:352–359.
- Slots J. Human viruses in periodontitis. Periodontol 2000. 2010;53:89–110.
- Centers for Disease Control and Prevention. 2015 Periodontal Disease. Available at: cdc.gov/ oralhealth/periodontal_disease/index.htm. Accessed November 13, 2015.
- Nibali L, Atkinson C, Griffiths P, et al. Low prevalence of subgingival viruses in periodontitis patients. J Clin Periodontol. 2009;36:928–932.
- Slots J. Low-cost periodontal therapy. Periodontol 2000. 2012;60:110–137.
- Dawson DR, Wang C, Danaher RJ, et al. Real-time polymerase chain reaction to determine the prevalence and copy number of Epstein-Barr virus and cytomegalovirus DNA in subgingival plaque at individual healthy and periodontal disease sites. J Periodontol. 2009;80:1133–1140.
- Slots J. Periodontology: Past, present, perspectives. Periodontol 2000. 2013;62:7–19.
- Whitley RJ. Herpesvirus. In S. Baron, ed. Medical Microbiology. 4th ed. Galveston, Texas: University of Texas Medical Branch at Galveston; 1996.
- Crawford DH, Macsween KF, Higgins CD, et al. A cohort study among university students: Identification of risk factors for Epstein-Barr virus seroconversion and infectious. Clin Infect Dis. 2006;43:276–282.
- Slots J, Saygun I, Sabeti M, Kubar A. Epstein-Barr virus in oral diseases. J Periodontal Res. 2006;41:235–244.
- Sunde PT, Olsen I, Enersen M, Grinde B. Patients with severe periodontitis and subgingival Epstein-Barr virus treated with antiviral therapy. J Clin Virol. 2008;42:176–178.
- Vincent-Bugnas S, Vitale S, Mouline CC, et al. EBV infection is common in gingival epithelial cells of the periodontium and worsens during chronic periodontitis. PLoS One. 2013;8:e80336.
- Centers for Disease Control and Prevention. 2014 Cytomegalovirus and Congenital CMV Infection. Available at: cdc.gov/epstein-barr/about-mono.html. Accessed November 13, 2015.
- Cannon MJ, Schmid DS, Hyde TB. Review of cytomegalovirus seroprevalence and demographic characteristics associated with infection. Rev Med Virol. 2010;20:202–213.
- Stein JM, Said Yekta S, Kleines M, et al. Failure to detect an association between aggressive periodontitis and the prevalence of herpesviruses. J Clin Periodontol. 2013;40:1–7.
From Dimensions of Dental Hygiene. December 2015;13(12):46–48,51.



