Best Practices for Surface Disinfection
Improve your understanding of cross-contamination and disease transmission in the dental setting.
With the focus of most infection control procedures centered around mitigating the spread of the novel coronavirus since early 2020, it is important to review and discuss infection control procedures in the dental setting for the post-COVID era. A survey of infection control practices among dentists during the pandemic showed an increase in knowledge and higher adherence to infection control guidelines.1
In addition to SARS-CoV-2, other highly infectious and potentially life-threatening diseases can spread in a dental setting. Infectious diseases transmissible in dental offices include viruses, such as herpes simplex virus (HSV), norovirus, coxsackievirus, hepatitis, human immunodeficiency virus (HIV), cytomegalovirus, measles, mumps, rubella, and respiratory viruses, and bacteria, such as Staphylococcus aureus, Escherichia coli, Neisseria gonorrhoeae, Treponema pallidum, Streptococcus pyogenes, Mycobacterium tuberculosis, Legionella pneumophila, and Pseudomonas aeruginosa.2
Multiple routes of disease transmission exist in a dental setting. Direct or indirect contact with contaminated fluids can occur during dental procedures. Additionally, infectious agents can be spread by aerosols.3 Transmission of infectious agents through indirect contact occurs by cross-contamination of environmental surfaces or objects.
COMMON INFECTIONS
Any microorganism that colonizes the oral cavity or the bloodstream can spread in a dental environment. Among the most commonly found microorganisms are HSV 1 and 2, HIV, hepatitis B and C, Staphylococcus, and Streptococcus.3 These pathogens are transmitted from bodily fluids, such as the blood or saliva of an infected individual, to a potentially susceptible host.
A 2015 study of environmental surfaces in a dental setting showed contamination in 38% of the cases.4 Based on the findings of the study, the most common microorganisms were Gram-negative bacteria (Klebsiella, Pseudomonas aeruginosa, Escherichia coli, and Enterobacter). The presence of P. aeruginosa and aerobic spore-baring bacilli was noted in 6% of the investigated samples. The presence of fungi was found only in one sample.4 Staphylococcus aureus is also spread by indirect contact at any clinical facility and is a potentially deadly nosocomial bacterial infection.2
Many of these microorganisms can survive outside of the host. Environmental surfaces serve as a reservoir for pathogens, which can remain pathogenic for prolonged time periods. Depending on the nature of the pathogen—such as type, pathogenicity, and weight—the length of time it is viable on environmental surfaces ranges from hours to days, weeks, and even months.3,5
ROUTES OF TRANSMISSION
Multiple modes of transmission are possible in a dental setting: direct contact (bodily fluids), indirect contact (contaminated objects or surfaces, and contact with mucous membranes/inhaling contaminated particles.3 Cross-transmission of microorganisms frequently occurs within the dental office; however, this does not usually lead to infections in patients or oral health professionals.2
Preventing the spread of potentially harmful microorganisms in the dental setting is possible by breaking the chain of transmission.2 To successfully break the chain of indirect disease transmission, surface disinfection protocols must be followed to the highest standard.6
According to a recent survey, 89 % of dental hygienists believe they can reduce the spread of disease by following infection control guidelines.7 Additionally, an increase in knowledge and adherence to infection guidelines has been noted among dentists post-pandemic.1
TYPES OF SURFACES
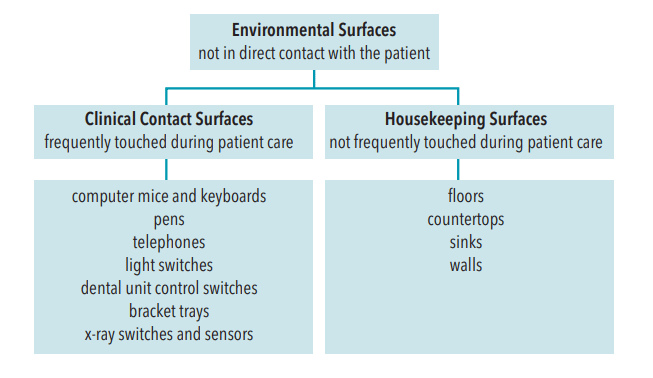
Surfaces not in direct contact with patients are called environmental surfaces.3 Those surfaces are further categorized as housekeeping or clinical contact surfaces based on the risk for contamination (Figure 1).3
Housekeeping surfaces are not frequently touched during patient care, such as floors, countertops, sinks, and walls.8
Clinical contact surfaces are frequently touched and pose a high risk of disease transmission through cross-contamination from gloved hands, aerosols, and contact with instruments.3,8 These include computer mice, keyboards, pens, telephones, light switches, dental unit control switches, bracket trays, Xray switches, sensors, and many more.3,8
The risk of infection from environmental surfaces is high, due to insufficient cleaning and disinfection practices.6 The actual level of viral contamination on surfaces is not well-researched. However, various detection methods are used to determine how many pathogens are present on surfaces, which surfaces are most susceptible to contamination, and what types of pathogens are of most concern for transmission.
One method is the use of luminol to detect the presence of blood particles on surfaces following dental procedures. A study using luminol revealed that 86.7% to 100% of surfaces, such as flooring below the patient’s headrest, instrument tray, operating light, dental chair armrests, cuspidor, and suction unit, were contaminated with blood.9
MAINTAIN ASEPSIS AND REDUCE CROSS-CONTAMINATION
The chances of contamination increase with the amount of time microorganisms are left undisturbed on the surface. Disease transmission is prevented by reducing the number of microorganisms on surfaces.3
To slow the spread of potentially pathogenic microorganisms on surfaces in the dental operatory, the chain of infection must be broken.2 The chain of infection includes the pathogen, a source, a mode of transmission, a port of entry, and a susceptible host. According to the United States Centers for Disease Control and Prevention (CDC) guidelines, this may be achieved through hand hygiene, use of personal protective equipment (PPE), engineering controls, and surface disinfection.8 The proper way to maintain surfaces in a dental operatory is by cleaning, disinfecting, and using surface barriers.3,10
The first step is to clean and disinfect contaminated surfaces. The CDC recommends the use of an Environmental Protection Agency (EPA)-registered intermediate-level disinfectant to treat contaminated surfaces at the beginning of the day, after each patient, and at the end of the day.8
Soiled surfaces and surfaces with visible contamination must be cleaned prior to disinfecting.11 A two-step cleaning and disinfecting approach is recommended, using the EPA-registered disinfectant.
EPA-registered disinfectants are available in multiple formulations, including ready-to-use or concentrated liquids.12 An ideal disinfectant is characterized by a short kill time. The labels of disinfectants will list the specific time required to deactivate each pathogen.6
Some EPA-registered disinfectants claim a 10-minute kill time. However, the effectiveness of these disinfectants against vegetative bacteria, yeasts, mycobacteria, and viruses is reported at exposure times of 30 seconds to 60 seconds.13
Studies show that ready-to-use disinfectants require a much shorter contact time to kill pathogens.12 Widely available, ready-to-use wipes are popular due to their ease of use. A study tested the efficacy of 10 brands of disinfecting wipes against S. aureus and P. aeruginosa. Results showed that the effectiveness of disinfecting wipes was correlated with the size of the area to clean: the larger the area, the less effective.14 Manufacturer instructions must be followed for each product to achieve maximum results.6
Barrier protection is another strategy in preventing cross-contamination.10 This approach includes the placement of water-resistant impervious barriers on frequently touched surfaces prior to contamination such as computer mice and keyboards, pens, hoses, handles, and switches. Barriers are either pre-cut pieces of thick film with mild adhesive to assure it remains on the surface, or plastic covers designed to fit specific equipment, cut to size, and secured by elastic.
Replacement of contaminated barriers after each patient encounter combined with disinfecting surfaces with proper disinfectant ensures compliance with infection control procedures. Additionally, thorough general cleaning and disinfection of clinical contact surfaces are performed regardless of barrier protection at the end of each workday.8
Bundling evidence-based practices is an approach designed to ensure infection control efficacy. The five most common practices are:6 create policies and procedures, appropriately select cleaning and disinfecting products, educate staff to include environmental services and patient equipment, monitor compliance and give feedback, and implement a “no touch” room. Two to three of the above recommendations should be followed simultaneously for the best outcomes.
CONCLUSION
Although the risk of disease transmission in the dental setting is relatively low, necessary steps should be taken to minimize the spread of infectious diseases.
References
- Cheng HC, Chang YJ, Liao SR, Siewchaisakul P, Chen SLS. The impact of COVID-19 on knowledge, attitude, and infection control behaviors among dentists. BMC Oral Health. 2021;21:584.
- Volgenant CMC, de Soet JJ. Cross-transmission in the dental office: does this make you ill? Curr Oral Health Rep. 2018;5:221–228.
- Schneiderman MT, Cartee DL. Surface disinfection. In: DePaola LG, Grant LE, eds. Infection Control in the Dental Office. New York: Springer International Publishing; 2020:169–191.
- Umar D, Basheer B, Husain A, Baroudi K, Ahamed F, Kumar A. Evaluation of bacterial contamination in a clinical environment. J Int Oral Health. 2015;7:53–55.
- Dhadse P, Oza R, Bhombe K. Disinfectants for dental setting in Coronavirus disease 2019 era. J Datta Meghe Inst Med. 2022;17(1):83–87.
- Rutala WA, Weber DJ. Best practices for disinfection of noncritical environmental surfaces and equipment in health care facilities: a bundle approach. Am J Infect Control. 2019;47S:A96–A105.
- Garland KV. A survey of United States dental hygienists’ knowledge, attitudes, and practices with infection control guidelines. J Dent Hyg. 2013;87:140–151.
- Kohn WG, Harte JA, Malvitz DM, et al. Guidelines for infection control in dental health-care settings—2003. MMWR Recomm Rep. 2003;52(RR-17):1–61.
- Al-Eid RA, Ramalingam S, Sundar C, Aldawsari M, Nooh N. Detection of visually imperceptible blood contamination in the oral surgical clinic using forensic luminol blood detection agent. J Int Soc Prev Community Dent. 2018;8:327–332.
- Upendran A, Gupta R, Geiger Z. Dental Infection Control. Treasure Island, Florida: StatPearls Publishing; 2022.
- United States Centers for Disease Control and Prevention. Summary of Infection Prevention Practices in Dental Settings. Available at: cdc.gov/oralhealth/infectioncontrol/summary-infection-prevention-practices/standard-precautions.html. Accessed February 15, 2023.
- Rutala WA, Weber DJ. Surface disinfection: treatment time (wipes and sprays) versus contact time (liquids). Infect Control Hosp Epidemiol. 2018;39:329–331.
- Rutala WA, Weber DJ. Disinfection, sterilization, and antisepsis: an overview. Am J Infect Control. 2019;47:A3–A9.
- West AM, Nkemngong CA, Voorn MG, et al. Surface area wiped, product type, and target strain impact bactericidal efficacy of ready-to-use disinfectant towelettes. Antimicrob Resist Infect Control. 2018;7:122.
From Dimensions of Dental Hygiene. March 2023; 21(3)12-14.

