
Avoid Implant Complications
Dental hygienists play an important role in recognizing, treating, and preventing peri-implant disease.
This course was published in the October 2012 issue and expires October 2015. The authors have no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Define peri-implant disease.
- Discuss the etiology of peri-implant disease.
- Explain standard protocol for detection, treatment, and maintenance of peri-implant disease.
Over the past 20 years, osseointegrated dental implants have revolutionized modern dentistry and long-term studies report implant therapy’s consistently high success rates.1 These “successes,” however, may include implants that also have existing pathology. The prevalence of this problem continues to grow as more data are collected on implant fixtures that have been in function for many years.1 The increase in the number of implants being placed, combined with the inherent risk of complications, underscores the need for dental hygienists to understand the incidence and etiology of implant breakdown, recognize active peri-implant disease, definitively treat early stage disease, appropriately refer patients, and prevent peri-implant disease through stringent surveillance and maintenance protocols.
PERI-IMPLANT DISEASE
Peri-implant disease is the presence of inflammation in the tissues surrounding the implant and is categorized into two states: peri-implant mucositis and peri-implantitis.2 Peri-implant mucositis is a reversible inflammatory process localized to the soft tissues surrounding a functioning implant (implant gingivitis).3 Peri-implantitis is similar but includes the loss of peri-implant bone (implant periodontitis).3 Peri-implant mucositis is more common than peri-implantitis.4 The primary contributing factor to peri-implant inflammatory disease is bacterial plaque. Research shows that peri-implant lesions are associated with Gram-negative anaerobic bacterial infiltrate within biofilm. The organisms isolated from these lesions are similar to those found in periodontal disease (eg, red complex and Aggregatibacter actinomycetemcomitans). The host inflammatory response to peri-implant lesions is similar to that observed in periodontitis and consists of plasma cells, macrophages, and T-lymphocytes.5,6 The same local or systemic factors that contribute to plaque accumulation and compromise the host response in natural teeth affect the onset and progression of disease around implants. Systemic diseases and medications that induce xerostomia can cause bacterial plaque accumulation and exacerbate disease.7 Systemic health problems and lifestyle habits that compromise the immunologic response to bacteria, such as diabetes and/or smoking, have also been associated with an increased risk of peri-implant disease. 8,9 Peri-implantitis is also more prevalent among patients with pre-existing periodontal diseases than in periodontally healthy individuals.10
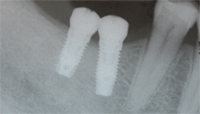
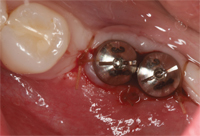
The position, surface characteristics, excessive restorative cement, and restorative design of the dental implant can all affect the development of peri-implant disease because they interfere with the patient’s ability to effectively clean the prosthesis. From a mesial-distal perspective, if implants are placed too close to each other or too close to a natural tooth, a poor embrasure form will result that can interfere with interproximal cleaning (Figure 1 and Figure 1A). If the implants are placed too far apart, a large cantilever will result that can be difficult to maintain (Figure 2 and Figure 2A).
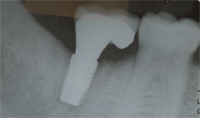
In apical and coronal areas, dentists will often place an implant well below the crest of bone in order to create enough “running room” for the tooth to properly emerge.11 Although this technique may be esthetic, it can create a deep implant sulcus that is difficult for the patient to access (Figure 3). If the implant is left too coronal, the implant platform or surface can be exposed. If the implant has a roughened texture and coated surface, this can provide an area that attracts bacterial plaque accumulation.12 From a buccal-lingual perspective, dentists often place an implant in a more palatal/lingual manner in order to avoid compromising the buccal plate and blood supply. If the entire axis of the implant is placed too far to the lingual/palatal, a large ridge lap can result that acts as a food trap. If the implant is placed too buccally, soft tissue dehiscence can occur—creating a welcoming environment for bacteria.
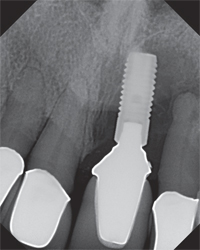
Excess cement below the restorative margin of an implant-supported prosthesis also invites peri-implant disease. In one study, excess dental cement was associated with signs of peri-implant disease in 81% of the cases. It also took anywhere from 4 months to more than 9 years after cementation for the signs of peri-implant disease to become evident, suggesting an insidious pattern of disease progression.13 Removal of cement is analogous to removal of subgingival calculus from a root surface. Once removed, resolution of this inflammation can occur if all other factors are controlled and bone loss has not been initiated.14
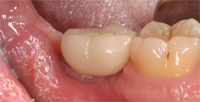
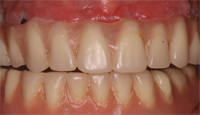
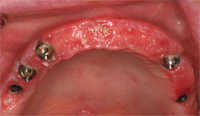
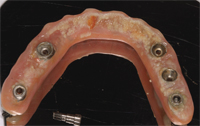
Finally, when placing a prosthesis on multiple implant units or restoring a full edentulous arch with an implant-supported porcelain fused to metal bridge, ceramoacrylic hybrid, or acrylic overdenture, the restoration must be cleanable. Unfortunately, esthetic compromises are often made that make performing adequate oral hygiene nearly impossible (Figure 4, Figure 4A, and Figure 4B).15 Large acrylic flanges or pink porcelain that mask hard and soft tissue defects can be extremely difficult to clean (Figure 5 and Figure 5A).
DETECTION
One of the most important roles dental hygienists can play in implant dentistry is the detection of peri-implant disease. The majority of implants suffering tissue breakdown in the peri-implant area are detected during maintenance visits.16 A typical implant suffering from peri-implant mucositis can be identified by the following: bleeding on probing/palpation, suppuration on probing/ palpation, rolled marginal tissue, change in color, change in texture, and movement in soft tissue, from adherent to loosely bound, and retractable on instrumentation (Figure 6 and Figure 6A). These factors, in addition to radiographic evidence of bone loss around the implant fixture, suggest progression to a peri-implantitis lesion. The exact amount of bone loss that defines peri-implantitis remains controversial.
Studies have used various markers for disease progression, such as exposed implant threads and millimeters of bone loss from the platform. These types of lesions initially affect the marginal part of the peri-implant tissues and the implant can remain in function for varying periods of time. Implant mobility is not an essential characteristic of peri-implantitis, but if left unabated, it may indicate complete loss of integration. As with natural teeth, early stage peri-implant mucositis lesions are reversible and easier to treat than late stage peri-implantitis lesions.
Early detection is crucial to implant survival. Dental hygienists can use a periodontal probe to detect the presence of peri-implant disease. Although early implant literature suggested that periodontal probing should not take place around implants due to the risk of attachment tearing, this antiquated theory is not supported by the literature.17 Periodontal probing is a prerequisite in the examination of peri-implant tissues.18
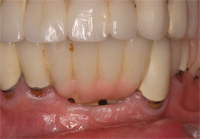
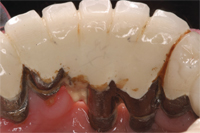
NONSURGICAL THERAPY
Nonsurgical treatments for peri-implant disease are similar to those used to treat gingivitis. The therapeutic goal is to reduce the bacterial load and promote healing of surrounding tissues. Unlike natural teeth, however, the grooved and roughened surfaces of implants are difficult to treat through mechanical means alone. Therefore, nonsurgical therapies include adjuncts such as antiseptics, antibiotics, and lasers. Research shows that nonsurgical therapy, such as scaling and root planing with the adjunctive use of local drug delivery, are effective in reversing bleeding on probing and pocketing around dental implants affected by peri-implant disease.19 In addition, systemic antibiotics with or without the use of a chlorhexidine mouthrinse show promise in reducing bleeding on probing and inflammation around implants among patients with peri-implant mucositis.20 A typical protocol for treating patients with peri-implant mucositis lesions is to prescribe amoxicillin 500 mg in combination with metronidazole 500 mg twice a day for 10 days. In addition, scaling and root planing should be performed using an implant-friendly ultrasonic insert/tip with chlorhexidine medicament and titanium hand scalers. Care must be taken to avoid scratching the implant components because this can lead to a roughened surface and further plaque attachment.21 Local drug delivery at the time of scaling and root planing is performed with 1 mg minocycline spheres.22 The patient is instructed to avoid using oral irrigators for 10 days and warned about the possibility of tissue recession once inflammation subsides. The patient is seen again in 4 weeks for clinical inspection and possible re-application of local drug delivery if the region is still bleeding on probing. This protocol works well to control peri-implant mucositis, however, nonsurgical therapy is not effective in treating peri-implantitis lesions.23
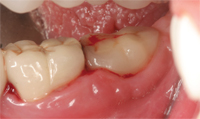
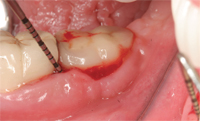
SURGICAL THERAPY
Patients with peri-implantitis must be referred to a specialist for surgical intervention. The long-term efficacy of surgical intervention with regenerative agents remains under investigation. Researchers are trying to identify the ideal method of treating the implant surface to remove surface contaminants, promote regeneration of lost or damaged tissues, and stimulate osseointegration. In a recent case series, 38 patients with 51 implants sites affected by peri-implantitis received surgical intervention and follow-up
over a 31?2 year to 71?2 year period.24 A surgical protocol was used that showed a significant gain in bone level ranging between 3.00 mm and 3.75 mm. Pocket depths were reduced by approximately 5 mm, soft tissue gain was enhanced by 1.0 mm to 1.3 mm, and bleeding on probing, as well as recession, were both reduced. These conclusions suggest that if proper surgical protocol is followed in combination with maintenance protocols, tissue around damaged implants can be repaired.
MAINTENANCE
Regardless of the treatment modality, the maintenance of proper self-care practices and supportive care at regular intervals is key to success. A consistent 3-month recare schedule is supported by research, which shows that patients on this schedule perform better than those seen at longer intervals. Patients with previous histories of periodontal disease should remain on a 3-month recare schedule until they demonstrate good self-care and/or absence of disease progression. The mantra remains if they lost their teeth to periodontal disease, they certainly can lose their implants to peri-implant disease.
A typical implant maintenance visit should include recording probing depths, noting bleeding on probing/suppuration, recognizing the presence of mobility, and recording the soft tissue texture and color. Radiographs should also be taken. After the clinical exam is completed, dental maintenance is performed with the proper armamentarium.
At the end of the visit, oral hygiene instruction is provided, which depends on the complexity of the implant restoration and the level of self-care demonstrated by the patient. Ultra-soft toothbrushes or mechanical toothbrushes should be recommended along with automated interproximal cleaning devices. If the patient still performs poorly with self-care, a separate visit to review and demonstrate effective self-care should be scheduled. The saying “an ounce of prevention is worth a pound of cure” holds true in implant dentistry and as the prevention experts, dental hygienists are key players in the fight for implant health and maintenance.
REFERENCES
- Berglundh T, Persson L, Klinge B. A systematicreview of the incidence of biological andtechnical complications in implant dentistryreported in prospective longitudinal studies of atleast 5 years. J Clin Periodontol. 2002;29(Suppl):197–212.
- Albrektsson T, Isidor F. Consensus report:implant therapy. In: Lang NP, Karring T, eds.Proceedings of the 1st European Workshop onPeriodontology. Berlin: Quintessence; 1994:365–369.
- Berglundh T, Lindhe J, Lang NP. Peri-impantmucositis and peri-implantitis. In: Lindhe J,Karring T, Lang NP, eds. Clinical Periodontologyand Implant Dentistry. 5th edition. Oxford, UK:Blackwell Munksgaard. 2007:529–538.
- Roos-Jansaker AM, Renvert S, Egelberg J.Treatment of peri-implant infections: a literaturereview. J Clin Periodontol. 2003;30:467–485.
- Tonetti M, Schmid J. Pathogenesis of implantfailures. Periodontol 2000. 1994;4:127–138.
- Berglundh T, Gislason O, Lekholm U, SennerbyL, Lindhe J. Histopathological observations onhuman periimplantitis lesions. J Clin Periodontol.2004;31:341–347.
- Ship JA, Pillemer SR, Baum BJ. Xerostomia andthe geriatric patient. J Am Geriatr Soc. 2002;50:535–543.
- Bain CA, Moy PK. The association between thefailure of dental implants and cigarette smoking.Int J Oral Maxillofac Implants. 1993:8:609–615.
- Lindquist LW, Carlsson GE, Jemt T. Associationbetween marginal bone loss aroundosseointegrated mandibular implants andsmoking habits: a 10-year follow-up study. J DentRes. 1997;76:1667–1674.
- Karoussis IK, Salvi GE, Heitz-Mayfield LJ,Brägger U, Hämmerle CH, Lang NP. Long-termimplant prognosis in patients with and without ahistory of chronic periodontitis: a 10-yearprospective cohort study of the ITI DentalImplant System. Clin Oral Implants Res.2003;14:329–339.
- Neale D, Chee WW. Development of implantsoft tissue emergence profile: a technique.J Prosthet Dent. 1994;71:364–368.
- Berglundh T, Gotfredsen K, Zitzmann NU,Lang NP, Lindhe J. Spontaneous progression ofligature induced peri-implantitis at implants withdifferent surface roughness: an experimentalstudy in dogs. Clin Oral Implants Res.2007;18:655–661.
- Wilson T, Jr. The positive relationship betweenexcess cement and peri-implant disease: aprospective clinical endoscopic study.J Periodontol. 2009;80:1388–1392.
- Froum SH. Supporting success: the role of thehygienist in implant complications. Dimensions ofDental Hygiene. 2012;10(2):19–22.
- Salierno C. The implant dilema. RDH.2012;32(6):22–25.
- Meffet RM. Maintenance and treatement ofthe ailing and failing implant. J Indiana DentAssoc. 1994;73:22–24
- Renvert S, Giovannoli JL. Peri-Implantitis.Hanover Park, Ill: Quintessence International;2012:251–252.
- Etter TH, Hakanson I, Lang NP, Trejo PM,Caffesse RG. Healing after standardized clinicalprobing of the periimplant soft tissue seal: ahistomorphometric study in dogs. Clin OralImplants Res. 2002;13:571–580.
- Renvert S, Roos-Jansåker AM, Claffey N. Nonsurgicaltreatment of peri-implant mucositis andperi-implantitis: a literature review. J ClinPeriodontol. 2008;35(Suppl):305–315.
- Mombelli A, Lang NP. Antimicrobial treatmentof peri-implant infections. Clin Oral Implants Res.1992;3:162–168.
- Meschenmoser A, d’Hoedt B, Meyle J, et al.Effects of various hygiene procedures on thesurface characteristics of titanium abutments.J Periodontol. 1996;67:229–235.
- Salvi GE, Persson GR, Heitz-Mayfield LJ, Frei M,Lang NP. Adjunctive local antibiotic therapy in thetreatment of peri-implantitis II: clinical andradiographic outcomes. Clin Oral Implants Res.2007;18:281–285.
- Leonhardt A, Dahlén G, Renvert S. Five-yearclinical, microbiological, and radiologicaloutcome following treatment of peri-implantitisin man. J Periodontol. 2003;74:1415–1422.
- Froum SJ, Froum SH, Rosen PS. Successfulmanagement of peri-implantitis with aregenerative approach: a consecutive series of 51treated implants with 3- to 7.5-year follow-up. IntJ Periodontics Restorative Dent. 2012;32:11–20.
From Dimensions of Dental Hygiene. October 2012; 10(10): 70-73.



