When Cancer Treatment Affects the Mouth
Dental hygienists play a critical role in preventing, identifying, and managing oral mucositis.
This course was published in the May/June 2026 issue and expires June 2029. The authors have no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
AGD Subject Code: 730
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Define oral mucositis (OM).
- Identify the etiologies of OM.
- Discuss the role of the dental hygienist in managing patients with OM.
Dental hygienists are uniquely positioned to identify, assess, and contribute to the management of complex oral manifestations of systemic diseases and their treatments. Among the most debilitating complications encountered in oncology patients is oral mucositis (OM), an inflammatory response of the oral mucosa marked by ulcerations caused by cytotoxic cancer therapies. OM profoundly impacts quality of life, often leading to severe pain, compromised nutrition, increased risk of systemic infection, and even interruption or modification of cancer treatment.
While medical oncologists, radiation oncologists, and oncology nurses collaborate to manage the primary disease and its systemic effects, cancer therapies can also significantly affect the oral cavity and require dedicated management. By understanding the intricate pathophysiology of OM, its clinical presentations, and evidence-based management strategies, dental hygienists can help alleviate patient suffering, improve treatment adherence, and contribute to better overall outcomes.
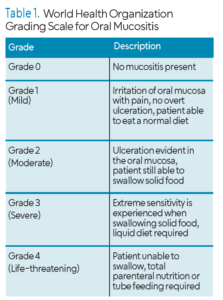
 OM can result in erythema, edema, and ulcerative lesions of the oral mucosa and may range from mild to severe including small ulcerations and those large enough to impair oral function (Table 1). For patients undergoing chemotherapy, approximately 40% will develop OM at some stage during treatment, typically 5 to 7 days after starting medication. For those receiving both chemo- and radiation therapy, this percentage increases to approximately 90%.1-3
OM can result in erythema, edema, and ulcerative lesions of the oral mucosa and may range from mild to severe including small ulcerations and those large enough to impair oral function (Table 1). For patients undergoing chemotherapy, approximately 40% will develop OM at some stage during treatment, typically 5 to 7 days after starting medication. For those receiving both chemo- and radiation therapy, this percentage increases to approximately 90%.1-3
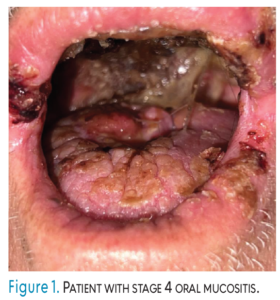
OM is a complex biological process of cellular injury, inflammation, and tissue breakdown (Figure 1). This intricate process is typically divided into five phases (Table 2), regardless of whether the etiology is chemotherapy or radiation.1-5
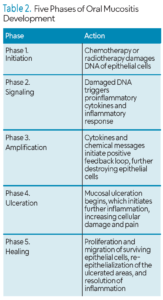 Phase 1. The initiation phase begins immediately upon exposure to chemo- or radiation therapies. Cancer treatments, which are designed to target rapidly dividing cancer cells, also damage rapidly proliferating healthy cells, including the basal epithelial cells of the oral mucosa.
Phase 1. The initiation phase begins immediately upon exposure to chemo- or radiation therapies. Cancer treatments, which are designed to target rapidly dividing cancer cells, also damage rapidly proliferating healthy cells, including the basal epithelial cells of the oral mucosa.
Phase 2. The signaling phase occurs after the cellular damage is initiated and activates several molecular pathways. Signaling molecules amplify the initial damage leading to further cell death and an escalating inflammatory response. The subepithelial connective tissue and vasculature are impacted in this phase, contributing to the overall tissue injury.
Phase 3. In the amplification phase, pro-inflammatory cytokines released in the signaling phase continue to recruit inflammatory cells and perpetuate the cycle of tissue damage. This leads to a further decrease in epithelial cell proliferation and a thinning of the oral mucosal lining. The delicate balance between cell death and cell renewal is severely disrupted, making the mucosa increasingly vulnerable.
Phase 4. During the ulceration phase, symptoms present clinically with the breakdown of the oral epithelial barrier. As the basal epithelial cells are destroyed and their regenerative capacity is compromised, the overlying mucosa thins and eventually sloughs, exposing the underlying connective tissue. This leads to the formation of painful, erythematous, and often pseudomembranous ulcers. The loss of mucosal integrity provides a direct pathway for oral microorganisms to enter the compromised tissues, thus significantly increasing the risk of local and systemic infections. This phase typically peaks around 7-14 days after the start of chemotherapy, or 2-3 weeks into radiation therapy.1-5
Phase 5. The healing phase begins as the cancer therapy is discontinued, or the mucosal cells have had a chance to recover. This phase involves the proliferation and migration of surviving epithelial cells, re-epithelialization of the ulcerated areas, and resolution of inflammation. The duration of healing can vary widely, from a few weeks to several months, depending on the severity of the mucositis and the patient’s overall health and immune status.
Impact of Oral Mucositis on the Patient
The effects of oral mucositis extend far beyond the oral cavity. Arguably, the most dominant symptom is pain. Interference with virtually all oral functions occurs as the palate, tongue, cheeks, floor of mouth, lips, and pharynx are damaged. Pain ranging from mild discomfort to debilitating pain can require strong opioid analgesics.
Chemotherapy and radiation often cause dysphagia, odynophagia, and dysgeusia. This, combined with pain, severely reduces appetite.5 Inadequate oral intake results in weight loss, nutritional deficiencies, and dehydration. Malnutrition can further impair immune function and delay wound healing, initiating a pattern of deterioration. In severe cases, patients may require enteral (feeding tube) or parenteral (intravenous) nutrition.6
The affected oral mucosa now functions as an entry point for opportunistic pathogens. Patients undergoing cancer treatment are often profoundly immunocompromised (neutropenic) because of therapy.7 This combination dramatically increases the risk of local oral infections and, more dangerously, systemic infections (sepsis). Oral sepsis originating from mucositis-related ulceration can be life-threatening and may require hospitalization.6,8
OM also negatively affects quality of life, limiting patients’ ability to perform activities of daily living and hindering their emotional well-being and social interactions.9 Speech may be impaired, affecting the patient’s communication. Patients may experience anxiety, depression, and frustration. This decreased quality of life may reduce the desire to continue with prescribed therapy.10
Severe OM is a common reason for delaying or reducing doses of cancer therapy.1,6 While these modifications aim to alleviate suffering and prevent life-threatening complications, they can compromise the efficacy of the cancer treatment. Effective OM management to support treatment adherence is critical. The management of OM, depending on the severity, may include hospitalizations, increased clinic visits, medications, and nutritional support. All of which can impose a significant economic burden on patients and healthcare systems.6,8,11
Etiology of Oral Mucositis
While the underlying mechanism of OM involves damage to rapidly dividing cells, specific cancer treatments and patient-related factors significantly influence the incidence and severity of OM. Certain chemotherapeutic agents are more cytotoxic than others. Antimetabolites and certain alkylating agents are particularly notorious for inducing severe OM.4,12 High-dose chemotherapy regimens, especially those used in preparation for hematopoietic stem cell transplantation (HSCT), are associated with a remarkably high incidence and severity of OM (70% to 100%).6,7 The use of multiple chemotherapeutic agents concurrently, or chemotherapy combined with radiation therapy, significantly increases the risk and severity of OM.6
Radiation to the head and neck region, particularly when the oral cavity is within the treatment field, is a primary cause of OM. The severity is related to the volume of oral mucosa irradiated and the total cumulative dose.6 Hypofractionated regimens (larger doses per fraction) can sometimes lead to more acute, severe mucositis compared to conventional fractionation.13
Patients undergoing HSCT receive extremely high doses of chemotherapy (myeloablative conditioning regimens), often followed by total body irradiation. This combination invariably leads to severe, often Grade 3 or 4, oral mucositis, which is frequently cited by patients as the most distressing side effect of their transplant.6
Proactive, Preventive, and Palliative
The dental hygienist is uniquely positioned to impact the comprehensive care of oncology patients at risk for or experiencing OM. Expertise in oral health assessment, preventive strategies, patient education, and supportive care make dental hygienists invaluable members of the interdisciplinary team. The role encompasses proactive intervention, vigilant monitoring, and empathetic palliative care. Optimizing oral health before the initiation of cancer therapy is paramount in reducing the incidence and severity of OM.
Pretreatment assessment and intervention are arguably the most critical phases. A detailed history of the patient’s cancer diagnosis, planned treatment regimen (chemotherapy, radiation, HSCT), anticipated side effects, current medications (including over-the-counter and herbal supplements), allergies, and any history of previous oral complications should be obtained. Clinically, a thorough examination of the head, neck, lymph nodes, lips, buccal mucosa, labial mucosa, gingiva, tongue (dorsal, ventral, lateral borders), floor of the mouth, hard and soft palate, and oropharynx should be performed. Documentation of any existing lesions, infections, or abnormalities is necessary. Intraoral photographs are an excellent form of documentation for this step.
Next, the patient’s periodontal status should be evaluated, including probing depths, bleeding on probing, clinical attachment loss, and presence of calculus. Active periodontal disease is a significant risk factor for OM. Prescribed periodontal therapy should be performed to reduce inflammation and bacterial load. Review of recent radiographs to identify periapical lesions, impacted teeth, or other bony pathologies that may pose a risk during immunosuppression.
An examination of the hard tissue is important to identify any existing carious lesions, especially those approaching the pulp or causing sensitivity. Restoration of carious lesions should be completed, and extraction of nonrestorable teeth considered, especially those with periapical lesions or severely periodontally involved teeth. These procedures need to be completed prior to head and neck radiation or bone marrow transplant during which the risk of osteoradionecrosis or osteonecrosis of the jaw is high.
Extractions should ideally be completed with sufficient time (7 to 14 days for healing) before the start of cancer therapy.14 If a patient uses a removable prosthesis of any kind, the fit and condition should be assessed to identify areas of irritation or potential trauma. Ill-fitting appliances can exacerbate OM, so sharp cusps should be smoothed, adjustments made, or temporary denture discontinuation may be recommended. While daily oral hygiene is crucial, routine preventive visits should also be maintained. Prophylactic steps taken with an oral healthcare provider can reduce the risk of mucositis by more than 25%.15
When speaking to the patient, define OM, why it occurs, and what symptoms it may cause. Use simple language and visual aids if available. Stress the critical importance of maintaining meticulous oral hygiene before, during, and after treatment.
Recommend an extra-soft toothbrush, emphasizing gentle, thorough brushing at least two to four times daily, especially after meals and before bed. Advise the patient to replace the toothbrush frequently (every 3-4 weeks, or after an infection). Toothbrush storage is also important. Patients may become immunocompromised; an improperly stored toothbrush may introduce new risks. Recommend storing the toothbrush in an area away from family toothbrushes that will allow air flow.16
Suggest a nonabrasive, fluoride-containing toothpaste with a mild flavor (avoid strong mints or whitening agents that can irritate). Encourage daily flossing but advise patients to stop if it causes excessive pain or bleeding that doesn’t subside within a couple of minutes. If patients were not consistent flossers prior to their diagnosis, now may not be the time to start. Instruct them to consult with their oncology team if platelet counts are low, as flossing may be contraindicated.
Nonirritating mouthrinses may be recommended. A sodium bicarbonate rinse is ideal. The standard recipe is 1 teaspoon of salt and 1 teaspoon of baking soda mixed into 4 cups of warm water. Advise rinsing frequently (four to six times daily, especially after meals and before bed, and even hourly if symptoms are severe). Patients should make their sodium bicarbonate rinse daily. Emphasize swishing for at least 30 seconds and spitting out. Patients with OM should avoid alcohol-containing hydrogen peroxide, chlorhexidine (unless specifically prescribed by the oncology team for a targeted infection), and highly acidic or irritating commercial mouthrinses. The Multinational Association of Supportive Care in Cancer recommends benzydamine mouthrinse for the prevention of OM. Note: This rinse is only available by prescription in the United States through a compounding pharmacy.2
Advise regular application of water-soluble or lanolin-based lip lubricants to prevent dryness and cracking.
Patients who wear dentures should remove them for at least 8 hours daily ( overnight) and clean them thoroughly after each meal. If mucositis develops, advise wearing dentures only during meals or discontinuing them altogether if they cause irritation or pain.
Guidance on appropriate dietary choices during mucositis onset should be provided. Recommend soft, bland, moist foods that are easy to chew and swallow, such as mashed potatoes, scrambled eggs, yogurt, smoothies, pureed fruits, and cooked cereals. Encourage adding gravies or sauces to foods to make them easier to consume.
Patients should be advised against eating acidic, spicy, hot, and crunchy foods as well as consuming carbonated beverages. Suggest lukewarm or cool foods and beverages, as extreme temperatures can increase pain. Encourage patients to take frequent sips of water throughout the day to keep the mouth moist and aid in cleansing.
Patients should understand when it is time to reach out to their oncology team; for instance, in light of worsening pain, inability to eat or drink, fever, signs of infection, and bleeding.
Emerging Therapies
While supportive care is still the cornerstone of OM management, research continues to explore new preventive and therapeutic interventions. Palifermin (recombinant human keratinocyte growth factor-1) is the only US Food and Drug Administration-approved medication for reducing the incidence and duration of severe OM in patients undergoing HSCT for hematologic malignancies. It works by promoting the growth and repair of epithelial cells.2
Growing evidence supports the use of lower-level laser therapy (LLLT) for the prevention and treatment of OM, particularly in head and neck cancer patients receiving radiation and in HSCT patients. LLLT is thought to reduce inflammation, promote cell proliferation and provide analgesic effects.2,8
Used during chemotherapy, cryotherapy can reduce the severity of OM due to vasoconstriction reducing the amount of drug delivered to the oral mucosa. Continued research is exploring the precise timing and duration for maximal benefit.8 Understanding the role of the oral microbiome in the pathogenesis and resolution of OM may lead to novel probiotic or prebiotic interventions.
Conclusion
OM demands a comprehensive and interdisciplinary approach to care. By thoroughly understanding the etiology, pathophysiology, impact, and evidence-based management of OM, dental hygienists can provide invaluable pretreatment preparation, vigilant monitoring during therapy, and empathetic palliative care.
References
- Pulito C, Cristaudo A, Porta CL, et al. Oral mucositis: the hidden side of cancer therapy. J Exp Clin Cancer Res. 2020;39:210.
- Blakaj A, Bonomi M, Gamez ME, Blakaj DM. Oral mucositis in head and neck cancer: Evidence-based management and review of clinical trial data. Oral Oncol. 2019;95:29-34.
- Wong HM. Oral complications and management strategies for patients undergoing cancer therapy. ScientificWorldJournal. 2014;2014:581795.
- Basile D, Di Nardo P, Corvaja C, et al. Mucosal injury during anti-cancer treatment: from pathobiology to bedside. Cancers. 2019;11:11060857.
- Kutuk T, Atak E, Villa A, Kalman NS, Kaiser A. Interdisciplinary collaboration in head and neck cancer care: optimizing oral health management for patients undergoing radiation therapy. Curr Oncol. 2024;31:2092-2108.
- Gugnacki P, Sierko E. Is there an interplay between oral microbiome, head and neck carcinoma and radiation-induced oral mucositis? Cancers (Basel). 2021;13:5902.
- Raber-Durlacher JE, Elad S, Barasch A. Oral mucositis. Oral Oncol. 2010;46:452-456.
- Lalla RV, Saunders DP, Peterson DE. Chemotherapy or radiation-induced oral mucositis. Dent Clin North Am. 2014;58:341-349.
- Al-Rudayni AHM, Gopinath D, Maharajan MK, Menon RK. Impact of oral mucositis on quality of life in patients undergoing oncological treatment: A systematic review. Transl Cancer Res. 2020;9:3126-3134.
- Jung YS, Park EY, Sohn HO. Oral health status and oral health-related quality of life according to presence or absence of mucositis in head and neck cancer patients. J Cancer Prev. 2019;24:43-47.
- Tang X, Li W, Zhong Q, Wan L. Effects of omega-3 fatty acids on oral mucositis induced by anticancer therapy: a meta-analysis. Nutr Cancer. 2025;77:600-609.
- Liu M, An R, Wu Z, Dai L, Zeng Q, Chen W. The trajectory of oral mucositis in head and neck cancer patients undergoing radiotherapy and its influencing factors. Ear Nose Throat J. 2025;104:NP257-NP269.
- Nahum AE. The radiobiology of hypofractionation. Clin Oncol. 2015;27:260-269.
- Strobl J, Ballicas N, Wachter B, et al. Dental health, conditioning and oral mucositis in allogeneic hematopoietic stem cell transplantation: a single-center study. Cytotherapy. 2025;27:1130-1136.
- Brown TJ, Gupta A. Management of cancer therapy-associated oral mucositis. JCO Oncol Pract. 2020;16:103-109.
- Dayoub MB, Rusilko D, Gross A. Microbial contamination of toothbrushes. J Dent Res. 1977;56:706.
From Dimensions of Dental Hygiene. May/June 2026; 24(3):32-35