Minimize Risks of Drug Interactions
The polypharmaceutical nature of modern health care, with an estimated 29% of Americans taking five or more medications on a regular basis, requires oral health professionals to have a thorough understanding of possible drug interactions.

The polypharmaceutical nature of modern health care, with an estimated 29% of Americans taking five or more medications on a regular basis, requires oral health professionals to have a thorough understanding of possible drug interactions. Drugs can interact with each other, as well as with herbal supplements, vitamin and mineral supplements, and even various foods. Understanding the many complex, underlying mechanisms of these interactions and resulting intricate drug relationships is crucial in preventing adverse drug reactions and potentially life-threatening emergencies. While the number and therapeutic categories of drugs prescribed in the dental setting is limited compared to medicine, a thorough review of the patient’s drug history and evaluation for potential adverse reactions—or reduced effect as a result of drug interaction—is required.
Photo Credit: Fahroni / iStock / Getty Images Plus
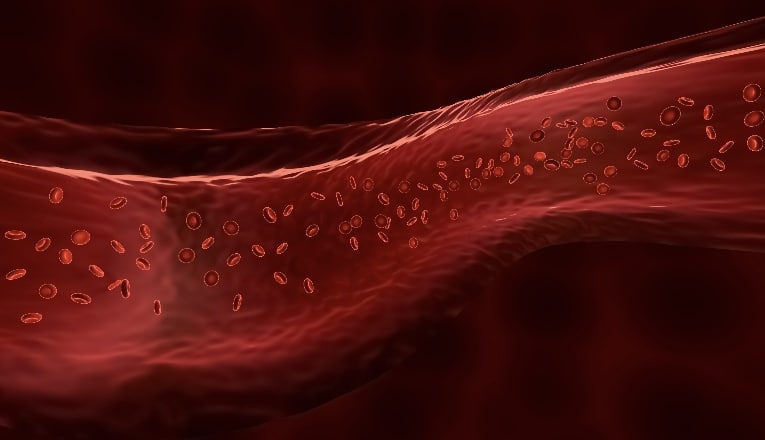
Drug Absorption
Once absorbed into the bloodstream, many drugs are distributed through the body either as available free drug or bound reversibly to plasma proteins, mainly albumin. The percentage bound to protein, and therefore not available at the intended site of action, depends on the drug concentration and its affinity for that plasma protein. Drug interactions may occur when protein binding sites, which are limited in number, are saturated to capacity. When two or more drugs compete for the same binding sites, with one drug displacing another, it increases its availability as a free active drug. With many drugs, a small increased percentage may have no clinically significant effect. However, displacement is problematic when a highly bound drug with a narrow therapeutic index, such as warfarin, is displaced by another drug, such as aspirin, potentiating warfarin’s potent anticoagulant effects. Similar interaction at protein-binding sites occurs with warfarin and most nonsteroidal anti-inflammatory drugs (NSAIDs). Significant increased bleeding risk warrants careful review of the international normalized ratio values, as well as physician consultation when NSAID use is unavoidable.
Photo Credit: libre de droit / iStock / Getty Images Plus
Different Patients, Different Responses
Variability in drug response is widespread and multifactorial, as estimates suggest most major drugs are effective in only 25% to 60% of the population. This irregularity encompasses environmental and genetic factors, including variations in the cytochrome P-450 enzymes (CYPs) involved in drug metabolism in the liver and small intestine. The role of this family of enzymes in the metabolism of drugs and endogenous substances—and their influence on drug effectiveness and toxicity—has been well established.
Photo Credit: Rasi Bhadramani / iStock / Getty Images Plus

Toxic Interactions
Because multiple drugs, as well as vitamins, herbal supplements, and foods, can be metabolized by the same enzyme, interactions, including an increase or decrease in drug concentration, can occur, making these drugs potentially toxic or ineffective. Various substances can function as inducers or inhibitors of CYPs. When a particular CYP is induced by drug A, for example, more CYPs are produced in response, and more enzymes can metabolize their other substrates faster. This can result in more efficient inactivation and therefore lower plasma concentration and availability of drug B that is metabolized by the same CYP at its site of action. This will render drug B ineffective, and if the two drugs have to be taken together, as is often the case, a dose adjustment of drug B may be required to maintain its effectiveness. Conversely, if drug A is an inhibitor of a CYP that also metabolizes drug B, it will slow metabolism of drug B, possibly raising its concentration to toxic levels.
Photo Credit: carlosgaw / E+
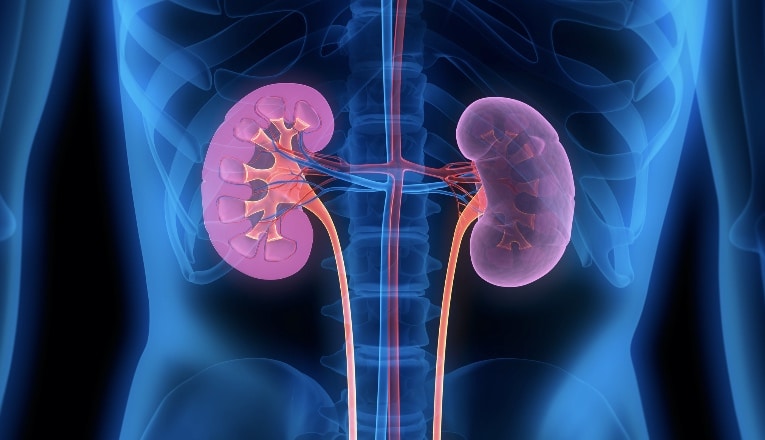
Drug Elimination
Drugs are most commonly excreted through the kidneys either as a free drug or metabolites into the urine and cleared from the body at a rate proportional to the blood concentration. Impaired renal function due to kidney disease and the normal aging process is associated with reduced drug clearance. More recently, decreased activity of gastrointestinal enzymes and renal transporters has been linked to chronic and acute kidney disease. This also contributes to prolonged drug elimination and increased risk of toxicity and drug-drug interactions, as multiple drugs may compete for the same excretion sites. When prescribing medications, the clinical implications of renal disease or insufficiency require careful evaluation of potential drug interactions. Oral health professionals should also consider reducing dosages or increasing dosing intervals due to prolonged clearance rates. Therefore, agents with a long half-life, such as tetracyclines, should be avoided in patients with impaired or reduced kidney function, including older adults.
Photo Credit: peterschreiber.media / iStock / Getty Images Plus

Pharmacodynamic Drug Actions
Pharmacodynamic drug actions occur at extra- and intracellular receptor sites of target tissues and nontarget tissues, exerting varying degrees of expected and unexpected effects. This may include the interaction of two or more drugs competing for the same receptor sites, producing various outcomes. Rather than affecting drug concentration (as with pharmacokinetic interactions), the net effect of the agent(s) on physiological functions may be enhanced or reduced by competitive binding or blocking of receptor sites. Although the ability of drugs to increase or decrease each other’s effects most often leads to adverse outcomes, it can also be beneficial and used therapeutically. An example of a negative outcome is the well-known interaction of alcohol (ethanol) with benzodiazepines (diazepam, chlordiazepoxide) and barbiturates (phenobarbital), which all bind to their respective sites on γ-aminobutyric acid (GABA) inhibitory GABAA receptors, enhancing GABAergic inhibition and leading to potentially dangerous central nervous system depression. Conversely, competitive binding of an opioid antagonist naloxone to the μ and κ opiate receptors reverses the effects of an opioid overdose; this is an example of beneficial pharmacodynamic antagonism.

