 PHOTOBAC / ISTOCK / THINKSTOCK
PHOTOBAC / ISTOCK / THINKSTOCK
Trends in the Prevention of Peri-Implant Diseases
Dental professionals who are well versed in the etiologies and risk factors for peri-implantitis can help prevent implant failure.
This course was published in the May 2016 issue and expires May 31, 2019. The authors have no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Identify the etiologies and risk factors for peri-implant diseases.
- Explain the factors involved in the diagnosis of peri-implant diseases.
- List the components of the cumulative interceptive supportive therapy protocol.
- Discuss the recommendations made by the American College of Prosthodontists regarding implant maintenance.
INTRODUCTION
As the number of patients with dental implants increases and dental implant therapy assumes a greater role in dental practice, clinical investigations are focusing on the prevention and management of the diseases that affect successfully osseointegrated implants. There is considerable evidence demonstrating that patients with implants will need lifelong professional recare regimens tailored to their individual needs. These regimens should be based on patient history, susceptibility to periodontitis and ability to control local factors at home. As there is minimal evidence related to recare regimens, this article includes a discussion of the American College of Prosthodontists Clinical Practice Guidelines for Recall and Maintenance of Patients With Tooth-Borne and Implant-Borne Dental Restorations, published in January 2016, which provide clinicians with suggested protocols to help improve clinical outcomes for implant patients. The Colgate-Palmolive Company is delighted to have provided an unrestricted educational grant to support “Trends in the Prevention of Peri-Implant Diseases” in collaboration with the American Academy of Periodontology.
—Matilde Hernandez, DDS, MS, MBA
Scientific Affairs Manager
Professional Oral Care
Colgate Oral Pharmaceuticals
FROM THE AMERICAN ACADEMY OF PERIODONTOLOGY
Often used to successfully manage a number of clinical issues, dental implants have proven to be a viable solution for patients and practitioners alike. Yet, despite implant therapy’s popularity and promise, it is not immune from complications associated with improper treatment planning, surgical and prosthetic execution, material failure, and lack of maintenance. Peri-implant disease, which is often characterized by an inflammatory reaction in tissues surrounding an implant, has long been a focus of periodontists—surgical specialists trained in the placement and maintenance of implants.
In this article, educator Akshay Kumarswamy, BDS, MS, highlights risk factors, diagnosis, and management therapies for peri-implant disease and the important role that dental hygienists play in the success of a implant therapy. The American Academy of Periodontology, which released a special report on this subject in 2013, is proud to work with Dimensions of Dental Hygiene and Colgate-Palmolive to bring you information that can aid in the diagnosis and treatment of implant-related disease.
—Wayne Aldredge, DMD
President, American Academy of Periodontology
Inflammation of the tissues or bone surrounding dental implants caused by plaque biofilm is broadly referred to as peri-implant disease.1 Inflammation limited to the peri-implant gingiva or mucosa is called peri-implant mucositis, while peri-implantitis refers to inflammation involving nearby bone. The terms “ailing” (mucositis), “failing” (peri-implantitis), and “failed” (clinically mobile implant) are used to describe the different stages of peri-implant diseases.2,3
The prevalence of peri-implantitis ranges from 6% to 56% in various populations.4,5 This wide range is likely due to a lack of standardized criteria for diagnosing these conditions. In order to reduce the prevalence of peri-implant diseases, dental professionals must assess patients’ risk profiles and educate them about proper implant maintenance before initiating implant therapy.
The formation of plaque biofilm is considered the primary etiological factor in peri-implant disease initiation, and it is essential for the development of typical peri-implant infections.6,7 Peri-implant mucositis is analogous to gingivitis around natural teeth, while peri-implantitis is similar to periodontitis. The host immune-inflammatory response to bacteria on an implant surface is similar to the response to plaque adhering to teeth.8 Accordingly, Gram-negative bacteria have been consistently associated with both periodontal and peri-implant lesions.6 Based on these collective findings, the frequent removal of plaque biofilm is a key objective when treating peri-implant diseases.
The Sixth European Workshop on Periodontology published a consensus report on peri-implant diseases in 2008.9 In 2013, the American Academy of Periodontology (AAP) published a consensus report on the purported risk factors for peri-implant diseases and evidence quality.10 Both acknowledged that strong evidence supports four risk factors for peri-implant diseases: history of periodontitis; smoking or other tobacco use; poor plaque control; and diabetes mellitus. The AAP report also identified residual prosthetic cement as a factor in peri-implant diseases. Occlusal disharmonies were deemed controversial because of inconsistent evidence.
Peri-implantitis is found more often in patients with a history of periodontitis. Systematic reviews have concluded that implants placed in patients with periodontitis have a higher incidence of biological complications and lower survival rates than those placed in periodontally healthy patients.11,12 Implant therapy success rates over 5 years or 10 years may decrease to as low as 60% in patients with periodontitis.13,14
Smoking is a major risk factor for peri-implantitis and implant failure.14 A dose-dependent effect also seems present. The frequency of implant complications is reported to be higher among those who smoke more than 10 cigarettes a day for more than 10 years compared with those who smoke less.15 The benefits of implant therapy should be used as a motivation for patients to quit smoking.
Oral hygiene expectations should be clear before initiating implant treatment. Prostheses designs can also impede good plaque control and oral hygiene.16 Unlike cement-retained prostheses, screw-retained restorations can be removed in order to facilitate professional maintenance. Regardless of how a prosthesis is retained, however, the dental team must educate patients on proper plaque control and establish a regular professional maintenance schedule.
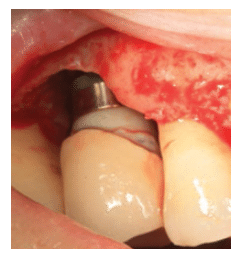
Residual cement impinging on the peri-implant tissues and concurrent inflammation of the proximate mucosa may affect implant therapy success (Figure 1). A case-control study found that in the majority of cases, excess dental cement was associated with signs of peri-implant diseases.17 Clinical signs of peri-implant diseases were eliminated in 74% of the cases after removal of excess cement. Residual cement may be left around an implant because removing subgingival cement in healthy peri-implant mucosa—which is firm and tightly adapted around the prosthesis—is difficult and detecting cement on radiographs can be challenging.18 Cement may act like an overhanging restoration on a natural tooth, which limits a patient’s ability to keep the site plaque-free. Although screw-retained restorations are the only way to completely eliminate the risk of residual cement being left during implant placement, other techniques may minimize residual cement extrusion such as the use of a lingual set screw, Teflon tape, abutment analog to flush out excess cement, and a rubber dam at the time of cementation.19
There is conflicting evidence that diabetes is associated with peri-implant diÂseases. One study involving 1,140 patients with diabetes reported a 10-year implant survival rate of 68.75%.20 Accordingly, diabetes was associated with implant failure with a relative risk ratio of 2.75 compared with patients without diabetes.20 Another review of nine cohort studies concluded that diabetes is not an absolute contraindication for implant placement if there is good glycemic control.21 A more recent systematic review found no statistically significant differences in implant failure rates between patients with and without diabetes.22 However, there is a strong association between diabetes and periodontal health.23 Thus, clinicians should thoroughly explain the potential risks of implant complications, especially to patients whose diabetes is poorly controlled.
Occlusal disharmonies and excess occlusal loading have been suggested as risk factors for implant structural failures. Research performed on animals, however, has shown no clear cause-and-effect relationships between occlusal loading and implant failure.24,25 The exception is one study on monÂkeys, in which extreme ocÂclusal forces caused implant failures.26 As such, the off-axis loading of implants may lead to imÂplant failures, but its effect on peri-implantitis risk is not yet understood.
DIAGNOSIS
Early diagnosis of peri-imÂplant diseases is important to ensure that the pathological processes can be arrested before the stability of the implant is compromised. After completion of dental implant treatment, the patient must be put on a tightly monitored maintenance schedule. A 5-year study found that without professional maintenance, patients with pre-existing peri-implant mucositis had a 44% higher incidence of peri-implantitis.27 Therefore, dental hygienists who perform these maintenance treatments play important roles in detecting the initial signs of peri-implant diseases.
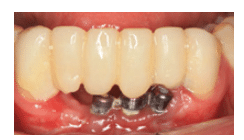
Gentle probing around dental implants should be performed at every maintenance appointment. The presence of bleeding on probing indicates inflammation in the peri-implant tissues. The consistent absence of bleeding on probing at consecutive maintenance appointments is an indicator of periodontal stability.28 In the absence of evidence to the contrary, it is reasonable to assume the same for peri-implant tissues. Probing should be performed followed by observation for bleeding or suppuration.29 Bleeding on probing typically indicates chronically inflamed tissues; suppuration is usually indicative of an acute infection that can be superimposed on chronic inflammation. Visual signs of inflammation such as erythema (redness) and edema help corroborate a diagnosis of peri-implant mucositis (Figure 2).
Deep probing depths and loss of clinical attachment levels often characterize progressing periodontitis. Similar depths around implants can be misleading, as the probing depths around healthy implants are typically greater compared with natural teeth.30 While probing natural healthy teeth, the probe tip ends closer to the junctional epithelium. In the presence of inflammation, the periodontal probe tip extends deeper into the attachment apparatus and may not stop until it contacts subjacent connective tissues. On the other hand, when probing healthy peri-implant mucosa, the probe may extend close to underlying bone, thereby producing higher probing depths than those typically seen around healthy teeth.30 Adding to this apparent discrepancy is the fact that implants are almost always placed at the level of the bone, thereby creating deeper sulcus depths. Even with tissue-level implants, the probe goes past the coronal aspect of the implant. Thus, deeper-than-expected probing depths around dental implants may not provide much information. Bleeding on probing, visual signs of inflammation, and radiographic bone loss when combined with peri-implant probing may offer better insights into the condition of the tissues surrounding an implant.
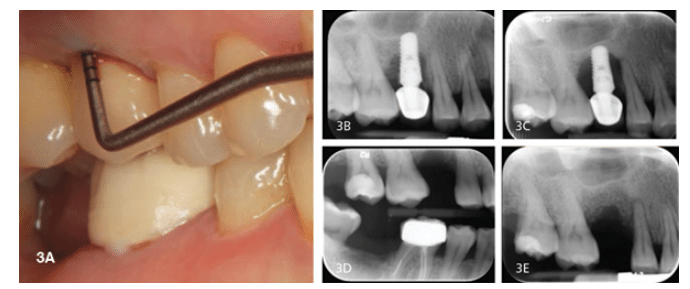
Radiographic evaluation of dental implants is crucial for monitoring long-term health. Failure to fully seat an implant-supported prosthesis can contribute to accumulation of dental plaque underneath which may, in turn, facilitate peri-implant diseases.31 Radiographs also serve as a baseline with which future X-rays can be compared for evidence of bone loss (Figure 3A to Figure 3E). Periapical radiographs are most useful when taken with the X-ray beam perpendicular to the implant fixture. A quality radiograph allows clear visualization of implant threads and adjacent bone, making it easier to observe initial bone levels and to monitor changes over time. Because bone loss around implants tends to be circumferential and subtle changes may not be evident on radiographs, clinical examinations must accompany radiographic examinations. Cone-beam computed tomography (CBCT) can provide three-dimensional diagnostic information on suspected peri-implant lesions; however, the aspect of burnouts due to adjacent metal restorations can render these images useless. The expense and additional radiation exposure of using CBCT should be considered.
Following placement and healing, dental implants are osseointegrated with direct bone to implant contact along the implant surface. Unlike teeth, which have a cushioning periodontal ligament and may become visibly mobile due to off-axis loading, implants may appear immobile, even when the bone level has diminished to the apical third of the implant fixture. Therefore, recognizing progressive bone loss can lead to a diagnosis of a “failed” implant even before the fixture actually becomes clinically mobile. Sometimes, mobility can be the result of a loose prosthetic component. Such circumstances should be corrected immediately.31
CUMULATIVE INTERCEPTIVE SUPPORTIVE THERAPY
The cumulative interceptive supportive therapy (CIST) protocol has been proposed as a management strategy for peri-implant diseases. It includes four sequential steps to be used depending on the severity and extent of lesions.32
The first step is mechanical debridement and is the treatment of choice for peri-implant lesions limited to the soft tissue, which are evidenced by slight inflammation and probing depths not exceeding 3 mm to 4 mm. The use of metal tips and curets on roughened implant surfaces is thought to be detrimental to the implant surface, possibly causing increased plaque attraction and accumulation.33 Clinicians should be careful to avoid damaging the implant surface. Curets and scalers made of plastic, carbon, and resins can be used, but research shows they provide incomplete biofilm removal and occasionally deposit the curet material on the implant surface.34 A recent study compared titanium-tipped hand instruments to ultrasonic devices with a specially designed tip for implant surfaces. While both groups showed a decrease in probing depths and bleeding scores, there were not many differences between the two sets of instruments.35 These could serve as instruments of choice for plaque debridement.
The second step is antiseptic therapy, which is performed in conjunction with mechanical debridement in areas where the probing depth is increasing along with the presence of plaque and bleeding on probing. Daily application of 0.1% chlorhexidine gluconate (CHX) gel or irrigation and rinsing with 0.12% CHX rinse for 10 days to 14 days has been effective in reducing peri-implant mucositis.36
Antibiotic therapy is the next step in the CIST protocol. Probing depths deeper than 6 mm usually harbor Gram-negative anaerobic bacteria.37 Antibacterial treatment may include antibiotics that can eliminate or reduce the microorganisms in this subgingival environment. According to the CIST protocol, mechanical debridement and antiseptic therapy should be completed before the antibiotic regimen is started. A systemic antibiotic such as metronidazole is the drug of choice and is directed toward Gram-negative microorganisms. Locally delivered antibiotics must have the kinetics to remain at the submucosal site for 7 days to 10 days to be effective. A recent systematic review concluded that locally delivered antibiotics as an adjunct to submucosal debridement resulted in reduction in bleeding on probing scores and probing pocket depths, but it remains unclear what these results mean in terms of treating all sites.38
An implant site with probing depths beyond 5 mm and progressive bone loss that is unresponsive to the first three steps of the CIST protocol usually require surgical intervention. Various methods such as guided bone regeneration,39 implant apicoectomy,40 implantoplasty41 have been suggested with varied success. The treatment approach depends on the size and morphologic characteristics of the lesions in addition to esthetic considerations. Lasers42 and photodynamic therapy43 are other treatment options but limited data on their effectiveness exist. Reosseointegration of an implant has been demonstrated histologically only in an animal model, and this approach needs further research to be considered viable.44
The CIST protocol should be followed if any sign of peri-implant disease is noted. After full-mouth debridement is completed, patient education on oral hygiene should be given.
IMPLANT MAINTENANCE
Implant maintenance is a cornerstone in the prevention of peri-implant diseases. Maintaining an implant patient is very similar to maintaining a patient with treated periodontitis. The recare interval is based on the patient’s past history, susceptibility to periodontitis, and ability to control local factors at home. A maintenance visit should consist of a thorough visual examination of peri-implant soft tissues, peri-implant probing, bleeding on probing, suppuration, and radiographs when indicated.
The American College of Prosthodontists (ACP) recently published Clinical Practice Guidelines for Recall and Maintenance of Patients With Tooth-Borne and Implant-Borne Restorations.45 These guidelines are based on systematic reviews, sound clinical practices, clinical feasibility, and risk-benefit ratios. Patients with fixed implant-borne restorations are advised to visit their dentist once every 6 months. Patients categorized as high-risk should receive professional care more often. At these maintenance appointments, patients should receive a full oral examination, oral hygiene instruction, and prophylaxis. CHX should be applied when antimicrobial action is needed. Only those instruments compatible with the type and material of restoration present should be used. Clinicians should recommend and perform adjustment, repair, replacement, or remake of any or all parts of the prosthesis and/or components that could impair optimal function. Clinical signs of bruxism or clenching should necessitate the fabrication of an occlusal guard to ensure longevity of the fixed restorations.45
At-home recommendations for patients with fixed implant-borne restorations include patient education regarding brushing and the use of oral hygiene aids. Patients with multiple or complex restorations should be advised to incorporate toothpaste containing 0.3% triclosan and limited, short-term use of CHX when needed. These are merely highlights of the extensive ACP guidelines, which cover a variety of restoration types. The full text is available at: jada.ada.org/article/S0002-8177(15)01154-X/fulltext.45
CONCLUSION
The prevalence of dental implant complications grows as more patients undergo implant therapy, despite the technique’s predictability and high success rates. Dental professionals who are knowledgeable about implant diseases will be best able to identify and treat problems as well as provide timely educational interventions aimed at preventing irreparable damage. Patients and each member of the dental team have roles to play to help optimize implant success rates. Dental hygienists should involve the restorative dentist and/or specialists as soon as signs of peri-implant disease are identified. Proactivity and collaboration between specialists, dentists, dental hygienists, and patients will help prevent or minimize implant complications, thus optimizing the utility and longevity of implant therapy.
ACKNOWLEDGMENT
The author would like to thank David Paquette, DMD, MPH, DMSc, for his help with this manuscript.
References
- Zitzmann NU, Berglundh T. Definition and prevalence of peri–implant diseases. J Clin Periodontol.2008;35(Suppl 8):286-291.
- Zablotsky M, Kwan J. Periimplantitis: etiology of the ailing, failing, or failed dental implant. In: Hall WB.Critical Decisions in Periodontology. London: Decker BC; 2003:256–257.
- Mefferet RM. How to treat ailing and failing implants. Implant Dent. 1992;1:25–33.
- Albrektsson T, Isidor F. Consensus report of session IV. In: Lang NP, ed. First European Workshop onPeriodontology. Berlin: Quintessence; 1994:365–369.
- Roos-Jansaker AM, Lindahl C, Renvert H, Renvert S. Nine- to fourteen-year follow-up of implant treatment.Part II: Presence of peri-implant lesions. J Clin Periodontol. 2006;33:290–295.
- Mombelli A, Lang NP. The diagnosis and treatment of peri-implantitis. Periodontol 2000. 1998;17:63–76.
- Salvi GE, Aglietta M, Eick S, Sculean A, Lang NP, Ramseier CA. Reversibility of experimental peri-implantmucositis compared with experimental gingivitis in humans. Clin Oral Implants Res. 2012;23:182–190.
- Zitzmann NU, Berglundh T, Marinello CP, Lindhe J. Expression of endothelial adhesion molecules in thealveolar ridge mucosa, gingiva and peri-implant mucosa. J Clin Periodontol. 2002;29:490–495.
- Lindhe J, Meyle J. Peri-implant diseases: Consensus Report of the Sixth European Workshop onPeriodontology. J Clin Periodontol. 2008;35(Suppl 8):282–285.
- American Academy of Periodontology. Peri-implant mucositis and peri-implantitis: a current understandingof their diagnoses and clinical implications. J Periodontol. 2013;84:436–443.
- Chambrone L, Chambrone LA, Lima LA. Effects of occlusal overload on peri-implant tissue health: asystematic review of animal-model studies. J Periodontol. 2010;8:1367–1378.
- Sousa V, Mardas N, Farias B, et al. A systematic review of implant outcomes in treated periodontitispatients. Clin Oral Implants Res. September 18, 2015. Epub ahead of print.
- Karoussis IK, Kotsovilis S, Fourmousis I. A comprehensive and critical review of dental implant prognosis inperiodontally compromised partially edentulous patients. Clin Oral Implants Res. 2007;18:669–679.
- Heitz-Mayfield LJ, Huynh-Ba G. History of treated periodontitis and smoking as risks for implant therapy. IntJ Oral Maxillofac Implants. 2009;24(Suppl):39–68.
- Schwartz-Arad D, Samet N, Mamlider A. Smoking and complications of endosseous dental implants.J Periodontol. 2002;73:153–157.
- Serino G, Strom C. Peri-implantitis in partially edentulous patients: association with inadequate plaquecontrol. Clin Oral Implant Res. 2009;20:169-174.
- Wilson TG Jr. The positive relationship between excess cement and peri-implant disease: a prospectiveclinical endoscopic study. J Periodontol. 2009;80:1388–1392.
- Wadhwani C, Hess T, Faber T, Pinô€€ƒyro A, Chen CSK. A descriptive study of the radiographic density ofimplant restorative cements. J Prosthet Dent. 2010;103:295–302.
- Present S, Levine RA. Techniques to control or avoid cement around implant-retained restorations.Compend Contin Educ Dent. 2013;34:432–437.
- Moy PK, Medina D, Shetty V, Aghaloo TL. Dental implant failure rates and associated risk factors. Int J OralMaxillofac Implants. 2005;20:569–577.
- Kotsovilis S, Karoussis IK, Fourmousis I. A comprehensive and critical review of dental implant placement indiabetic animals and patients. Clin Oral Impl Res. 2006;1:587–599.
- Chrcanovic BR, Albrektsson T, Wennerberg A. Diabetes and oral implant failure: a systematic review. J DentRes. 2014;93:859–867.
- Loe H. Periodontal disease—the sixth complication of diabetes mellitus. Diabetes Care. 1993;16(Suppl1):329–334.
- Heitz-Mayfield L, Schmid B, Weigel C, et al. Does excessive occlusal load affect osseointegration? Anexperimental study in the dog. Clin Oral Implants Res. 2004;15:259–268.
- Asikainen P, Klemetti E, Vuillemin T, Sutter F, Rainio V, Kotilainen R. Titanium implants and lateral forces.Clin Oral Implants Res. 1997;8:465–486.
- Isidor F. Loss of osseointegration caused by occlusal load of oral implants. A clinical and radiographic studyin monkeys. Clin Oral Implants Res. 1996;7:143–152.
- Costa FO, Takenaka-Martinez S, Cota LO, Ferreira SD, Magalhaes Silva GL, Costa JE (2012). Peri-implantdisease in subjects with and without preventive maintenance: a 5-year follow-up. J Clin Periodontol.2012;39:173–181.
- Lang NP, Adler R, Joss A, Nyman S. Absence of bleeding on probing. An indicator of periodontalstability. J Clin Periodontol. 1990;17:714–721.
- Etter TH, Hakanson I, Lang NP, Trejo PM, Caffesse RG. Healing after standardized clinical probing of the periimplantsoft tissue seal: a histomorphometric study in dogs. Clin Oral Implants Res. 2013;24:91–95.
- Mombelli A, Lang NP. Clinical parameters for evaluation of dental implants. Periodontol 2000. 1994;4:81–86.
- Broggini N, McManus LM, Hermann JS, Medina R, Schenk RK, Buser D, Cochran DL. Peri-implantinflammation defined by the implant- abutment interface. J Dent Res. 2006;85:473–478.
- Berglundh T, Lang NP, Lindhe J. Clinical Periodontology and Implant Dentistry. 2nd ed. Ames, Iowa:Blackwell Munksgaard; 2008.
- Quirynen M, Marcchal M, Busscher HJ, Weerkamp AH, Darius PL, van Steenberghe D. The influence ofsurface free energy and surface roughness on early plaque formation. An in vivo study in man. J ClinPeriodontol. 1990;17:138–144.
- Louropoulou A, Slot DE, Van der Weijden FA. Titanium surface alterations following the use of differentmechanical instruments: a systematic review. Clin Oral Implants Res. 2012;23:643–658.
- Renvert S, Samuelsson E, Lindahl C, Persson GR. Mechanical non-surgical treatment of peri-implantitis: adouble-blind randomized longitudinal clinical study. I: Clinical results. J Clin Periodontol. 2009;36:604-609.
- Porras R, Anderson GB, Caffesse R, Narendran S, Trejo PM. Clinical response to two different therapeuticregimens to treat peri-implant mucositis. J Periodontol. 2002;73:1118–1125.
- Tamura N, Ochi M, Miyakawa H, Nakazawa F. Analysis of bacterial flora associated with peri-implantitisusing obligate anaerobic culture technique and 16S rDNA gene sequence. Int J Oral Maxillofac Implants.2013;28:1521–1529.
- Muthukuru M, Zainvi A, Esplugues EO, Flemmig TF. Non-surgical therapy for the management of periimplantitis:a systematic review. Clin Oral Implants Res. 2012;23(Suppl 6):77–83.
- Froum SJ, Froum SH, Rosen PS. A regenerative approach to the successful treatment of peri-implantitis: aconsecutive series of 170 implants in 100 patients with 2- to 10-year follow-up. Int J Periodontics RestorativeDent. 2015;35:857–863.
- Balshi SF, Wolfinger GJ, Balshi TJ. A retrospective evaluation of a treatment protocol for dental implantperiapical lesions: long-term results of 39 implant apicoectomies. Int J Oral Maxillofac Implants.2007;22:267–272.
- Lang NP, Wilson TG, Corbet EF. Biological complications with dental implants: their prevention, diagnosisand treatment. Clin Oral Impants Res. 2000;11:146–155.
- Takasaki AA, Aoki A, Mizutani K, Kikuchi S, Oda S, Ishikawa I. Er:YAG laser therapy for peri-implant infection:a histological study. Lasers Med Sci. 2007;22:143–157.
- Schar D, Ramseier A, Eick S, Arweiler NB, Sculean A, Salvi GE. Anti-infective therapy of peri-implantitis withadjunctive local drug delivery or photodynamic therapy: six-month outcomes of a prospective randomizedclinical trial. Clin Oral Implants Res. 2013;24:104–110.
- Perrson LG, Berglundh T, Lindhe J, Sennerby L. Re-osseointegration after treatment of peri-implantitis atdifferent implant surfaces. An experimental study in the dog. Clin Oral Implants Res. 2001;12:595–603.
- Bidra AS, Daubert DM, Garcia LT, et al. Clinical practice guidelines for recall and maintenance of patientswith tooth-borne and implant-borne dental restorations. J Am Dent Assoc. 2016;147:67–74.
From Dimensions of Dental Hygiene. May 2016;14(05):49–54.



