
The Ethics of Off-Label Drug Use
With a continued rise in the use of off-label drug therapies, oral health professionals need to be aware of related legal and ethical considerations.
This course was published in the March 2018 issue and expires March 2021. The authors have no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Explain the process of federal drug approvals, and the ethical and legal implications of off-label therapy.
- Describe the need for careful clinical decisions regarding off-label indications, and the levels of evidence supporting such use.
- Discuss the importance of informed consent when providing off-label drug treatment.
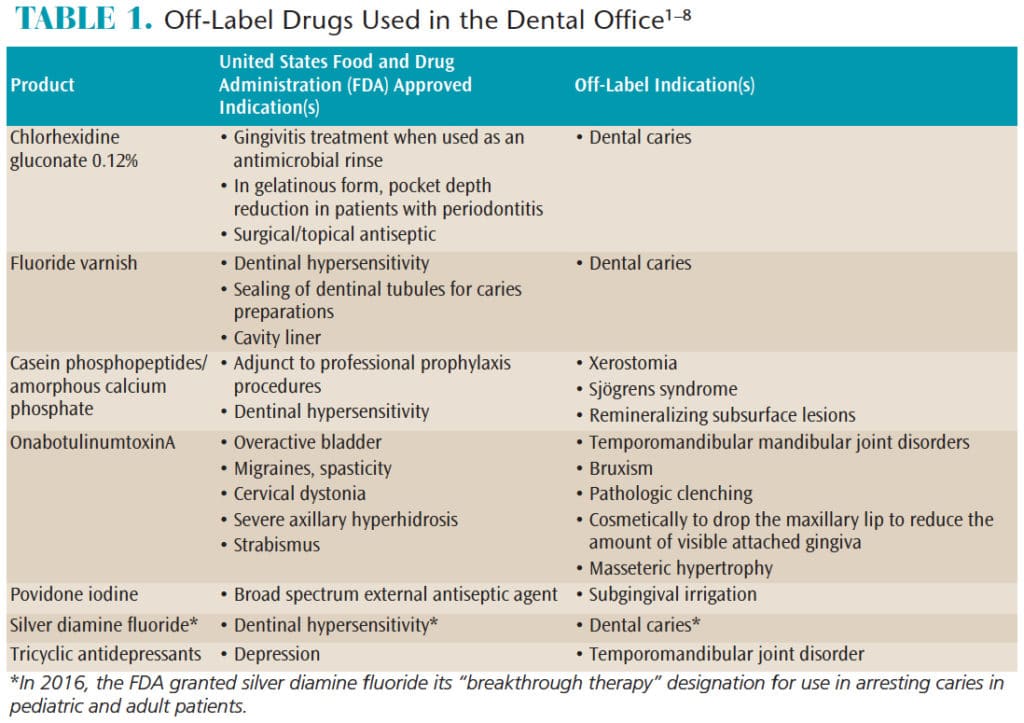
Although medical advances have made it possible for individuals to live longer, advanced age is often influenced by medications used for indications not approved by the United States Food and Drug Administration (FDA). Using drugs off-label has become a common practice in dentistry (Table 1),1–8 and this increase in off-label therapy underscores the clinical need for an advanced knowledge of pharmacology, as there can be ethical and legal implications associated with drugs used in this manner.
The Food, Drug, and Cosmetics Act of 1938, including amendments made in 1962, requires that all drugs and medical devices pass FDA review for a specific use.9 Based on a long process of drug development, review, and clinical trials, the FDA approves drugs and medical devices for specific therapies stated on the FDA-approved label (Figure 1). While this process can be arduous and might vary slightly among drugs,10 once the FDA has endorsed the safety and efficacy of a drug, companies may begin to market and sell the drug in the US for the approved and specified disease or condition. Once on the market, however, such drugs are often used for off-label therapies. The possibility of adverse reactions, ineffective treatment, lack of evidence supporting such indications, and prescribing practices are all concerns regarding off-label drug use. Despite these concerns, the FDA recognizes that off-label drug uses may be relevant treatment options—and, in some cases, could even represent a standard of care.11,12 However, legal implications can arise resulting from an absence of informed consent when using drugs for indications that lack the appropriate FDA approval confirming safety and efficacy.13
 As off-label indications are discovered for a particular drug, the FDA does not evaluate the safety and efficacy of such uses. Because lack of approval for these indications does not prohibit prescribing the drug for that purpose, manufacturers typically opt out of entering yet another long and expensive review process to obtain approval for off-label uses. Pharmaceutical companies are prohibited from advertising these off-label uses, but, under the FDA Modernization Act of 1997, may distribute peer-reviewed journal articles discussing off-label uses.14 One stipulation of this act is that the “company commits itself to file, within a specified time frame, a supplemental application based on appropriate research to establish the safety and effectiveness of the unapproved use.”14
As off-label indications are discovered for a particular drug, the FDA does not evaluate the safety and efficacy of such uses. Because lack of approval for these indications does not prohibit prescribing the drug for that purpose, manufacturers typically opt out of entering yet another long and expensive review process to obtain approval for off-label uses. Pharmaceutical companies are prohibited from advertising these off-label uses, but, under the FDA Modernization Act of 1997, may distribute peer-reviewed journal articles discussing off-label uses.14 One stipulation of this act is that the “company commits itself to file, within a specified time frame, a supplemental application based on appropriate research to establish the safety and effectiveness of the unapproved use.”14
ETHICS OF OFF-LABEL DRUG USE
As noted, among the concerns for prescribing drugs off-label is the risk of adverse reactions or ineffective treatment.15 Drugs used off-label for conditions in which little or no approved treatment indications exist in particular patient populations (such as children, pregnant women, or older adults) are highly understudied.16 Drug safety is a concern in off-label use and continued monitoring is recommended. Factors that might affect the safety of the medication include patient age, range of comorbidities, polypharmacy, drug-disease interactions, and differences in pharmacokinetics and pharmacodynamics.16 Despite these safety concerns, monitoring is difficult due to a lack of controlled studies and observations.
In order to provide the safest and most ethically sound treatment, clinical decisions should be based on relevant scientific evidence, after weighing the potential risks and rewards associated with each treatment option. Clinical decisions lacking sufficient scientific evidence can increase the risk of adverse reactions.
EVALUATION AND PRIORITIZATION
Largent et al17 identified four characteristics that demand increased scrutiny of evidence when prescribing drugs off-label, and the levels of evidence needed to guide prescription practices. These characteristics included drugs that are new, costly, possess novel off-label uses, or contain known adverse effects.17 Three levels of evidence were suggested supporting the use of off-label drugs. These included supported, suppositional, and investigational evidence corresponding to the level of certainty, based on existing evidence showing a patient would benefit from the off-label treatment.
Supported off-label use correlated to a moderate to high level of certainty, in which a drug may be prescribed off-label without concern. Higher levels of evidence, such as meta-analyses or systematic reviews, reinforce supported off-label use. Suppositional off-label use corresponds with a low level of certainty, and, therefore, risk and benefit analysis should be completed, including consultation with colleagues and securing informed consent from patients. Higher-level studies are not available to support suppositional evidence. Investigational off-label use corresponds with a very low level of certainty, so these drug uses should be limited to research protocols due to the unpredictability of a benefit to the patient.9 Proper evaluation of the evidence is necessary to make the appropriate treatment recommendations and increase the safety of off-label therapies.
LEGAL IMPLICATIONS
While the laws and regulations concerning off-label drug use are controversial and sometimes unclear, legal claims have been made against physicians for adverse reactions related to drugs used off-label. As off-label drug use continues to increase in dentistry, legal claims can potentially arise, making it vital for dental professionals to understand the legal implications associated with off-label drug use. Medical lawsuits over off-label indications addressed issues of unregulated research, inadequate informed consent, and medical negligence.13
Unregulated Use of a Research Drug. When using drugs and medical devices, it is often difficult to differentiate research vs practice.18 When clinical trials begin after obtaining an Investigational New Drug approval from the FDA, institutional review boards are proactive in the protection of human subjects. Once a drug is approved, the FDA has no part in the regulation of off-label uses and physician practice. According to Riley and Basilius:18 If a physician’s use of a drug qualifies as experimental research, the physician has an increased risk of professional discipline and civil liability if such experimental use does not comply with the safeguards and oversight of a formal study carefully designed to monitor the drug’s safety and effectiveness.
The National Commission for the Protection of Human Subjects of Biomedical and Behavioral Research differentiates research and practice based on outcome goals.18 Research tests hypotheses, draws conclusions, and adds to a knowledge base, while practice aims to diagnose, prevent, or treat.
In 2007, Bax Global was sued in a malpractice case by a patient who was the recipient of an artificial disk inserted into her spine.18The disk was used off-label due to the location of placement in her spine. She argued that because the use was off-label, it was also experimental. The court did not find this use experimental because the off-label placement was widely used in Europe and constituted an easier procedure than the approved use. Riley and Basilius18 stated, based on this case, that a physician can avoid professional liability if the patient’s needs are foremost and scientific data supporting the off-label use are available.
Informed Consent. There is no court-mandated law that requires a prescribing provider to disclose information regarding the off-label use of a drug.13 Wittich et al13 reported that physicians do not obtain informed consent for off-label use due to concerns of unnecessarily frightening patients, and the possibility of the information overshadowing more critical patient care issues. In light of the high prevalence of off-label drug use, researchers have explored the informed consent practices among physicians that employ drugs for off-label purposes. To et al19 conducted a study involving 105 members of the Australian and New Zealand Society of Palliative Medicine providers. This study showed that 26% of physicians never obtained verbal consent from the patient/caregiver, while 49% sometimes did, and 24% always did. Additionally, 74% never obtained written consent, 23% sometimes did, and 1% always did. When asked about documentation practices, 49% never documented their reasons for off-label prescribing, 38% sometimes did, and 10% always did. A study by Culshaw et al20 involving 332 participants showed that 22% of prescribers do not inform patients that prescribed drugs were for off-label indications, while only 3% stated they always do.
In the practice of dental hygiene, there are professional standards that must be upheld. According to the American Dental Hygienists’ Association’s Standard for Clinical Dental Hygiene Practice, practitioners must “obtain and document informed consent and/or informed refusal” as part of the planning process.21 This informed consent includes the dental hygiene diagnosis, treatment recommendations, risks, and benefits of the procedure, prognosis, alternative treatment options and their associated risks, and the consequences of noncompliance.22–24 The treatment plan with a signed informed consent represents an agreement between the patient and the clinician acknowledging the problems that will be addressed, services to be rendered, and financial obligation of the patient. The consent obtained can be either verbal or written; however, written consent is advised. Verbal consent would be difficult to prove if a lawsuit was pursued by the patient.23 If the consent is verbal, the dental hygienist must fully document the consent in the patient’s chart should legal documentation need to be produced.24 To establish a strong case supporting a lack of informed consent the patient must prove that the dental hygienist:
- Had a duty to disclose sufficient information about the proposed treatment to obtain the patient’s informed consent
- Breached that duty
- The breach of duty was the cause of the injury sustained by the patient23
Failure to obtain informed consent from a patient prior to treatment is considered battery.26
Despite providing all of the appropriate information about a proposed treatment, including any off-label use, the patient has a right to exercise informed refusal.23,24 Such refusal should be documented thoroughly and it must be established that the patient understood the risks and possible outcomes associated with the refusal.23,24
The off-label use of products during dental therapy should necessitate informed consent from patients; for example, informed consent should be obtained when applying fluoride varnish as an anti-caries treatment, as this is considered an off-label use. Table 1 provides examples of other off-label uses for which informed consent should be obtained.1–8
Medical Negligence. Another legal consideration when using drugs off-label is medical negligence. There are four elements of tort law concerning negligence that must be proved in order to justify liability: the prescribing physician must have a duty to the patient, that duty must be breached, there must be some injury requiring compensation, and there must be a causal link between the breach and injury.13 Helm25 stated that for medical negligence to be ascertained, it must be proven that the physician strayed from the standard of practice in the community, and that FDA regulation of off-label promotion by the manufacturers makes it difficult for the physician to gather information regarding the standard of practice with certain off-label uses. Additionally, oral health professionals should be mindful that if a patient consents to treatment without the appropriate information provided to him or her, the clinician might be liable for negligence.26
PRACTICAL IMPLICATIONS AND CONCLUSION
To ensure patient safety, oral health professionals should recognize and inquire about medications and the conditions for which they are being used, particularly when off-label use is suspected. Thoroughly reviewing the patient’s medical history and discussing current medications are essential to an accurate diagnosis and the provision of safe, effective care. Tam et al27 performed a systematic review analyzing studies discussing the frequency, type, and clinical importance of medication history errors at hospital admission. After reviewing 22 studies that included 3,755 participants, prescription medication history errors were found in 67% of all cases. Additionally, 34% to 95% of patients had at least one error related to the identification of prior allergies or adverse drug reactions. Considering the lack of scientific evidence related to off-label prescriptions, clinicians should be cautious and accurate when reviewing medical histories and evaluating the drugs their patients are using. In the event of medication reconciliation errors, oral health professionals should protect themselves and their patients legally via informed consent.
Studies showing a lack of informed consent from patients using drugs off-label mandate that oral health professionals be knowledgeable in this aspect of pharmacology in order to prevent possible medical emergencies in the dental office. Although many clinicians have been casual about gaining informed consent, it might be timely to consider the legal ramifications of using off-label drugs, and to be more prescriptive about informing patients and documenting that patients have consented to this application of an off-label drug. It is possible that clinicians may have taken for granted the use of some of these products without recognizing they are off-label applications, and assume that patients understand the drugs are being used in a different manner than for what they were approved. In light of this, oral health professionals should educate patients and offer the option to consent or decline treatment in this capacity.
As the use of off-label drug therapies in medicine continues to increase, it is likely the products and medications used in dentistry will see a corresponding rise in off-label indications. As such, clinicians need to be aware of the legal and ethical considerations related to off-label drug use. Adopting informed consent practices is one means to help legally protect providers and patients. Additionally, the lack of scientific evidence supporting some off-label indications requires an advanced knowledge of pharmacology in order to make the appropriate treatment modification for patients using drugs in this fashion.
Examples of an informed consent form that could be used when providing off-label therapy, as well as an informed refusal form are available with the web version at: dimensionsofdentalhygiene.com.
REFERENCES
- United States Food and Drug Administration. Inspections, Compliance, Enforcement, and Criminal Investigations. Available at: fda.gov/iceci/enforcementactions/warningletters/2012/ucm330758.htm. Accessed February 9, 2018.
- US Food and Drug Administration. Premarket Notification: Dentsply International. Available at: accessdata.fda.gov/cdrh_docs/pdf12/k122331.pdf. Accessed February 9, 2018.
- US Food and Drug Administration. Botox Label: Highlight of Prescribing Information. Available at: accessdata.fda.gov/drugsatfda_docs/label/2016/103000s5252lbl.pdf. Accessed February 9, 2018.
- Nayyar P, Kumar P, Nayyar PV, Singh A. Botox: broadening the horizon of dentistry. J Clin Diagn Res. 2014;8:ZE25–ZE29.
- Lexicomp. Chlorhexidine Gluconate. Available at: wolterskluwercdi.com/lexicomp-online/. Accessed February 9, 2018.
- Li Y, Tanner A. Effect of antimicrobial intervention on oral microbiota associated with early childhood caries. Pediatr Dent. 2015;37:226–244.
- Hoang T, Jorgensen MG, Keim RG, Pattison AM, Slots J. Povidone-iodine as a periodontal pocket disinfectant. J Periodontal Res. 2003;38:311–317.
- Cascos-Romero J, Vázquez-Delgado E, Vázquez-Rodríguez E, Gay-Escoda C. The use of tricyclic antidepressants in the treatment of temporomandibular joint disorders: systematic review of the literature of the last 20 years. Med Oral Patol Oral Cir Bucal. 2009;14:E3–E7.
- Klein DB, Tabarrok A. Do off-label drug practices argue against FDA efficacy requirements? A critical analysis of physicians’ argumentation for initial efficacy requirements. Am J Econ Sociol. 2008;67:743–775.
- US Food and Drug Administration. The FDA’s Drug Review Process: Ensuring Drugs Are Safe and Effective. Available at: fda.gov/drugs/resourcesforyou/consumers/ucm143534.htm. Accessed February 9, 2018.
- US Food and Drug Administration. Guidance for Industry: Responding to Unsolicited Requests for Off-Label Information About Prescription Drugs and Medical Devices. Available at: fda.gov/downloads/drugs/guidancecomplianceregulatoryinformation/guidances/ucm285145.pdf. Accessed February 9, 2018.
- Eguale T, Buckeridge DL, Verma A. et al Association of off-label drug use and adverse drug events in an adult population. JAMA Intern Med. 2015;176:1–9.
- Wittich, C, Burkle, C, Lanier, W. Ten common questions (and their answers) about off-label drug use. Mayo Clin Proc. 2012;87:982–990.
- US Food and Drug Administration. Regulatory Information. FDA Backgrounder on FDAMA. Available at: fda.gov/RegulatoryInformation/LawsEnforcedbyFDA/SignificantAmendmentstotheFDCAct/FDAMA/ucm089179.htm. Accessed February 9, 2018.
- Dresser R, Frader J. Off-label prescribing: a call for heightened professional and government oversight. J Law Med Ethics. 2009;37:476–486.
- Dal Pan GJ. Monitoring the safety of off-label medicine use. WHO Drug Inf. 2009;23:21–22.
- Largent E, Miller FG, Pearson SD. Going off-label without venturing off-course: evidence and ethical off-label prescribing. Arch Intern Med. 2009;169:1745–1747.
- Riley JB, Basilius PA. Physicians’ liability for off-label prescriptions. Nephrol News Issues. 2007;21:43–44,46–47.
- To TM, Agar M, Shelby-James T, et al. Off-label prescribing in palliative care—a cross-sectional national survey of palliative medicine doctors. Palliat Med. 2013;27:320–328.
- Culshaw J, Kendall D, Wilcock A. Off-label prescribing in palliative care: a survey of independent prescribers. Palliat Med. 2013;27:314–319.
- American Dental Hygienists’ Association. Standards for Clinical Dental Hygiene Practice. Available at: adha.org/resources-docs/2016-Revised-Standards-for-Clinical-Dental-Hygiene-Practice.pdf. Accessed February 9, 2018.
- Moreira NC, Pachêco-Pereira C, Keenan L, Cummings G, Flores-Mir C. Informed consent comprehension and recollection in adult dental patients: A systematic review. J Am Dent Assoc. 2016;147:605–619.
- Cameron CA, Zarkowski PM. Dental hygienist-patient relationship. In: Beemsterboer PL, ed. Ethics and Law in Dental Hygiene. 3rd ed. St. Louis: Elsevier Saunders; 2010:79–90.
- Zarkowski P. Legal and ethical decision making. In: Darby ML, Walsh MM, eds. Dental Hygiene Theory and Practice. 3rd ed. St. Louis: Elsevier Saunders; 2010:1183–1209.
- Helm KA. Protecting public health from outside the physician’s office: a century of FDA regulation from drug safety labeling to off-label drug promotion. Fordham Intellect Prop Media Entertainment Law J. 2007;18:117–187.
- Davison JA. Legal and Ethical Considerations For Dental Hygienists and Assistants. St. Louis: Mosby Inc; 2000.
- Tam VC, Knowles SR, Cornish PL, Fine N, Marchesano R, Etchells EE. Frequency, type and clinical importance of medication history errors at admission to hospital: a systematic review. CMAJ. 2005;173:510–515.
From Dimensions of Dental Hygiene. March 2018;16(3):48-51



