 JANULLA / ISTOCK / THINKSTOCK
JANULLA / ISTOCK / THINKSTOCK
Severe Tooth Wear: A Case Report
When addressing the oral health and esthetic concerns of a patient with gastroparesis, effective patient education, risk assessment, and appropriate therapies are integral to successful treatment.

Oral health professionals should be prepared to identify clinical findings associated with diagnosed or undiagnosed gastric diseases, dietary habits, and psychological disorders (eg, bulimia) that may result in vomiting and reflux. Gastric fluids are composed of hydrochloric acid, which may have a pH of 1.0 to 2.0.1 These extremely strong acids can cause surface softening of enamel and dentin, increasing the risk of tooth surface loss caused by bruxism, toothbrushing, and consuming hard or abrasive foods.2 In addition to the intrinsic effects of gastric fluids, erosive tooth wear may also be related to the extrinsic dietary acids found in many foods and sugar-sweetened beverages.3 As such, when erosion is found, oral health professionals need to identify its etiology and institute behavioral and preventive strategies to help manage this problem. Among oral health practitioners, dental hygienists are in a unique position to diagnose and manage key aspects of this irreversible oral health issue.4 This article presents a case study of erosive tooth wear caused by gastroparesis to help guide practitioners on effective management strategies.
GASTROPARESIS
Gastroparesis is a chronic gastrointestinal motility disorder characterized by delayed gastric emptying.5 Patients experience partial paralysis of the stomach, which results in food retention in the stomach for long periods. Gastroparesis symptoms include nausea, vomiting, and bloating.5,6 Patients with gastric disorders who seek medical treatment are provided with dietary counseling and may be prescribed medications to alleviate symptoms. Oral health professionals need to question patients regarding a gastroparesis diagnosis on the medical history in order to effectively treatment plan.7
CASE REPORT
A young woman with the chief complaint of extremely sensitive teeth presented to the dental office. Her medical history revealed a diagnosis of gastroparesis. The patient was taking esomeprazole and omeprazole, both of which
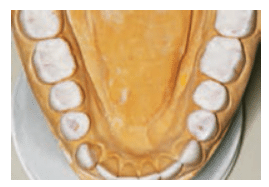
are proton pump inhibitors that suppress production of gastric acids. In addition to the pain and functional difficulties her dentition was causing, the patient was self-conscious about how she looked when she smiled (Figure 1).
A comprehensive examination was performed. The clinical exam revealed erosion with dentin exposure. Staining, decalcification, noncarious cervical lesions, and gingival recession were also found and noted in the patient’s record. The patient’s gingival display was adequate and within normal limits. A caries risk assessment was completed and her caries risk level was deemed high because of low salivary pH and hyposalivation.
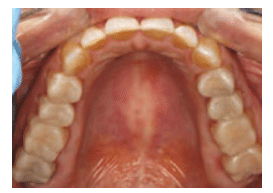
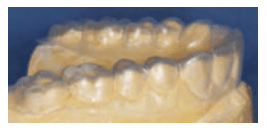
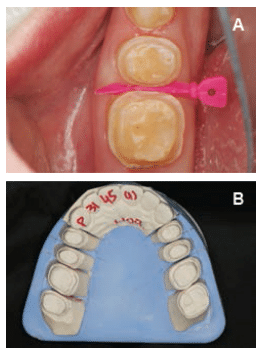
A treatment plan was formulated and discussed with the patient. A diagnostic wax-up of the proposed occlusal scheme was completed (Figure 2). Oral hygiene instructions were presented. The patient was prescribed a high-fluoride toothpaste and given xylitol mints and oral spray containing a pH-neutralizing agent.8 The patient was also counseled on dietary habits. Additionally, oral side effects of the gastroparesis medications, such as decreased salivary flow leading to xerostomia, were discussed.
After re-evaluating the patient’s caries risk level, an esthetic treatment plan was formulated. Lithium disilicate all-ceramic onlays were planned for tooth #18, #19, #20, and #29. Lithium disilicate all-ceramic crowns were planned for tooth #21, #28, #30, and #31. These all-ceramic crowns have high success rates.9 Composite buildups were planned for tooth #22 through #27. Because the patient’s periodontal condition was stable and there were no biologic width problems, crown lengthening surgery and gingivectomy were not required. The decision was made to increase the vertical dimension of occlusion due to the loss of tooth structure, but mainly for restorative convenience—meaning more room was required for the restorations without the need for excessive reduction of the teeth.10
SUSAN DO, DDS
The patient accepted the proposed treatment plan. A smile design was done based on the wax-up. The patient was enthusiastic about beginning treatment. Before preparing the teeth, the patient was placed in temporary splinted composite overlays at the new vertical dimension of occlusion (Figure 3 and Figure 4). The patient adapted well to these changes.
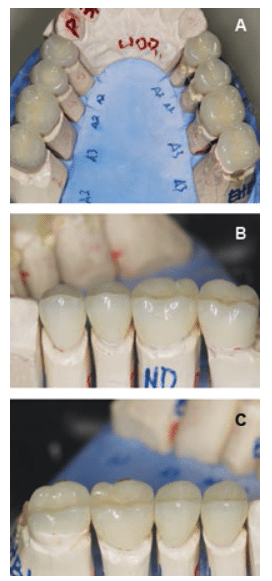
With the new vertical dimensions of occlusion, the maxillary anterior teeth were prepared for crowns. Anterior teeth #6 to #11 were prepared, impressed, and temporized for lithium disilicate all-ceramic restorations. At the next appointment, lithium disilicate all-ceramic crowns were applied to teeth #6 to #11. The patient was extremely pleased with the esthetic result. At the following appointments, posterior quadrants were prepared and restored sequentially, using bonded lithium disilicate all-ceramic onlays and crowns (Figure 5A to Figure 6C). Composite buildups were completed on the lower anterior teeth #22 to #27.
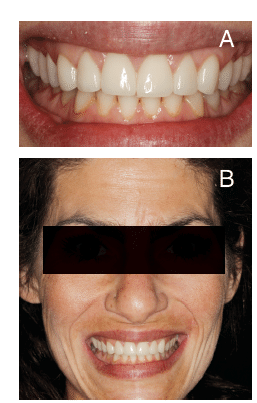
The patient was pleased with the result. Her sensitivity was eliminated, and her function and appearance were improved (Figure 7A and Figure 7B).
DISCUSSION
Dental hygienists need to be able to identify dental erosion and erosive tooth wear.11,12 They should recognize signs of dental erosion, determine the etiology, and discuss clinical findings with the patient. Sensitive findings in the medical history, such as the effects of eating disorders (anorexia, bulimia) should be handled cautiously.7 Patients who are unaware that gastric acid may cause erosion should be referred to their physician for additional diagnostic testing. In addition, patients should be educated regarding the risks of acid reflux to their general and oral health. In cases where it is determined that the erosive tooth wear is due to dietary habits, patients should be counseled to limit their intake of acidic foods and beverages.13 They need to understand that it is far better to manage tooth surface loss early, as opposed to a “wait and watch” approach, which may lead to the need for more extensive and expensive restorative dentistry.
Patients should be placed on a frequent recare schedule that includes caries risk assessment, monitoring of dietary acid intake, and reinforcement of preventive strategies. This will help to support patients’ oral health, as well as the stability and longevity of their restorations.
CONCLUSION
Even though the etiology of dental erosion is multifactorial, oral health professionals should be able to identify possible causes and discuss these with the patient. Educating patients on causes of dental erosion is important in clinical practice. Oral health professionals should work with patients’ physicians to ensure proper counseling and treatment management. In particular, dental hygienists can play a key role in the identification and management of severe erosive tooth wear.
ACKNOWLEDGEMENT
The authors would like to thank Susan Do, DDS, for providing the images.
REFERENCES
- Moazzez R, Bartlett D. Intrinsic causes of dental erosion. Monogr Oral Sci. 2014;25:180–196.
- Ranjitkar S, Smales RJ, Kaidonis JN. Oral manifestations of gastroesophageal reflux disease. J Gastroenterol Hepatol. 2012;27:21–27.
- Barbour ME, Lussi A. Erosion in relation to nutrition and the environment. Monogr Oral Sci. 2014;25:143–154.
- Lussi A, Caravalho TS. Erosive tooth wear. A multifactorial condition of growing concern and increasing knowledge. Mono Oral Sci. 2014;25:1–15.
- Camilleri M, Grover M, Farrugia G. What are the important subsets of gastroparesis? Neurogastroenterol Motil. 2012;24:597–603.
- Camilleri M, Parkman HP, Shafi MA, Abell TL, Gerson L, American College of Gastroenterology. Clinical guideline: management of gastroparesis. Am J Gastroenterol. 2013;108:18–37.
- Lussi A. Dental erosion clinical diagnosis and case history taking. Euro J Oral Sci. 1996;104:191–198.
- Bellamy R, Harris R, Date RF, et al. In situ clinical evaluation of a stabilized stannous fluoride dentifrice. Int Dent J. 2014;25:197–205.
- Christensen GJ. The all-ceramic restoration dilemma: where are we? J Am Dent Assoc. 2011;142:668–671.
- Spear FM, Kokich VG, Marhew D. Interdisciplinary management of anterior dental esthetics. J Am Dent Assoc. 2006;137:160–169.
- Barbour M, Lussi A, Shellis R. Screening and predication of dental erosion. Caries Res. 2011;45(Suppl 1):24–32.
- Hellwig E, Lussi A. Oral hygiene products, medications and drugs—hidden etiologic factors for dental erosion. Monogr Oral Sci. 2014;25:155–162.
- Cochrane NJ, Cai F, Yuan Y, Reynolds EC. Erosive potential of beverages sold in Australian schools. Aust Dent J. 2009;54:238–244.
From Dimensions of Dental Hygiene. October 2016;14(10):24–26,29.

