
Reducing Stroke Risk
Improving periodontal health may also help prevent stroke.
This course was published in the April 2015 issue and expires April 30, 2018. The authors have no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Define stroke and its signs and symptoms.
- Discuss the guidelines for preventing stroke.
- Explain the relationship between periodontal diseases and stroke.
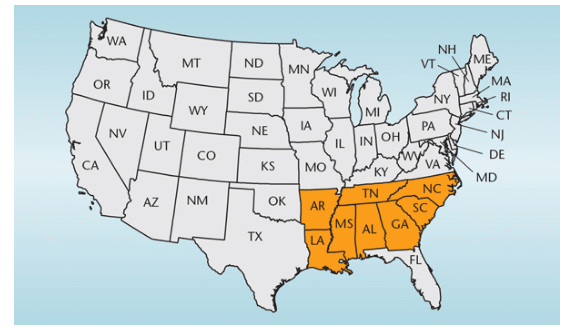
Stroke is the fifth leading cause of death, the chief cause of preventable disability, and a major economic burden in the United States.1 Approximately 795,000 Americans experience a new or recurring stroke each year, with one stroke occurring every 40 seconds. Stroke also kills more than 137,000 people each year. As of 2010, US patients were paying approximately $74 billion annually for stroke-related medical costs. In the US, stroke mortality rates remain highest among residents of the Southeastern “stroke belt,” which comprises eight states: Alabama, Arkansas, Georgia, Louisiana, Mississippi, North Carolina, South Carolina, and Tennessee (Figure 1).2 In this region, the stroke mortality rate has remained higher than the rest of the US since 1940.
As the US population grows, the prevalence of stroke also is projected to increase. By 2030, it is estimated that nearly 4% of Americans will have experienced a stroke. The combination of an aging population and increased stroke prevalence translates to potential swelling of stroke-related health-care costs.3 Thus, steps need to be taken to improve stroke prevention and treatment.
STROKE DEFINED
Stroke is the sudden onset of neurological deficits of a vascular origin lasting for greater than or equal to 24 hours, and confirmed through a brain scan provided either by computed tomography or magnetic resonance imaging.4 Stroke occurs when an area of the brain does not receive adequate blood flow due to a blood clot or ruptured blood vessel, resulting in the deprivation of oxygen and nutrients and potential loss of function in the affected area.
Signs and symptoms of stroke include the onset of one-sided weakness or numbness and difficulty with vision, speech, thought, and/or coordination.1 Symptoms that improve within 24 hours, or even within minutes, with no evidence of a stroke identified on a brain scan, are categorized as a mini stroke—or transient ischemic attack (TIA), which is caused by a temporary blood clot. Patients who have experienced a mini-stroke are at increased risk for a subsequent stroke compared to people who have not experienced a TIA.1
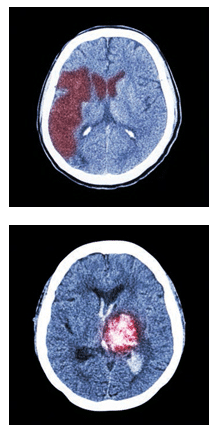
There are two types of stroke: ischemic and hemorrhagic (Figure 2). Ischemic stroke occurs when there is a lack of blood supply to the brain, while hemorrhagic stroke refers to bleeding in and around the brain. Ischemic strokes are 87% more common and are similar in mechanism to heart attacks in that they are caused by the hardening of the blood vessel (atherosclerosis) and/or a clot (thrombus)—earning the nickname “brain attack.” Strokes, when diagnosed early, are treatable; however, strokes often go undiagnosed and untreated due to misdiagnosis.5
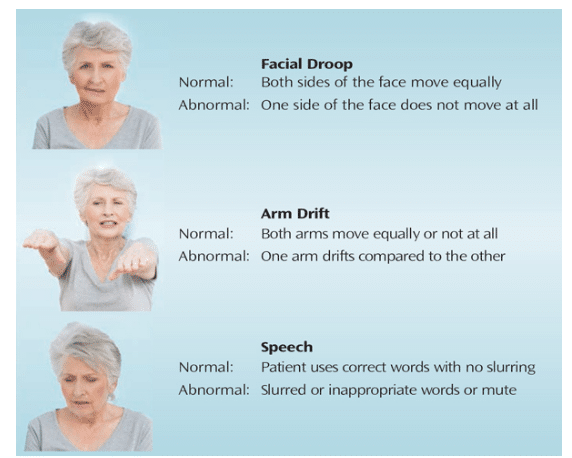
A simple means of screening for stroke is referred to as FAST: facial droop, arm/limb weakness, speech difficulty, time to dial 911 (Figure 3). Abnormal facial droop is present when one side of the face does not move at all or moves unequally to the opposite side. When one arm drifts either slightly or considerably compared to the other, this is abnormal and can be an early sign of stroke. An individual may have slurred speech, use inappropriate or incorrect words, or may be unable to speak at all. When these abnormalities are present, call 911 immediately.
When patients experiencing a stroke are admitted to an emergency department, brain scans can be taken and the brain can be treated with a clot-busting medication. Intravenous tissue plasminogen activator (IV t-PA) is a clot-dissolving medicine that helps to eliminate the blood vessel blockage and restore blood flow to the affected brain region, and is considered the gold standard for treating ischemic strokes. If administered within 3 hours of stroke-symptom onset, IV t-PA may significantly improve the chances of recovery. Results from the National Institute of Neurological Disorders and Stroke study found that patients who received IV t-PA in accordance with a strict protocol were 30% more likely to recover from stroke within 3 months without significant disability.6 In medical centers with intervention capabilities, catheter-based clot retrieval devices can be used (up to 8 hours from symptom onset), further reducing the likelihood of long-term deficits due to stroke.7
PREVENTION GUIDELINES
According to the American Stroke Association, 80% of strokes are preventable. The current standards for prevention of ischemic stroke include blood thinners, cholesterol-lowering medications, lifestyle changes, and treatment of risk factors such as high blood pressure.5
The 2014 American Heart Association/ American Stroke Association Guidelines for the Primary Prevention of Stroke suggest that modifiable risk factors include high blood pressure, high cholesterol, diabetes, physical inactivity, poor diet and nutrition, obesity, and cigarette smoking. High blood pressure is the most important, modifiable risk factor because its treatment is one of the most effective strategies for preventing both ischemic and hemorrhagic strokes. Patients who have high blood pressure should be treated with anti-hypertensive drugs to target blood pressure at 140 mm/90 mm Hg. Patients with high cholesterol and an increased risk of cardiovascular events should seek treatment with a statin for the prevention of stroke. Individuals with diabetes should receive a comprehensive program that includes tight control of high blood pressure and glycemic management, which can reduce microvascular complications and the risk of stroke.8
Capable adults should take part in moderate-to-vigorous physical activity for at least 40 minutes, 3 days to 4 days per week. Epidemiological studies and randomized trials have determined that diets low in sodium, high in potassium, and rich in fruits and vegetables reduce the risk of stroke, including the Mediterranean and Dietary Approaches to Stop Hypertension, or DASH diets. Individuals who are overweight (BMI=25 kg/m2 to 29 kg/m2) and obese (BMI >30 kg/m2) should aim for weight reduction in order to lower blood pressure and reduce the risk of stroke. Cigarette smoking is known to increase the risk of ischemic stroke and subarachnoid hemorrhages. Studies have shown a reduction in stroke risk because of smoking cessation and community-wide smoking bans. To assist smokers in quitting, both drug therapy (nicotine replacement) and counseling are recommended.8
Individuals who have experienced an initial ischemic stroke or TIA are at high risk for a recurrent event. On average, this population has a 3% to 4% risk for another ischemic event. The estimated risk for individuals is dependent on many factors, including comorbidities and adherence to preventive therapy. Because the risk for recurrent ischemic events is high, the 2014 American Heart Association/American Stroke Association Guidelines for the Prevention of Stroke in Patients with Stroke and Transient Ischemic Attack presents guidelines for prevention of future ischemic events among survivors of stroke or TIA.9
The most important intervention for the prevention of secondary stroke is the treatment of high blood pressure (systolic blood pressure greater than or equal to 140 mm Hg or a diastolic pressure greater than or equal to 90 mm Hg). The prevalence of high blood pressure among patients with a recent ischemic stroke is approximately 70%. Therefore, patients who have blood pressure greater than or equal to 140 mm Hg systolic or greater than or equal to 90 mm Hg diastolic after the first several days of an initial ischemic stroke or TIA should be started on a blood pressure therapy regimen. For patients who have an initial stroke or TIA assumed to be of atherosclerotic origin and a low-density lipoprotein cholesterol level greater than or equal to 100 mg/dL, a statin therapy with lipid-lowering effects is recommended to reduce the risk of recurrent events. All patients after an initial stroke or TIA should also be screened for diabetes by testing fasting plasma glucose, glycated hemoglobin (HbA1c), or via an oral glucose tolerance test. However, the HbA1c may be more accurate in the immediate period following the event. Existing American Diabetes Association guidelines for glycemic control are recommended for patients with stroke or TIA who may also be prediabetic or have diabetes.9
For patients with a history of ischemic stroke or TIA, nutritional assessments should be conducted and patients should be referred for individualized dietary counseling as necessary. Additionally, these patients should reduce their sodium intake to 1.5 g to 2.4 g per day to further reduce blood pressure. Patients at risk for secondary stroke should also follow diets low in sodium, high in potassium, and rich in fruits and vegetables. Capable adults with ischemic stroke or TIA should participate in moderate to vigorous physical activity at least three times to four times a week to reduce secondary risk factors. These sessions should last an average of 40 minutes and should be sufficient enough to break a sweat or raise the heart rate (eg, walking briskly, bicycling). Health-care providers should advise patients who have had a stroke or TIA and smoked in the past year to quit and avoid secondhand tobacco smoke. In an effort to help these patients quit smoking, health-care providers should offer counseling, nicotine replacement products, and oral smoking cessation medications.9
THE RELATIONSHIP BETWEEN PERIODONTAL DISEASES AND STROKE
The etiological theory of periodontal diseases has changed drastically over the past 50 years, from simplistic hypotheses to a more specific understanding of bacterial interactions and the systemic effect of periodontitis. In the 1960s, it was believed that periodontal diseases were caused by an irritation due to the deposit of calculus near gingival tissue. Over the next 15 years, it became more widely accepted that the quantity of nonspecific plaque, not the virulence of individual bacteria species, was responsible for periodontal destruction. During the late 1970s and early 1980s, researchers began to realize that periodontitis could vary greatly between patients, regardless of the quantity or quality of bacteria levels. Additionally, it became apparent that the existence of bacterial plaque alone was not enough to explain the progression of periodontal diseases—specifically the loss of connective tissue and alveolar bone. After this realization, researchers theorized that periodontal diseases were more likely mediated by a host-bacterial interaction, and that the host’s response to the presence of bacteria determines periodontal destruction.10
Periodontitis is present in nearly half of the US population. Among adults older than 30, 8.7% have mild periodontitis, 30% have moderate periodontitis, and 8.5% have severe periodontitis.11 Tooth loss as a result of severe periodontal diseases, which is associated with high stroke risk and stroke risk factors, disproportionately impacts residents of the stroke belt.12 Evidence from observational studies indicates that stroke is approximately 1.5 times to 3 times more common among individuals with periodontal diseases compared to those without.13
A recent prospective study by Whitely et al14 found an association between inflammatory markers with recurrent vascular events in stroke and TIA patients. Results showed that patients with recent stroke or TIA and higher levels of interleukin-6, C-reactive protein, fibrinogen, and white blood cells were associated with an increased probability of recurrent stroke.14 Data from this study also suggest that deaths resulting from stroke may be strongly linked to higher levels of inflammation. Periodontal diseases may be the result of a complex interaction between infection and immunity, resulting in an inflammatory response.15 The inflammatory response may be the link to the association of inflammatory response and recurrent vascular events. Thus, the inflammatory markers could potentially serve as modifiable stroke risk factors.16
Evidence suggests that inflammation plays a dominant role in the development of atherosclerosis, and atherosclerosis is a major component of cardiovascular and cerebrovascular diseases.17,18 More specifically, the presence of periodontal bacteria and periodontitis has been associated with increased thickness of carotid intima media and endothelial dysfunction present in the early stages of arterial disease.19 Mercanoglu et al20 determined that endothelium-dependent dilation and endothelium-independent dilation, measured by high-resolution vascular ultrasound, were significantly impaired in patients with chronic periodontitis.20 Furthermore, Tonetti et al19 found that patients with periodontitis who received intensive treatment saw an improvement in endothelial function, specifically flow-mediated dilation, over time. In this study, intensive treatment included full-mouth intensive removal of subgingival dental plaque biofilms through scaling and root planing. When necessary, teeth were extracted and minocycline microspheres were delivered to the dental pockets. The intensive periodontitis treatment initially resulted in a temporary, acute systemic inflammatory response and impaired endothelial function. However, at 6 months post-therapy, intensive treatment as opposed to standard mechanical scaling and root planing was associated with reduced periodontal disease severity and improved endothelial function.19
THEORIZED MECHANISMS BEHIND THE ASSOCIATION
Periodontal diseases may cause local and systemic inflammation, and growing evidence suggests that periodontal infection may be linked to an increase in atherosclerosis-induced conditions such as stroke.10,21 Microorganisms may enter the bloodstream during normal activities, such as chewing food, in direct relation to the severity of periodontal diseases.22,23 The introduction of these microorganisms can cause a systemic inflammation response, interfering with the body’s insulin action,24,25 which, in turn, leads to hyperglycemia and consequently results in cholesterol abnormalities and high blood pressure.26
The theorized mechanism linking periodontal diseases to systemic inflammatory damage is actually a combination of three etiological mechanisms: Gram-negative bacteria speeding to the bloodstream as a result of compromised gingival tissue; injury of the epithelial lining due to the circulation of periodontal pathogens and toxins; and systemic inflammation caused by the immunologic response to the pathogens and their toxins.10 More specifically, oral flora initially creates a subgingival biofilm and triggers a local inflammatory challenge. The inflammation of the gingiva then mediates pocketing and bone loss in which oral bacteria are able to invade, creating an inflammatory site of sepsis. The sepsis disperses cytokines, bacteria, and bacterial products into the bloodstream, and both the inflammatory and microbial challenge trigger an acute phase response in the liver. The liver then releases acute phase proteins to include fibrinogen, C-reactive protein, and interleukin 6. The C-reactive proteins and the interleukin 6 promote atherogenesis and oxidized low-density lipoprotein accumulation. The accumulation of atheroma increases the instability of plaque on the artery walls, which, in turn, increases the risk for atherosclerosis-induced disease, such as stroke.10
TREATMENT OF GINGIVITIS AND STROKE PREVENTION
Chronic inflammation plays a critical role in the development of atherosclerosis and systemic diseases, such as cerebrovascular disease. Periodontitis is a prevalent yet preventable chronic disease linked to the introduction of inflammatory markers into the circulatory system.10 This disease is preventable in that it can be attributed to poor oral hygiene, and prevention can be implemented to combat its onset and effects.
De Oliveira et al27 suggest that an increase in the frequency of toothbrushing can decrease the concentration of C-reactive protein. Studies have found a strong association between periodontal diseases and the rise of C-reactive proteins, which is an independent predictor of cardiovascular disease among intermediate-risk individuals and those who are middle age.27
Chen et al28 found that regular scaling and root planing decreases the incidence of stroke in comparison to those not receiving full-mouth or localized periodontal debridement. During a 7-year follow-up period, patients receiving at least one full-mouth or localized scaling and root planing had significantly lower incidence of myocardial infarction and stroke, as well as total cardiovascular event-free survival than the subjects not receiving treatment.28
Lee et al29 examined the strength of the association between periodontal treatment and the incidence of ischemic stroke. Results suggest that periodontitis, even after adjustment for known risk factors, is a significant risk factor for stroke. In this study, individuals diagnosed with periodontitis who did not receive treatment experienced the greatest risk for ischemic stroke among all periodontally involved and nonperiodontally involved groups. Furthermore, the patients with periodontal diseases who required intensive treatment or tooth extraction had more severe, irreversible stages of periodontal disease and higher stroke incidence rates. Patients who received regular dental treatment in this study were less likely to develop cerebrovascular disease. Over a 10-year follow-up period, the patients with periodontitis who received periodontal maintenance therapy had the lowest stroke incidence rate among all age and comorbidity groups.29
GUIDELINES FOR DENTAL CARE
All new dental patients should have their blood pressure taken at the first and subsequent appointments. Patients with high blood pressure, and those who are at risk for developing high blood pressure, should be managed by taking blood pressure readings during certain dental procedures such as oral surgery, restorative treatments, placement of dental implants, and periodontal surgery.
Patients who are new to the practice should be allowed to rest for 5 minutes upon arriving prior to receiving a blood pressure reading. During the reading, patients should be sitting upright with their arms supported at heart level. Two or more readings should be taken at least 5 minutes apart and then averaged to determine appropriate action based on the mean blood pressure.30
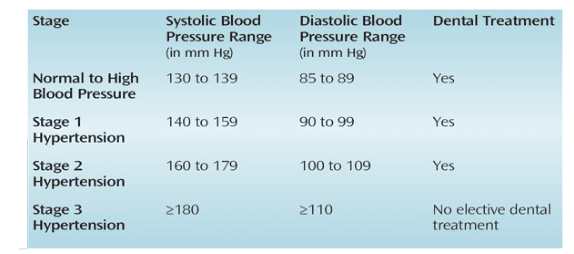
Suggestions for dental care based on blood pressure readings and varying stages of hypertension are available (Table 1).30 For example, patients with stage 3 hypertension should not receive any elective dental treatment and should immediately be referred to a physician. Patients with stage 1 and stage 2 hypertension can have their blood pressure rechecked in 1 week to 2 weeks. If, at that time, their blood pressure is still greater than 140/90 mm Hg, they should be referred to a physician prior to the start of dental treatment. Patients with optimal or slightly elevated blood pressure may receive dental treatment and should routinely have their blood pressure checked.30
Patients with a history of stroke or TIA should be screened for either antiplatelet or anticoagulant intervention before beginning dental treatment. Ischemic stroke patients may be divided into two subgroups based on the origin of the stroke: cardioembolic or noncardioembolic. Patients can then be prescribed antiplatelets or anticoagulants in an effort to prevent a recurrent event.9
Secondary stroke prevention for patients with a history of noncardioembolic stroke includes treatment with antiplatelets, such as aspirin, clopidogrel, and aspirin, plus extended-release dipyridamole.9 Dental patients seeking an elective dental procedure who are also taking antiplatelets should be managed with simple hemostasis. Clinicians can also request that patients stop taking the medication 7 days prior to the appointment. Secondary stroke prevention for patients with a history of ischemic stroke of cardioembolic origin includes treatment with anticoagulants, such as warfarin, or a direct thrombin inhibitor, including dabigatran, apixaban, and rivaroxaban.9
For stroke or TIA patients on warfarin who seek elective dental treatment, the prothrombin time international normalized ratio test can be ordered to ensure normal clotting behavior is present and to measure the effectiveness of the prescription. This test should be done no more than 3 days before the procedure. Warfarin affects the function of the coagulation cascade and helps inhibit the formation of blood clots. For patients taking this medication, the prothrombin time international normalized ratio should be between 2.0 and 3.0 for basic blood thinning needs. Patients within the normal range while on warfarin are manageable during dental procedures, but they should be carefully monitored.31,32 These patients also can be treated with a hemostatic mouthrinse, such as amniocaproic acid, to control localized bleeding.33 Patients with a history of stroke or TIA who are taking direct thrombin inhibitors can also be tested for drug effectiveness; however, there is not standard antidote for these relatively newer anticoagulants. Studies suggest that patients seeking elective dental procedures should be at no additional risk of bleeding while taking direct thrombin inhibitors because local hemostatic measures, such as pressure and topical agents, are adequate to control bleeding.34
CONCLUSION
Periodontal diseases will continue to be associated with risk factors for stroke and stroke itself. Mounting evidence suggests that treatment and prevention of periodontitis may result in the reduction of this risk. Thus, there is a growing need for treatment trials, specific to the treatment of periodontal diseases and prevention of stroke, as well as screening tools that can be utilized by physicians to assess gingival health and provide proper intervention in the effort to prevent stroke.
REFERENCES
- American Stroke Association. About Stroke. Available at: strokeassociation.org/STROKEORG/AboutStroke/AboutStroke_UCM_308529_SubHomePage.jsp. Accessed March 9, 2015.
- Borhani NO. Changes and geographic distribution of mortality from cerebrovascular disease. Am J Public Health Nations Health. 1965;55:673–681.
- Ovbiagele B, Goldstein LB, Higashida RT, et al. Forecasting the future of stroke in the United States: a policy statement from the American Heart Association and American Stroke Association. Stroke. 2013;44:2361–2375.
- Sacco RL, Kasner SE, Broderick JP, et al. An updated definition of stroke for the 21st century: a statement for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2013;44:2064–2089.
- National Stroke Association. What Is Stroke? Available at:stroke.org/understand-stroke/what-stroke. Accessed March 9, 2015.
- . Tissue plasminogen activator for acute ischemic stroke. The National Institute of Neurological Disorders and Stroke rt-PA Stroke Study Group. N Engl J Med. 1995;333:1581–1587.
- Berkhemer OA, Fransen PS, Beumer D, et al. A randomized trial of intraarterial treatment for acute ischemic stroke. N Engl J Med. 2015;372:11–20.
- Meschia JF, Bushnell C, Boden-Albala B, et al. Guidelines for the primary prevention of stroke: a statement for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2014;45:3754–3832.
- Kernan WN, Ovbiagele B, Black HR, et al. Guidelines for the prevention of stroke in patients with stroke and transient ischemic attack: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2014;45:2160–2236.
- Hein C, Cobb C, Clacopino, A. Report of the Independent Panel of Experts of the Scottsdale Project. Grand Rounds Oral Systemic Med. 2007:1–27.
- Eke PI1, Dye BA, Wei L, Slade GD, et al. Update on prevalence of periodontitis in adults in the United States: NHANES 2009-2012. J Periodontol. 2015;17:1–18.
- You Z, Cushman M, Jenny NS, Howard G. Tooth loss, systemic inflammation, and prevalent stroke among participants in the reasons for geographic and racial difference in stroke (REGARDS) study. Atherosclerosis. 2009;203:615–619.
- Sfyroeras GS, Roussas N, Saleptsis VG, Argyriou C, Giannoukas AD. Association between periodontal disease and stroke. J Vasc Surg. 2012;55:1178–1184.
- Whiteley W, Jackson C, Lewis S, et al. Association of circulating inflammatory markers with recurrent vascular events after stroke: a prospective cohort study. Stroke. 2011;42:10–16.
- Johansson A, Johansson I, Eriksson M, Ahrén AM, Hallmans G, Stegmayr B. Systemic antibodies to the leukotoxin of the oral pathogen Actinobacillus actinomycetemcomitans correlate negatively with stroke in women. Cerebrovasc Dis. 2005;20:226–232.
- Diaz J, Sempere AP. Cerebral ischemia: new risk factors. Cerebrovasc Dis. 2004;17(Suppl 1):43–50.
- Vlachopoulos C, Dima I, Aznaouridis K. Acute systemic inflammation increases arterial stiffness and decreases wave reflections in healthy individuals. Circulation. 2005;112:2193–2200.
- Schillinger M, Exner M, Mlekusch W, et al. Inflammation and Carotid Artery—Risk for Atherosclerosis Study (ICARAS). Circulation. 2005;111:2203–2209.
- Tonetti MS, D’Aiuto F, Nibali L, et al. Treatment of periodontitis and endothelial function. N Engl J Med. 2005;356:911–920.
- Mercanoglu F, Oflaz H, Oz O, et al. Endothelial dysfunction in patients with chronic periodontitis and its improvement after initial periodontal therapy. J Periodontol. 2004;75:1694–1700.
- Burt B, Research, Science and Therapy Committee of the American Academy of Periodontology. Position paper: epidemiology of periodontal diseases. J Periodontol. 2005;76:1406–1419.
- Kinane DF, Riggio MP, Walker KF, MacKenzie D, Shearer B. Bacteraemia following periodontal procedures. J Clin Periodontol. 2005;32:708–713.
- Lockhart PB, Brennan MT, Thornhill M, et al. Poor oral hygiene as a risk factor for infective endocarditis-related bacteremia. J Am Dent Assoc. 2009;140:1238–1244.
- Horng T, Hotamisligil GS. Linking the inflammasome to obesity-related disease. Nat Med. 2001;17:164–165.
- Hotamisligil GS. Inflammatory pathways and insulin action. Int J Obes Relat Metab Disord. 2003;27(Suppl 3):S53–55.
- Grundy SM. Hypertriglyceridemia, atherogenic dyslipidemia, and the metabolic syndrome. Am J Cardiol. 1998;81:18B–25B.
- de Oliveira, C, Watt R, Hamer M. Toothbrushing, inflammation, and risk of cardiovascular disease: results from Scottish Health Survey. BMJ. 2010;340:c2451.
- Chen ZY, Chiang CH, Huang CC, et al. The association of tooth scaling and decreased cardiovascular disease: a nationwide population-based study. Am J Med. 2012;125:568–575.
- Lee YL, Hu HY, Huang N, Hwang DK, Chou P, Chu D. Dental prophylaxis and periodontal treatment are protective factors to ischemic stroke. Stroke. 2013;44:1026–1030.
- Little JW. The impact on dentistry of recent advances in the management of hypertension. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2000;90:591–599.
- Schafer AI. Approach to the Patient with Bleeding and Thrombosis. In: Goldman L, Schafer AI, eds. Cecil Medicine. 24th ed. Philadelphia: Saunders Elsevier; 2011:174.
- Brewer AK. Continuing warfarin therapy does not increased risk of bleeding for patients undergoing minor dental procedures. Evid Based Dent. 2009;10:52.
- Patatanian E, Fugate SE. Hemostatic mouthwashes in anticoagulated patients undergoing dental extraction. Ann Pharmacother. 2006;40:2205–2210.
- Breik O, Cheng A, Sambrook P, Goss A. Protocol in managing oral surgical patients taking dabigatran. Aust Dent J. 2014;59:296–301.
From Dimensions of Dental Hygiene. April 2015;13(4):49–54.



