
Polishing Particulars
Although polishing is a basic procedure in dental hygiene care, there is new research about its effects on restorations and the efficacy of prophy paste additives.
Polishing as part of a dental prophylaxis is a staple of dental hygiene practice. While consensus has changed over the years regarding best practices, much of dental hygiene polishing technique has remained the same. Today, research is uncovering more details about the safety of polishing esthetic restorations and the possible benefits of additives to prophy pastes. Dental hygienists need to consider all of the available evidence in order to make the best decisions regarding polishing for their patients.
EFFECT ON DENTAL MATERIALS
Esthetic direct restorative materials include various types of resin composites, compomers, and glass ionomers. When polishing esthetic restorations, the primary concerns are surface roughness and loss of gloss. Surface roughness is related to bacterial adhesion, while gloss is an esthetic quality. Composite restorations are made of a resin matrix and filler particles; the size of the filler particles varies with the type of composite material. The low-Moh value resin matrix abrades easily when polished with prophy paste, exposing filler particles or creating craters where particles have dislodged. This produces a rougher surface and decreases gloss. While using prophy pastes on large-particle composites tends to produce a rougher surface than polishing on small-particle composites (eg, micro/nanofilled restorations),1,2 most studies show a general increase in surface roughness for all composites when prophy paste is used.
Not all surface roughness values, however, are above the biological limit of 0.2 um, which is the point of increased plaque accumulation.1,3,4 A general decrease in surface gloss as also been noted with prophy pastes.4 Recommended pastes for polishing composite restorations are aluminum oxide or diamond paste, with the diamond paste producing a smoother finish.5 Glass ionomers are less common than composites and are not as smooth or glossy. Little research is available on the effect of polishing these restorations, but Barnes6 recommends a fine finishing disc or rubber cup without prophy paste to remove stain.
Porcelain is an extremely hard and glossy substance used in crown and bridge applications. Covey et al4 found no increase in surface roughness when porcelain was polished with fine or coarse prophy paste, but did detect a statistically significant, though not clinically significant, decrease in gloss. Polishing with prophy pastes generally increased surface roughness values of smooth amalgam restorations, but they were not raised above the biological threshold of 0.2 um.3
When amalgam restorations require polishing to reduce roughness, pumice followed by tin oxide was shown to produce a surface approaching the ideal level of roughness.7 There is little research examining prophy paste abrasiveness on gold restorations. One study found that treatment with prophy paste produced varying results, but all increased surface roughness values, although none above the biological threshold, and all increases were less than those experienced with amalgam.3
Titanium is an intensely strong and biocompatible metal used in dental implants and abutments. Studies by both McCollum et al and Homiak et al demonstrated that rubber cup polishing with light, intermittent pressure and pumice flour or tin oxide produced a smooth surface with less manufacturing grooves than the controls, which were untreated abutments and abutment condition prior to instrumentation, respectively.8,9
EFFECT OF ADDITIVES
Prophylaxis pastes may include supplemental ingredients for added benefits, including fluoride, xylitol, and calcium phosphate compounds. The federal Food and Drug Administration considers these ingredients secondary, so they are not regulated.10 Studies show that aggressive polishing can remove several microns of fluoride-rich enamel, but fluoride levels of the outer enamel remain constant or become slightly higher when the prophy paste contains fluoride.11,12 Most pastes now include fluoride to replace any that might be lost in the polishing process.

Polishing with a fluoridated prophy paste is not considered a fluoride treatment because the levels provided are much lower than those obtained by professional topical application. Xylitol is a noncariogenic sugar alcohol used as a sweetener. While it can have antimicrobial benefits, these are associated with frequent use and are most likely unobtainable through professional dental polishing.13 Current dental management of noncavitated lesions focuses on remineralizing incipient lesions before they require restoration. Fluoride’s actions in the mineralization cycle of enamel are well-known, but now other compounds are available that may increase remineralization and decrease demineralization.
Three are available in prophy pastes: amorphous calcium phosphate (ACP), casein phosphopeptide-amorphous calcium phosphate (CPP-ACP or Recaldent™), and calcium sodium phosphosilicate (NovaMin®) prophy paste.14 All of these compounds form calcium and phosphate salts in solution. Application of calcium phosphates provides higher ionic mineral concentrations outside the dental tissues than are present inside, thus increasing mineral deposition onto tooth structure. This can be helpful for patients with low salivary flow who have insufficient salivary minerals to provide buffering and remineralization.
Research on prophy pastes containing calcium phosphates is limited, but evidence suggests these compounds may effectively deposit minerals onto tooth structure.15 Developed by the American Dental Association in 1991, ACP is an unstabilized calcium phosphate delivery system that causes ACP to precipitate in the presence of water.16 ACP then transforms into hydroxyapatite, or fluorapatite, in the presence of fluoride crystals.
Early studies indicate that ACP might smooth enamel irregularities, remineralize tooth structure, and decrease dental permeability. 15,17–19 However, because it precipitates quickly, the ions may not have a chance to diffuse deep into the tooth structure and can even block diffusion into the enamel by forming large clusters on tooth surfaces.20 Polishing with ACP prophy paste has been shown to reduce pitted surface roughness significantly more than prophy paste with no ACP.17 CPP-ACP was originally developed in Australia for the treatment of dentinal hypersensitivity.16 CPP-ACP is a stabilized calcium phosphate that remains in ionic form longer than ACP.20 Recent studies indicate CPP-ACP increases fluoride uptake and promotes subsurface remineralization of enamel lesions with fluorapatite when delivered in a dentifrice.21 It also enhances regression of orthodontic white spot lesions when applied topically as a cream twice daily for 12 weeks.22
Calcium sodium phosphosilicate was originally developed to repair and regenerate bone. It occludes dentinal tubules when applied with a rubber cup for 30 seconds.23 Studies show it also reduces surface roughness of artificial defects created by both sandpaper abrasion and acid etching when used as a dentifrice twice a day for 12 weeks. Furthermore, enamel thickness was restored by deposition of minerals onto the defects.24
RECOMMENDATIONS
The materials used in dentistry have varying characteristics, so specific polishing recommendations are needed for each. Because gold is relatively soft, caution is necessary when polishing. A cleansing paste or tin oxide, an extremely fine powder used for metallic restorations when mixed in a slurry, can be used.25 Porcelain is resistant to scratching, so gentle polishing for biofilm removal on posterior porcelain restorations should be safe.
Anterior ceramic restorations, however, should be avoided unless a nonabrasive paste is used due to the potential for loss of gloss. A brief, gentle polish on amalgam for plaque removal will not increase surface roughness significantly, but if the surface is already rough, the clinician should consider first using rubber cups and points, then rubber-cup polishing with pumice, followed by tin oxide to provide a smoother surface that is less retentive for bacteria. Lightly polishing titanium implant abutments with a fine prophy or cleansing paste is recommended to remove plaque and smooth the surface.26
Susceptibility to abrasion in composites is material dependent.1–3 When the specific type of material is unknown, determining whether polishing will damage the surface is difficult. In general, polishing composite restorations with prophy paste should be avoided. If composite restorations are to be polished for stain removal, an aluminum oxide or diamond paste should be used for the composites, followed by a fine prophy paste or cleaning agent for the rest of the teeth.
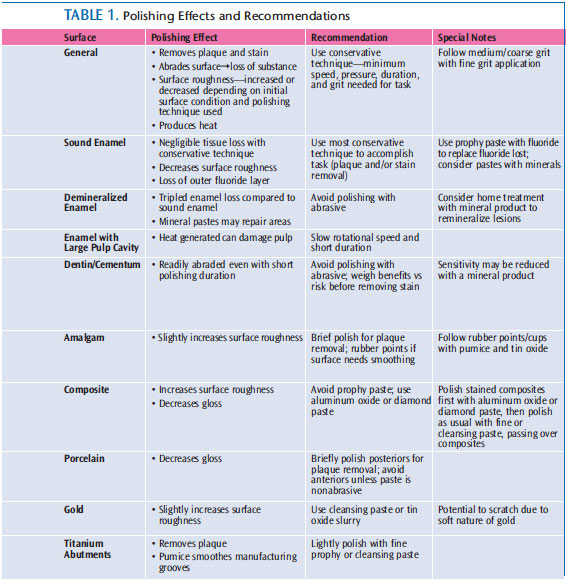
When possible, abrasive pastes should contain fluoride to maintain fluoride levels in outer enamel. Calcium phosphate pastes may fill in surface irregularities and diffuse into enamel to remineralize decalcified areas. Table 1 provides a review of polishing effects and recommendations.
SUMMARY
Dental hygiene is a dynamic, ever-changing field that relies on scientific study. While rubber cup polishing is common, there is a dearth of well designed, clinically-relevant research. More scientific study is required in order to provide dental hygienists with an evidence base from which to determine appropriate treatment options.
REFERENCES
- Yap AU, Wu SS, Chelvan S, Tan ES. Effect of hygiene maintenance procedures on surface roughness of composite restoratives. Oper Dent. 2005;30:99–104.
- Neme AM, Wagner WC, Pink FE, Frazier KB. The effect of prophylactic polishing pastes and toothbrushing on the surface roughness of resincomposite materials in vitro. Oper Dent. 2003;28:808–815.
- Roulet JF, Roulet-Mehrens TK. The surface roughness of restorative materials and dental tissues after polishing with prophylaxis and polishing pastes. J Periodontol. 1982;53:257–266.
- Covey DA, Barnes C, Watanabe H, Johnson WW.Effects of a paste-free prophylaxis polishing cup and various prophylaxis polishing pastes on tooth enamel and restorative materials. Gen Dent. 2011;59:466–473.
- Sen D, Göller G, Issever H. The effect of two polishing pastes on the surface roughness of bis-acryl composite and methacrylate-based resins. J Prosthet Dent. 2002;88:527–532.
- Barnes CM. Polishing esthetic restorative materials:The successful maintenance of esthetic restorations.Dimensions of Dental Hygiene. 2010;8(1):24–28.
- Bollen CM, Lambrechts P, Quirynen M. Comparison of surface roughness of oral hard materials to the threshold surface roughness for bacterial plaque retention: a review of the literature. Dent Mater. 1997;13:258–269
- McCollum J, O’Neal RB, Brennan WA, Van Dyke TE,Horner JA. The effect of titanium implant abutment surface irregularities on plaque accumulation in vivo.J Periodontol. 1992;63:802–805.
- Homiak AW, Cook PA, DeBoer J. Effect of hygiene instrumentation on titanium abutments: A scanning electron microscopy study. J Prosthet Dent. 1992;67:364–369.
- US Food and Drug Administration. Jurisdictional update: dental prophylaxis pastes with drug components. Available at: www.fda.gov/CombinationProducts/JurisdictionalInformation/JurisdictionalUpdates/ucm106560.htm. Accessed May 8, 2013.
- Stearns RI. Incorporation of fluoride by human enamel. 3. In vivo effects of nonfluoride and fluoride prophylactic pastes and APF gels. J Dent Res.1973;52:30–35.
- Vrbic V, Brudevold F, McCann HG. Acquisition of fluoride by enamel from fluoride pumice pastes. HelvOdontol Acta. 1967;11:21–26.=
- American Academy of Pediatric Dentistry. Policyon the use of xylitol in caries prevention, revised2010. Available at: www.aapd.org/media/Policies_Guidelines/P_Xylitol.pdf. Accessed May 8,2013.
- Focus on prophy pastes and air polishingpowders. Dimensions of Dental Hygiene.2012;10(6):46–50.
- Tung MS, Malerman R, Huang S, McHale WA.Reactivity of prophylaxis paste containing calcium,phosphate and fluoride salts. J Dent Res.2005;84:Special Issue A.
- Hurlbutt M. Caries management with calciumphosphate: Expand your armamentarium with thesenew technologies. Dimensions of Dental Hygiene.2010;8(10):40–46.
- Winston AE, Charig A, Patel V, McHale WA,Malerman R. Effect of prophy pastes on surface oftooth enamel. J Dent Res. 2005;84:Special Issue A.
- Tung MS, O’Farrell TJ, Liu DW. Remineralization byamorphous calcium phosphate compounds. J DentRes. 1993;72:1738.
- Tung MS, Eichmiller FC. Amorphous calciumphosphates for tooth mineralization. CompendiumCont Educ Dent. 2004;25(Suppl):9–13.
- Cochrane NJ, Cai F, Huq NL, Burrow MF, ReynoldsEC. New approaches to enhanced remineralization oftooth enamel. J Dent Res. 2010;89:1187–1197.
- Reynolds EC, Cai F, Cochrane NJ, et al. Fluorideand casein phosphopeptide-amorphous calcium phosphate. J Dent Res. 2008;87:344–348.
- Bailey DL, Adams GG, Tsao CE, et al. Regression ofpost-orthodontic lesions by a remineralizing cream.J Dent Res. 2009;88:1148–1153.
- Sauro S, Watson TF, Thompson I. Dentine desensitization induced by prophylactic and airpolishing procedures: an in vitro dentine permeability and confocal microscopy study. J Dent.2010;38:411–422.
- Burwell AK, Litkowski LJ, Greenspan DC. Calciumsodium phosphosilicate (NovaMin): remineralization potential. Adv Dent Res. 2009;21:35–39.
- Anusavice KJ. Phillips’ Science of Dental Materials.11th ed. St. Louis: Saunders; 2003:362, 370–372.
- Rose LF, Mealey BL, Genco RJ, Cohen DW.Periodontics: Medicine, Surgery and Implants. St. Louis:Elsevier Mosby; 2004:270.
From Dimensions of Dental Hygiene. June 2013; 11(6): 24, 26, 28.

