Maternal Infection – Understanding the Threat
What the evidence says about the relationship between periodontal infection and preeclampsia.
This course was published in the October 2008 issue and expires October 2011. The author has no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Understand the connection between intrauterine infection and preterm birth.
- Identify the specific relationship between periodontal infections and preeclampsia.
- List research findings to support the association between periodontal infections and preterm birth.
- Be able to identify recommended considerations when treating pregnant patients.
1996 was the year that Offenbacher published his groundbreaking research suggesting that periodontal infection may increase the risk of adverse outcomes.1 Since then, the potential relationship between infection of periodontal origin and preterm birth (PTB), resulting from preeclampsia, has been substantially investigated.2-18 Preeclampsia is a precursor to PTB and is threatening to human pregnancy, placing both the mother and fetus at risk. The exact nature of the relationship between periodontal infection and preeclampsia is still unclear. Further research is necessary to confirm or refute a causal relationship. However, the medical, dental, public health, and insurance communities have already responded to existing evidence by instituting various interventions, including health promotion and preventive strategies in prenatal care, screening women who may be at risk, and implementing treatment to reduce the threat that periodontal disease may pose to pregnancy.
ABOUT PREECLAMPSIA
Preeclampsia is a pregnancy specific, multifactorial condition that refers to a set of symptoms rather than any causative factor. It is one of the major causes of maternal and fetal morbidity and mortality.15 The condition affects 7%-10% of pregnancies,19 usually presenting after 20 weeks of gestation. First pregnancies appear to be more vulnerable.15 Preeclampsia is diagnosed when a pregnant woman develops high blood pressure (two separate readings taken at least 4 hours apart of 140/90 or higher) and proteinuria (300 mg of protein in a 24-hour urine sample).20 Unusual swelling of the hands, feet, or face can also be a symptom of preeclampsia. The condition is reversible through abortion or delivery.20 Risk factors include nulliparity (never have carried a pregnancy), primiparity (first child pregnancy), familial history of preeclampsia, uterine abnormalities, obesity, chronic hypertension, renal diseases, diabetes, multiple gestation, and maternal age.15
Although significant progress has been made toward understanding preeclampsia, the etiology of this life threatening disorder remains elusive.8 It is considered a syndrome caused by an excessive systemic inflammatory response to pregnancy.19 A mild increase in the systemic inflammatory response (proinflammatory state) is typical in normal pregnancy.8,11 However, the inflammatory response is significantly enhanced in preeclampsia, characterized by a severe state of inflammation and down-regulation of cellular immunity.11 Therefore, chronic infections from any source increase the inflammatory burden already amplified in normal pregnancy.
INFECTION IN OBSTETRIC OUTCOMES
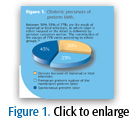
The idea that infection plays a role in an increased risk for adverse pregnancy outcomes is supported by a large body of evidence.8,21-25 Subclinical fetal infection is far more common than traditionally acknowledged.26 Approximately 30%-35% of the obstetric precursors leading to PTB are related to maternal or fetal infections (Figure 1). Intrauterine infection is an important mechanism leading to PTB and may account for 25%-40% of PTBs.26 Because intrauterine infection is difficult to identify, this estimate may be understated.26 An inverse relationship exists between the frequency of intrauterine infection and gestation age at the time of labor—the greater the frequency of intrauterine infection, the earlier the gestational age. Early treatment of urinary and vaginal infections during pregnancy can reduce the incidence of preeclampsia.27
Intrauterine infection can be limited to the decidua (the mucous membrane lining the uterus enveloping the embryo that undergoes special modifications in preparation for and during pregnancy) or it can expand to the space between the amnion (the thin membrane forming a closed sac around the embryo/fetus containing the amniotic fluid) and the chorion (the highly vascular outer embryonic membrane) and reach the amniotic cavity of the fetus.26 The amniotic cavity is usually sterile for bacteria.26 One study28 found that bacteria in the membranes and an associated inflammatory response in the amniotic fluid were present in the predominance of women who experienced early preterm labor.
In gaining access to the amniotic cavity, microorganisms can translocate in a number of ways.26 The most common pathway is the ascending route,26 which is manifested as vaginosis. Vaginosis is an infection characterized by the replacement of Gram-positive microflora present in healthy vaginal tissue by a predominance of Gram-negative, anaerobic bacteria. When this genital tract infection ascends, it leads to intrauterine infection and inflammation.29 Bacterial vaginosis is associated with a one-and-a-half- to three-fold increase in the rate of PTB.26 Translocation of periodontal pathogens from the oral cavity is through haematogenous dissemination (the spread of microorganisms through blood vessels).30 Mounting evidence suggests that periodontal disease confers close to a two-fold increase in the risk for preeclampsia.8,15,10
INTRAUTERINE EXPOSURE
An increasing number of risk factors for preeclampsia are associated with upregulation of systemic inflammation. Infection from periodontal origin may be a risk factor capable of stimulating the cascade of inflammatory events associated with preeclampsia.
The maternal-fetal interface may be exposed to endotoxins, inflammatory cytokines, and oxidative stressors associated with periodontal disease.9 Further, this exposure event may pose a systemic burden to periodontally infected pregnant women,9 creating a vascular stressor that may play a role in the development of preeclampsia.19 In the progression of this inflammatory cascade, maternal periodontal disease may cause intrauterine fetal exposure to oral organisms.31 When microbial invasion of the amniotic cavity occurs, the risk for intra-amniotic inflammation and fetal inflammatory response is increased26 in addition to poor placental perfusion and inflammation. The aftermath is frequently fetal growth restriction, which increases neonatal morbidity and the risk for preterm membrane rupture, premature labor, preterm delivery, and fetal injury.30
The sequelae of fetal injury are serious. Up to 50% of neurologic defects in children may be associated with PTB.32 Extreme low birth weight is associated with asthma, low IQ, cerebral palsy, poor motor skills, and other functional abnormalities.32 Further, maternal oral infectious exposure during pregnancy not only poses a threat during gestation, but may also compromise neurological growth and development around the time of birth.33
About 40% of pregnant women have some form of periodontal infection34 and periodontal infections are more prevalent in pregnant women than all other obstetric and sexually transmitted infections combined. One in five PTBs may be attributable to periodontal infections.1
A LOOK AT THE EVIDENCE
Conde-Agudelo and colleagues8 systematically reviewed nine studies2,3,5,7,9,10,12,13,35 on the relationship between periodontal disease and preeclampsia. Seven2,3,5,9,10,13,35 of the nine studies reported an association between periodontal disease and preeclampsia, with two studies7,12 reporting no association. The meta-analysis also included17 studies on the relationship be tween pre e clamp sia and urinary tract infection, seven studies on the relationship with HIV infection, six studies on the relationship with Chlamydia pneumoniae, four studies on the relationship with malaria, three studies on the relationship with cytomegalovirus, two studies on the relationship with Helicobacter pylori, and one study on the relationship with Epstein-Barr virus, herpes simplex virus type 2, Mycoplasma hominis, adeno-associated virus-2, and bacterial vaginosis. Of all the maternal infections studied, only urinary tract infection and periodontal disease during pregnancy were associated with an increased risk for preeclampsia.
This study emphasizes the magnitude of the threat that periodontal infection may pose to the systemic health of pregnant women and the role periodontal disease may play in preeclampsia. The investigators concluded that more studies are needed to determine whether the relationship between these infections and preeclampsia are causal or simply associative, and to explore the potential mechanisms that may be involved.8
Five studies6,11,15, 18,19 add further credence to the findings of Conde-Agudelo and colleagues. In evaluating the link between the severity of periodontal disease and preeclampsia, Canakci and colleagues6 found that periodontitis— defined as the presence of pocket depth ? 4 mm, clinical attachment level ? 3 mm, and increased percentage of sites with bleeding on probing—is associated with an increased risk for preeclampsia. This is in agreement with previous studies.3,5,9,10,35 The researchers also found that severe preeclamptic women were close to four times more likely to present with severe periodontal disease than women with normal pregnancies, and serum and gingival crevicular fluid levels of Il-1?, TNFa, and PGE2 were significantly higher in the preeclamptic group of subjects than those with normal pregnancies.19
Another recently published report by Canakci and colleagues6 hypothesized that periodontal disease may be a potential risk factor for the severity, progression, and the initiation of preeclampsia as a result of reduced antioxidant capacity and/or increased oxidative stress. The researchers investigated the role of periodontal disease in increasing oxidative stress as an important contributing factor in the pathogenesis of preeclampsia by determining the total antioxidant capacity (shift from oxidant/ antioxidant balance toward oxidative state) in blood serum, saliva, and gingival crevicular fluid in preeclamptic women and women undergoing normal pregnancies. The researchers concluded that oxidative stress (oxidant/antioxidant balance shifts toward the oxidative state) may be an important contributing factor in both preeclampsia and periodontal disease.
Ruma and colleagues36 recently published findings from the secondary analysis of data from the “Oral Conditions and Pregnancy Study,” a prospective cohort study of maternal periodontal disease and obstetric outcomes. The investigators hypothesized that chronic exposure to oral pathogens causes both systemic inflammation and preeclampsia, and that systemic inflammation may explain the biological plausibility of this relationship. The conclusion was that maternal periodontal disease accompanied by C-reactive protein (CRP) (the acute phase protein produced by the liver that is a reliable maker for systemic inflammation) levels > 15.2 ?g/mL early in pregnancy is associated with an increased risk for preeclampsia. Longitudinal data39 on CRP fluctuation in normal pregnancy indicate that CRP levels may be significantly higher in pregnant women compared to nonpregnant women, and that throughout gestation, CRP levels may remain fairly steady at approximately 3 ?g/mL, with no values higher than 15 ?g/mL.
The recently published research of Herrera and colleagues11 confirmed the effect of periodontal disease on increasing CRP levels and the potential risk this may pose in pregnancy. The investigators found that preeclamptic subjects presented a positive dose response relationship between high sensitivity (hs) CRP and periodontal status (after adjusting for body mass index and gestational age). The findings that hs-CRP levels were significantly higher in preeclamptic women with moderate/severe periodontitis, compared to pregnant women with the same periodontal status led researchers to conclude that “inflammation resulting from the most advanced states of the infection may be related to the development of preeclampsia.”
In a case control study of 1,206 Brazilian women, Siqueira and colleagues15 found that maternal periodontitis conveyed a higher risk of developing preeclampsia, and this association remained significant after matching for age, chronic hypertension, and primiparity. These findings are similar to those reported by Cota and colleagues.10 The odds of preeclampsia were associated with an increase in the number of sites with bleeding on probing, pocket depth, and clinical attachment loss ? 4 mm, demonstrating a dose-response effect. The researchers concluded that “maternal periodontitis is a risk factor associated with preeclampsia” and that “periodontal care is an important part of prenatal programming.”
PRACTICAL APPLICATION
In a systematic review of studies published in 2006, Xiong and colleagues17 concluded that the evidence was insufficient to recommend performing periodontal treatment during pregnancy in order to reduce the risk of adverse pregnancy outcomes. However, the inconclusiveness of this body of research and the call for more studies have not precluded a movement toward more progressive intervention of periodontal disease during pregnancy. Following are a number of ideas that dental practitioners may want to consider implementing in the care of women of childbearing years.
1. Establish preconceptional care/counseling as a standard in caring for women of childbearing age. Do not wait to address diseases and conditions of the oral cavity after conception has occurred.
In response to the Michalowicz study38 that found that periodontal treatment did not significantly alter the rates of PTB, Goldenberg and Culhane39 wrote: “Once the inflammatory cascade is activated during pregnancy, interventions targeting this pathway may be ineffective in reducing the rate of PTB. Treatment during pregnancy may be too late; it is possible that treatment either before pregnancy (in nulliparous women) or in the period between pregnancies (for multiparous women, especially those with a history of PTB) may yield more promising results.” Accordingly, diagnosing and treating periodontal disease prior to conception may be more effective in reducing the risk for an adverse pregnancy outcome. Preconceptional counseling is an intervention that focuses on the identification and modification of risks for preventable or modifiable adverse outcomes of pregnancy.
The United States Public Health Service expert panel on the content of prenatal care proposed that “the preconceptional visit may be the single most important health care visit when viewed in the context of its effect on pregnancy outcome.”40 Various risk factors that negatively impact maternal and child oral health, should be identified, treated, and/or modified prior to conception. Risk assessment and preconceptional counseling can easily be incorporated into routine dental care.
The American Academy of Periodontology (AAP) has also issued a statement regarding the periodontal management of the pregnant patient.41 The AAP suggests that women should be encouraged to achieve a high level of oral hygiene prior to becoming pregnant and throughout their pregnancies.
2. Educate the patient regarding the possible impact of periodontal infection on pregnancy outcomes. This is a critical component of preconceptional counseling. Pregnancy is a very teachable time. A number of educational tools are available from the AAP for women and expectant mothers regarding the importance of maintaining oral health. Visit www.perio.org for more information.
4. Perform periodontal treatment as necessary during pregnancy. Periodontal treatment during pregnancy is safe,38 however, the AAP recommends that treatment be scheduled early in the second trimester.41
5. Consider whether consultation with the patient’s medical care provider would be beneficial. This may help determine whether other risk factors such as gestational diabetes or high blood pressure are present. Reporting the periodontal status of the patient and any proposed treatment to the medical care provider may also be beneficial.41
6. Encourage pregnant patients to adhere to the recommendations of their prenatal medical care providers, including frequency of prenatal care, vitamin supplementation, nutrition, weight management, etc. Further information about the recommendations of prenatal care providers can be found in the document “Oral Health Care During Pregnancy and Early Childhood: Practice Guidelines,” developed by the New York State Department of Health, and is available at: www.health.state.ny.us/publications/0824.pdf.
7. Increase awareness within the medical community of the threat periodontal disease may pose to pregnancy. Build a network of prenatal care providers who are willing to collaborate in the comanagement of pregnant patients. Facilitate medical care providers’ referral of pregnant patients to dental providers for oral health consultation. An excellent referral form “Consultation for Pregnant Women to Receive Oral Health Care,” developed by the New York State Department of Health, is available at: www.health.state.ny.us/publications/0824.pdf.
8. Carefully observe pregnant patients for signs or symptoms of preeclampsia. Preeclampsia is often asymptomatic; its detection depends on recognition of signs and symptoms.20
REFERENCES
- Offenbacher S, Katz V, Fertik G, et al. Periodontal infection as a possible risk factor for preterm low birth weight. J Periodontol. 1996;67(10 Suppl):1103-1113.
- Barak S, Oettinger-Barak O, Machtei EE, et al. Evidence of periopathogenic microorganisms in placentas of women with preeclampsia. J Periodontol. 2007;78:670-676
- Boggess KA, Lieff S, Murtha AP, et al. Maternal periodontal disease is associated with an increased risk for preeclampsia. Obstet Gynecol. 2003;101:227-231.
- Boggess KA, Edelstein BL. Oral health in women during preconception and pregnancy: implications for birth outcomes and infant oral health. Matern Child Health J. 2006;10(Suppl):S169-S174.
- Canakci V, Canakci CF, Canakci H, et al. Periodontal disease as a risk factor for pre-eclampsia: a case control study. Aust N Z J Obstet Gynaecol. 2004;44:568-573.
- Canakci V, Canakci CF, Yildirim A, et al. Periodontal disease increases the risk of severe pre-eclampsia among pregnant women. J Clin Periodontol. 2007;34:639-645.
- Castaldi JL, Bertin MS, Giménez F, et al. Periodontal disease: Is it a risk factor for premature labor, low birth weight or preeclampsia? Rev Panam Salud Publica. 2006;19:253-258. Spanish.
- Conde-Agudelo A, Villar J, Lindheimer M. Maternal infection and risk of preeclampsia: systematic review and metaanalysis. Am J Obstet Gynecol. 2008 ;198:7-22.
- Contreras A, Herrera JA, Soto JE, et al. Periodontitis is associated with preeclampsia in pregnant women. J Periodontol. 2006;77:182-188.
- Cota LO, Guimarães AN, Costa JE, et al. Association between maternal periodontitis and an increased risk of preeclampsia. J Periodontol. 2006;77:2063-2069.
- Herrera JA, Parra B, Herrera E, et al. Periodontal disease severity is related to high levels of C-reactive protein in preeclampsia. J Hypertens. 2007 ;25:1459-1464.
- Khader YS, Jibreal M, Al-Omiri M, et al. Lack of association between periodontal parameters and preeclampsia. J Periodontol. 2006;77:1681-1687.
- Kunnen A, Blaauw J, van Doormaal JJ, et al. Women with a recent history of early-onset pre-eclampsia have a worse periodontal condition. J Clin Periodontol. 2007;34:202-207.
- Kunnen M, Boggess K, Moss K, et al. Maternal periodontal disease, systemic inflammation, and risk for preeclampsia. Am J Obstet Gynecol. 2008;198:389.
- Siqueira FM, Cota LO, Costa JE, et al. Maternal periodontitis as a potential risk variable for preeclampsia: a case-control study. J Periodontol. 2008;79:207-215.
- Siqueira JN. Studies suggest an association between maternal periodontal disease and pre-eclampsia. Evid Based Dent. 2008;9:46-47.
- Xiong X, Buekens P, Vastardis S, et al. Periodontal disease and pregnancy outcomes: state-of-the-science. Obstet Gynecol Surv. 2007;62:605-615.
- Ruma M, Boggess K, Moss K, et al. Maternal periodontal disease, systemic inflammation, and risk for preeclampsia. Am J Obstet Gynecol 2008;198:389.
- Canakci V, Yildirim A, Canakci CF, et al. Total anti oxidant capacity and antioxidant enzymes in serum, saliva, and gingival crevicular fluid of pre eclamptic women with and without perio dontal disease. J Periodontol. 2007;78:1602-1611.
- MayoClinic.com. Pregnancy, Preeclampsia. Available at: www.mayoclinic.com/health/preeclampsia/DS00583. Accessed September 16, 2008.
- Minkoff H. Prematurity: infection as an etiologic factor. Obstet Gynecol 1983;62:137–144.
- Gibbs RS, Romero R, Hillier SL, et al. A review of premature birth and subclinical infection. Am J Obstet Gynecol 1992;166: 1515–1528.
- Gibbs RS. The relationship between infections and adverse pregnancy outcomes: an overview. Ann Periodontol 2001;6:153–163.
- Redman CW, Sacks GP, Sargent IL. Preeclampsia: an excessive maternal inflammatory response to pregnancy. Am J Obstet Gynecol 1999;180:499–506.
- von Dadelszen P, Magee LA. Could an infectious trigger explain the differential maternal response to the shared placental pathology of preeclampsia and normotensive intrauterine growth restriction. Acta Obstet Gynecol Scand 2002;81:642–648.
- Goldenberg RL, Culhane JF, Iams JD, et al. Epidemiology and causes of PTB. Lancet. 2008;371:75-84.
- Herrera JA, Chaudhuri G, Lopez-Jaramillo P. Is infection a major risk factor for preeclampsia? Med Hypotheses 2001;57:393–397.
- Romero R, Espinoza J, Kusanovic JP, et al. The preterm parturition syndrome. Br J Obstet Gynaecol. 2006;113:17-42.
- Ferguson JE 2nd, Hansen WF, Novak KF, et al. Should we treat periodontal disease during gestation to improve pregnancy outcomes? Clin Obstet Gynecol. 2007;50: 454-467.
- Offenbacher S. Role of infection as systemic inflammatory stressor and impact on maternal-infant health. Presented at: Global Oral and Systemic Health Summit: Present Evidence and Future Directions. Basking Ridge, NJ: July 24, 2006.
- Romero BC, Chiquito CS, Elejalde LE, et al. Relationship between periodontal disease in pregnant women and the nutritional condition of their newborns. J Periodontol. 2002;73:1177-1183.
- Hack M, Taylor HG, Drotar D, et al. Chronic conditions, functional limitations, and special health care needs of schoolaged children born with extremely low-birth-weight in the 1990s. JAMA. 2005;294:318-325.
- Offenbacher S, Riche EL, Barros SP, et al. Effects of maternal Campylobacter rectus infection on murine placenta, fetal and neonatal survival, and brain development. J Periodontol. 2005;76 (Suppl):2133-2143.
- Lieff S, Boggess KA, Murtha AP, et al. The oral conditions and pregnancy study: periodontal status of a cohort of pregnant women. J Periodontol. 2004;75:116-1126.
- Oettinger-Barak O, Barak S, Ohel G, et al. Severe pregnancy complication (preeclampsia) is associated with greater periodontal destruction. J Periodontol. 2005;76:134-137.
- Ruma M, Boggess K, Moss K, et al. Maternal periodontal disease, systemic inflammation, and risk for preeclampsia. Am J Obstet Gynecol. 2008;198:389.
- Belo L, Santos-Silva A, Rocha S, et al. Fluctuations in C-reactive protein concentration and neutrophils activation during normal pregnancy. Eur J Obstet Reprod Biol. 2005;123:46-51.
- Michalowicz BS, Hodges JS, DiAngelis, AJ, et al. Treatment of periodontal disease and the risk of PTB. N Engl J Med. 2006;355:1885-1894.
- Goldenberg R, Culhane J. PTB and periodontal disease. N Engl J Med. 2006;355:1925-1927.
- Department of Health and Human Services. Healthy People 2000: National Health Promotion and Disease Prevention Objectives. Washington, DC: US Department of Health and Human Services, Public Health Service. 1990 DHHS publication no (PHS) 91-50212
- American Academy of Periodontology. Statement Regarding Periodontal Management of the Pregnant Patient. Available at: www.perio.org/resources-products/pdf/44-pregnancy.pdf. Accessed August 20, 2008.
- American Academy of Periodontology Statement on Risk Assessment. Available at: www.perio.org/resources-products/pdf/risk-assessment.pdf. Accessed August 20, 2008.
From Dimensions of Dental Hygiene. October 2008; 6(10):32-35.