 MAGICMINE/ISTOCK/GETTY IMAGES PLUS
MAGICMINE/ISTOCK/GETTY IMAGES PLUS
A Closer Look at the AHA’s Antibiotic Prophylaxis Guidelines
In order to prevent infective endocarditis, oral health professionals need to remain up to date on the American Heart Association guidelines.
This course was published in the December 2021 issue and expires December 2024. The author has no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Define infective endocarditis (IE).
- Discuss the rationale behind the American Heart Association’s update to the antibiotic prophylaxis regimen.
- Explain the role of oral health professionals in preventing cardiac infection.
In May 2021, the American Heart Association (AHA) updated its antibiotic prophylaxis guidelines for the prevention of infective endocarditis (IE).1 As some dental and dental hygiene procedures may increase the risk of this potentially life-threatening infection in at-risk patients, oral health professionals must have a thorough understanding of this serious, albeit rare infection of the heart and understand appropriate precautions for treatment. This article focuses on the AHA’s guidelines; however, oral health professionals should also consider the IE guidelines from the American Academy of Orthopædic Surgeons.
IE is an infection of the inner lining of the heart, frequently involving the heart valves. Sequela of an IE infection may lead to heart failure, stroke, heart valve damage that requires valve replacement, and death.1 There are two types of IE: native valve endocarditis and prosthetic valve endocarditis. This article will focus on native valve endocarditis as it is more common. The prevalence of prosthetic valve endocarditis, however, has grown over the past decade due to an increase in the number of surgical replacements of damaged valves. Requiring complex medical management, prosthetic valve endocarditis has higher mortality and morbidity rates than native valve endocarditis.2 Patients with prosthetic valve endocarditis may be better served in a hospital-based dental setting.
A significant public health issue, IE affects three to 10 individuals per 100,000 globally with a 20% mortality rate.3 IE is most common among persons who inject drugs (PWID).4 Also at increased risk for IE are patients with cardiovascular disease who develop nosocomial infections and patients undergoing dental procedures that produce significant bacteremia.
DAISUKE KOYA, KAZUYUKI SHIBUYA, RYUICHI KIKKAWA AND MASAKAZU HANEDA., CC BY 2.0
<HTTPS://CREATIVECOMMONS.ORG/LICENSES/BY/2.0>, VIA WIKIMEDIA COMMONS
Etiology and Pathophysiology of Infective Endocarditis
A combination of factors, including an injured (roughened) endothelial lining, damaged or roughed heart valve, host immune response, and bacteremia in the systemic circulation, can lead to IE. IE cannot occur unless the heart has a nonbacterial thrombotic endocarditis (NBTE) lesion. A NBTE lesion forms in the presence of inflammation or injury to the heart’s endothelial cells or valves, which causes an immune response. This response results in the platelets and fibrins adhering to the area of injury, causing vegetations or NBTE. When the integrity of the dermis is disrupted via an open skin wound, needle puncture, or surgical procedure, bacteremia may result, possibly leading to a NBTE lesion. Bacteremia may also originate from an internal source such as an abscess or dental infection.5 Due to bacteremia, an NBTE lesion in the heart can transition from noninfected tissue to infected tissue, causing IE.
IE most frequently impacts the left side of the heart (70%), especially the mitral or aortic valves. IE occurs in the right side of the heart involving the tricuspid or pulmonic valve approximately 10% to 20% of the time.6 The only exception to this pattern is among PWID, who most often present with right side IE due to a needle skin puncture with Staphylococcus aureus as the etiologic pathogen.
Some bacteria are more likely to cause IE. Approximately 90% of community-acquired cases are caused by streptococci, staphylococci, or enterococci species.7 S. aureus is a normal inhabitant of the skin and mucous membranes. When S. aureus enters the bloodstream due to drug injection or stab wound, it may cause IE in at-risk patients.8
The streptococci species is the most common cause of native valve IE among those who are not intravenous drug users. Viridans streptococci, a heterogeneous group of alpha-hemolytic streptococci, are part of the normal flora of the mouth, and play a role in the formation of dental caries (Streptococcus mutans, S. sanguinis) and pericoronitis. They also cause subacute IE.9 Viridans streptococci are responsible for approximately 40% to 60% of native valve IE.9 They are also the most common dental-derived pathogen to cause IE.10
Another virulent but rare (1% to 3%) IE pathogen is a group of Gram-negative bacteria composed of Haemophilus, Aggregatibacter, Cardiobacterium hominis, Eikenella corrodens, and Kingella species (HACEK).11 HACEK organisms are a normal part of the human microbiota living in the oral-pharyngeal region.
Aggregatibacter actinomycetemcomitans is a Gram-negative bacterium linked to periodontal diseases. It is found in 90% of localized aggressive periodontitis cases and 30% to 50% of severe adult periodontitis.12 This is important information for dental hygienists, as the presence of periodontal diseases in patients at risk for IE will increase the virulence of their bacteremia.
IE is classified as subacute and acute. Subacute IE is related to dental treatment, and typically presents a week to several months after a procedure. It is predominantly caused by S. viridans. On the other hand, acute IE is a bacterial endocarditis—most common among PWID—caused by an infection or port of entry. Most commonly caused by S. aureus, acute IE is a significant public health problem. PWID with acute IE are typically young, and present with native valve infections 90% of the time.13 An acute IE related to intravenous drug use is more complex to manage.
![TABLE 1. Indications for Antimicrobial Prophylaxis]() Medical Evaluation and Diagnosis
Medical Evaluation and Diagnosis
Physicians are trained to make an IE diagnosis, which is based on medical history and the presence of clinical symptoms. Patients with a history of intravenous drug use, valvular disorders, or recent heart procedures are at high risk for IE.
Clinical manifestations of IE may include the following: fever, night sweats, fatigue, Olser nodes (painful, red blisters on the hands and feet), Janeway lesions (irregular, nontender hemorrhagic macules found on the hands and toes), splinter hemorrhages (thin, reddish-brown lines under the nails), conjunctival petechiae (broken capillaries in the eyes) and/or hemorrhages, Roth spots (retinal hemorrhages), and nail clubbing (enlarged fingertips with the nails curving around the tips).14 Olser nodes and Janeway lesions are cutaneous nodules associated with an IE infection. Roth spots can be seen with other medical conditions, but are most often associated with IE, detected in 80% of subacute bacterial endocarditis.15
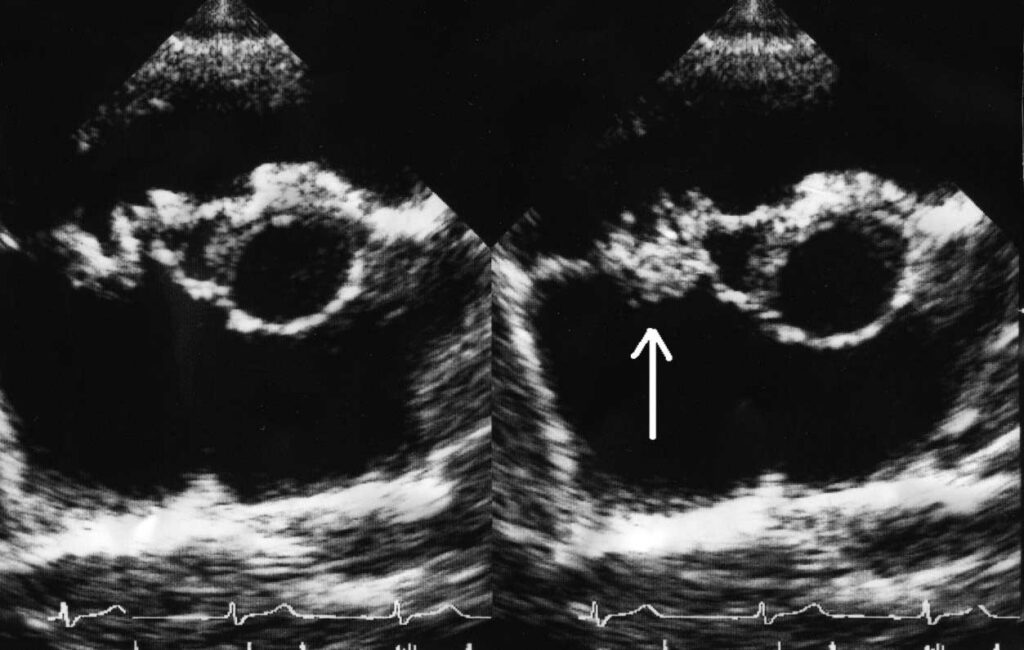
The major Duke criteria for IE diagnosis include a positive blood culture, presence of common IE-related microorganisms from two different blood cultures, signs of endocardial involvement, and new valvular regurgitation (mitral valve that does not close properly).6 Blood cultures evaluate the volume of the bacteremia and type of organisms present. An echocardiogram is done to assess whether a patient has vegetations on the heart valve (Figure 1, page 33). Minor Duke criteria for IE encompass predisposing heart conditions or intravenous drug use; fever; vascular phenomena including embolic events; immunologic phenomena; microbiologic findings; and echocardiographic evidence.13
Once a diagnosis is confirmed, an appropriate intravenous antibiotic regimen will be started. Patients with native valve endocarditis are given 15 mg/kg to 20 mg/kg of vancomycin intravenously every 8 hours to 12 hours.6
American Heart Association Guidelines
In 1955, the AHA recommended antibiotic prophylaxis for bacterial endocarditis.16 Since then, as empirical information has been analyzed and reviewed, the AHA has updated its antibiotic prophylaxis guidelines approximately eight times in an effort to prevent IE. In the mid-1980s, concerns arose surrounding the overuse of antibiotics, which was causing allergic reactions and microbial resistance.16 Due to these concerns, the antibiotic clindamycin was introduced as an antibiotic prophylaxis for people allergic to penicillin. According to the American Dental Association, the antibiotic prophylaxis is recommended for all dental procedures that involve “manipulation of gingival tissue or the periapical region of the teeth, or perforation of the oral mucosa.”17
According to the AHA’s 2021 guidelines, antibiotic prophylaxis continues to be the recommended course of treatment for patients at high risk for IE (Table 1 and Table 2). However, in the AHA’s most recent update, clindamycin is no longer recommended as a prophylactic antibiotic to prevent IE. The rationale for this change is that clindamycin is more likely to cause negative reactions compared to other antibiotics.1 A single dose of clindamycin can cause a Clostridioides difficile infection, which can lead to serious complications or death.1 Up to 15% of C. difficile infections may be attributable to antibiotics prescribed for a dental procedure.1
The AHA recommends amoxicillin or doxycycline antibiotic prophylaxis. Amoxicillin’s recommended oral dose in adults is 2 g and 50 mg/kg for children.1 Patients who are allergic to penicillin or ampicillin may be prescribed cephalexin (2 g for adults and 50 mg/kg for children); azithromycin or clarithromycin (500 mg for adults and 15 mg/kg for children); or doxycycline (100 mg for adults, 2.2 mg/kg for children weighing less than 45 kg and 100 kg for children weighing more than 45 kg).1 Antibiotics should be taken 30 minutes to 60 minutes prior to beginning dental treatment. Patients who do not take their prescribed antibiotic before the dental appointment are allowed to take the medication up to 2 hours after treatment is provided.
![TABLE 2. Antimicrobial Prophylaxis Treatment Options]() The Role of Oral Health Professionals
The Role of Oral Health Professionals
Dental hygienists play an important role in decreasing the prevalence of IE by educating patients on the best biofilm control practices tailored for their needs. S. viridans-related IE is more likely to be caused by bacteremia originating in the oral cavity due to daily activities such as chewing, brushing, and flossing in inflamed gingival tissue, than from a dental procedure.1 Transient bacteremia is frequently caused by chewing food (7% to 51%), toothbrushing (20% to 68%), using a wooden toothpick (20% to 40%), and water flossing (7% to 50%).18 The AHA’s 2007 statement on IE noted that bacteremia from chewing food is more likely among patients with periodontal diseases as higher bacteria counts are present.18
For dental hygienists, the most effective way to prevent IE is to support patients in maintaining their oral health.1 To reduce plaque biofilm and inflammation, clinicians can recommend power toothbrushes,19 floss or other interdental aids, and antimicrobial mouthrinses. Good oral hygiene will decrease S. viridans more effectively than a single dental procedure. Controlling oral inflammation is a better strategy to prevent IE than prescribing antibiotics.
Oral health professionals should be familiar with current information regarding the oral-systemic connection, including which practices can improve health or increase the risk of disease. First, the medical history should be used to identify patients who may be at risk for an oral bacteremia. Second, dental hygienists can implement motivational interviewing to promote positive oral health practices. Third, appropriate recare intervals to decrease the formation of subgingival biofilms should be recommended.
Research shows that maintaining oral health is more important in reducing the risk of subacute IE in the long-term than preprocedural antibiotic prophylaxis.1 However, in situations when it is indicated, antibiotic prophylaxis is essential in preventing serious illness and possibly death. Therefore, oral health professionals should be well-informed of the current recommendations and important changes concerning antibiotic prophylaxis.
References
- Wilson WR, Gewitz M, Lockhart PB, et al. Prevention of viridans group streptococcal infective endocarditis: a scientific statement from the American Heart Association. Circulation. 2021;143:e963–e978.
- Luehr M, Bauernschmitt N, Peterss S, et al. Incidence and surgical outcomes of patients with native and prosthetic aortic valve endocarditis. Ann Thorac Surg. 2020;110:93–101.
- Montano TCP, Wanderley MIA, Sampaio RO, et al. Cardiological, microbiological, and dental profiles of Brazilian ˇpatients who developed oral bacteria-related endocarditis. Oral Surg, Oral Med, Oral Pathol Oral Radiol. 2021;132:418–425.
- Pericàs JM, Llopis J, Athan E, et al. Prospective cohort study of infective endocarditis in people who inject drugs. J Am Coll Cardiol. 2021;77:544–555.
- Chu VH. Pathogenesis of vegetation formation in infective endocarditis. Available at: uptodate.com/contents/pathogenesis-of-vegetation-formation-in-infective-endocarditis. Accessed November 15, 2021.
- Armstrong GP. Infective endocarditis. Available at: merckmanuals.com/professional/cardiovascular-disorders/endocarditis/infective- endocarditis#. Accessed November 15, 2021.
- Fowler VG Jr, Bayer A. Infective endocarditis. In: Goldman L, Ausiello D, eds. Cecil Medicine. 23rd ed. Philadelphia: Saunders; 2008:537–548.
- Taylor TA, Unakal CG. Staphylococcus aureus. Available at: ncbi.nlm.nih.gov/books/NBK441868/. Accessed November 15, 2021.
- Birlutiu V, Birlutiu RM, Costache VS. Viridans streptococcal infective endocarditis associated with fixed orthodontic appliance managed surgically by mitral valve plasty: A case report. Medicine (Baltimore). 2018;97:e11260.
- Raja M, Ummer F, Dhivakar CP. Aggregatibacter actinomycetemcomitans—a tooth killer? J Clin Diagn Res. 2014;8:ZE13-ZE16.
- Bläckberg A, Morenius C, Olaison L. et al. Infective endocarditis caused by HACEK group bacteria—a registry-based comparative study. Eur J Clin Microbiol Infect Dis. 2021;40, 1919–1924.
- Dijkstra GW, Glaudemans AWJM, Erba PA, et al. Relationship between 18F-FDG uptake in the oral cavity, recent dental treatments, and oral inflammation or infection: a retrospective study of patients with suspected endocarditis. Diagnostics (Basel). 2020;10:625.
- Pericàs JM, Llopis J, Athan E, et al. Prospective cohort study of infective endocarditis in people who inject drugs. J Am Coll Cardiol. 2021;77:544–555.
- Topan A, Carstina D, Slavocivic A, et al Assessment of the Duke criteria for the diagnosis of infective endocarditis after 20 years. A analysis of 241 cases. Clujul Med. 2015;88:321–326.
- Ruddy SM, Bergstrom R, Tivakaran VS. Roth spots. Available at: ncbi.nlm.nih.gov/books/NBK482446/. Accessed November 15, 2021.
- Pallasch TJ. Antibiotic prophylaxis. Endodontic Topics. 2003;4(1):46–59.
- American Dental Association. Antibiotic Prophylaxis Prior to Dental Procedures. Available at: ada.org/en/member-center/oral-health-topics/antibiotic-prophylaxis. Accessed November 15, 2021.
- Wilson W, Taubert KA, Gewitz M, et al. Prevention of infective endocarditis: guidelines from the American Heart Association: a guideline from the American Heart Association rheumatic fever, endocarditis, and Kawasaki disease committee, council on cardiovascular disease in the young, and the council on clinical cardiology, council on cardiovascular surgery and anesthesia, and the quality of care and outcomes research interdisciplinary working group. Circulation. 2007;116:1736–1754.
- Yaacob M, Worthington HV, Deacon SA, et al. Powered vs manual toothbrushing for oral health. Cochrane Database Syst Rev. 2014;2014:CD002281.
From Dimensions of Dental Hygiene. December 2021;19(12)32-35.





