 Jon Fraze
Jon Fraze
Caring for Patients Who Abuse Methamphetamine
The nonmedical use of this drug significantly impacts oral health.
This course was published in the November 2016 issue and expires November 30, 2019. The authors have no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Discuss the increase in methamphetamine abuse and prevalence of oral health issues as a comorbidity in long-term users.
- Describe the potential oral manifestations of methamphetamine use.
- List therapeutic approaches to address the oral health consequences of the nonmedical use of this drug.
- Explain the physiological effects of this stimulant and the appropriate response when patients present with signs of use.
According to a 2008 study by the United Nations, approximately 25 million individuals around the world use methamphetamine.1 Between 2008 and 2014, however, the number of methamphetamine users more than doubled. Worldwide, as many as 52 million people between the ages of 15 and 64 are estimated to have used amphetamine-type stimulants for nonmedical purposes in 2014.2 It is the second most widely abused recreational drug (following cannabis).2
Methamphetamine can be snorted, smoked, ingested, injected and inserted rectally.3 It exerts a powerful effect through its activation of the dopamine, norepinephrine, and serotonin systems. Its highly addictive properties are associated with the drug’s prolonged half-life (10 hours to 12 hours) and long duration of action, as well as its elevation of dopamine levels.2
A group heavily impacted by methamphetamine use is men who have sex with men; however, the drug seems to appeal equally to men and women.4 Research indicates that women are more likely than men to use methamphetamine for weight loss and to relieve symptoms of depression.4 A 2010 study by Shetty et al5 on the dental effects of methamphetamine use reported dental disease—at 41.3%—as one of the most prevalent comorbidities in long-term methamphetamine users.
A more recent investigation by Shetty et al6 notes that cigarette smokers who abuse methamphetamine were nearly three times as likely as nonsmokers to experience untreated caries, and that, overall, posterior teeth (especially occlusal surfaces) were most affected. The researchers suggest the patterns of root caries in methamphetamine users are distinct and potentially could be used to identify covert drug users. Additionally, they report that while 37% of adults ages 35 to 49 in the US have periodontitis, more than 89% of methamphetamine users present with periodontitis.
Caries risk is highly elevated among chronic users of methamphetamine due to a high prevalence of xerostomia; heavy bruxing, leading to exposed tooth surfaces and fractured restorations; and poor diet and nutrition. Methamphetamine use is associated with sugared soda consumption. Murphy et al7 studied 541 methamphetamine users and found their soda consumption was higher than the adult population average, with longer methamphetamine use correlated with higher rates of sugared soda consumption. Users with high soda consumption rates were also more likely to experience more oral health problems than users without high soda-consumption rates.7
Formication, or the sensation that insects are crawling under the skin, is common among chronic methamphetamine users. This causes users to frequently scratch at their skin, resulting in significant abrasions. These skin lesions often appear on the face and may be noted during routine dental examinations.8
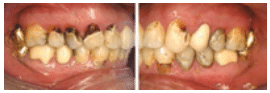
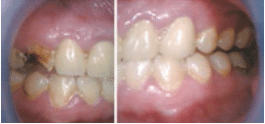
Chronic methamphetamine use is also likely to damage the stomatognathic system. The direct consequences of abuse—including xerostomia, bruxism, decreased salivary buffering capacity, and accompanying risk factors, such as consumption of other toxic substances—may contribute to an overall neglect of dental care, significantly increasing oral health risks9 (Figure 1A to Figure 3B).
PATIENT MANAGEMENT
Tatlock et al10 suggest the following protocol when treating patients with methamphetamine-induced oral conditions:
- Include questions about drug use when taking a medical history
- Consider the potential for drug interactions to rule out possible contraindications
- Be aware that administering nitrous oxide or prescribing narcotics could endanger the sobriety of a recovering patient
- Offer education about regular checkups and oral hygiene (eg, flossing, interdental cleaning, and brushing with a fluoride toothpaste for 2 minutes at least twice daily)
- Suggest a mouthguard for patients who brux
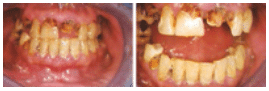
FIGURE 3A and FIGURE 3B. Oral manifestations in the advanced stages of metham phetamine abuse include: (3A) teeth broken off at the gingival margin; and (3B) grayish- brown dentition with enamel that is reduced to a soft, leathery texture, along with gingivitis and acute periodontitis
FIGURE 1A TO FIGURE 3B COURTESY OF MITCHELL A. GOODIS, DDS
Discussions with patients about the oral health risks associated with nonmedical amphetamine use should be tactful and nonjudgmental (Table 1). In addition, patient education efforts should explain the benefits of consuming a balanced diet and drinking water rather than sugary beverages or diuretics (such as caffeine and alcohol) that can exacerbate xerostomia. Table 2 offers a sampling of methamphetamine educational resources.
It is difficult, if not impossible, to mitigate the oral effects of methamphetamine if substance abuse is ongoing. For patients in all stages of methamphetamine use, clinicians should explain optimal short-term and long-term treatment options. Patients are often embarrassed by their teeth and appreciate guidance for esthetic and functional improvements. Treatment of cavitated lesions using glass ionomer as an interim restoration—with or without removal of decay—can slow or possibly stop lesion progression. Stabilizing lesions in this manner can help see the patient through a drug addiction rehabilitation period with reasonable esthetic and functional improvement.
If a patient is in recovery, or has expressed a desire to save his or her teeth, the authors present the following method for caries management using silver diamine fluoride (SDF). In 2014, the US Food and Drug Administration cleared SDF for marketing as a dental desensitizing agent, and many state regulatory boards have approved its application by dental auxiliaries. This agent has been used off-label to arrest caries, primarily in high-risk populations. Although such use has been shown to be effective and safe,11,12 as with all off-label therapy, clinicians are advised to exercise professional judgment.
Methamphetamine users experience rampant decay for a variety of reasons. The drug contributes to xerostomia, and, among this patient population, oral health is further compromised by the tendency to neglect proper nutrition, oral hygiene, and regular dental visits.9,10 Individuals subject to drug addiction often do not have the financial resources for extensive dental reconstruction. In these cases, SDF offers a potential first step for oral rehabilitation by stabilizing the progression of caries lesions until recovery and rehabilitation can begin.
The first step in treatment is to engage the patient and obtain consent with the understanding that SDF is not definitive care, but, rather, the beginning of care. Clinicians should also explain that SDF will darken cavitated surfaces (Figure 4).13 Improvement in the appearance of teeth can begin when decay is arrested and the patient is ready to move forward and participate actively in recovery, leading to improved oral health. If resources are available, esthetic concerns can be addressed with conventional dental services (eg, direct or indirect restorations and fixed or removable prostheses).
Teeth to be treated must be free of symptoms of pulpitis, signs of draining fistula, or pulpal exposure. One drop to two drops of SDF are dispensed into a plastic dappen dish. The affected dentition is polished with prophylaxis paste, or thoroughly cleaned with a toothbrush. The teeth are then isolated with cotton rolls and dried. Using a microbrush, the solution is applied to the affected area for 1 minute, and the excess is rinsed off. Most patients report a metallic or bitter taste during treatment, but there is no other discomfort.13 Patients are instructed not to eat, drink, or brush for 1 hour following treatment.
For severe caries, some protocols recommend reapplication of SDF after 2, 4, 6, and 8 weeks.14 Although other protocols recommend fewer initial treatments,11 consensus is that treatment should continue until lesions are arrested (or restored), and repeated biannually or annually to maintain results.
Care must be used in handling SDF because it will stain surfaces; consequently, plastic wrap or other barrier products are recommended to protect operatory surfaces. This agent should not be splashed onto clothing or skin. If it contacts skin, SDF will leave a dark stain, which will exfoliate in 1 week to 2 weeks.11
REMINERALIZATION AND REFERRAL
Because methamphetamine users are subject to high caries risk, remineralization therapies are often indicated to prevent the development of new lesions or progression of existing decay. Thus, concentrated fluoride products may be prescribed as part of the treatment plan. Clinicians can refer to the caries management by risk assessment—or CAMBRA—protocol for further information about caries management strategies.15
As noted, clinicians should be mindful that mitigating oral health problems when methamphetamine use is ongoing is difficult. If a patient is abusing methamphetamine, referral to a substance abuse treatment center should be offered.10,16 Dental offices should maintain a list of local and national resources for this purpose. Oral health professionals need to have written information available detailing addiction support services.16
Whenever possible, dental teams should coordinate with the patient’s physician so that he or she may participate in the referral. If the patient does not wish to discuss illicit drug use with oral health professionals, the team should document findings in an objective, nonjudgmental manner. The patient should also be encouraged to return to the practice for follow-up care.
TREATMENT PRECAUTIONS
For the safety of patients and staff, all team members should be able to identify the signs of an intoxicated methamphetamine user. When taking the drug, users report feelings of euphoria, an abundance of energy, increased motivation, anxiety, increased self-confidence, and decreased appetite. Physiological effects include elevated heart rate, increased blood pressure, hypertension, hyperthermia, pupil dilatation, sweating, insomnia, dermatologic reactions, and psychomotor agitation.4,8,17 It is not unusual for individuals who abuse methamphetamine to engage in binge episodes in which they use large amounts of the drug for several days, and neglect proper dietary, hygienic and personal care.
If the clinician suspects that a patient is intoxicated due to signs of recent use—such as repetitive physical activity, rapid speech, shortness of breath, irritability, or bruxing—dental treatment should not be administered for physiological reasons.16 Furthermore, due to the patient’s mental functioning, he or she may not be able to make informed decisions regarding treatment or consent. In these situations, the clinician should inform the patient in a calm and clear manner that, for safety reasons, the team will not be able to provide treatment at this time. Table 3 provides guidelines and precautions for working with intoxicated patients.16
CONCLUSION
Drug addiction has severe psychosocial and physical consequences. This is especially true with methamphetamine users, who are subject to a variety of oral health issues. Through therapeutic interventions, including glass ionomer, SDF, and concentrated fluoride—and by offering patient education and referral to substance abuse resources—dental professionals can help recovering users by restoring their oral health.
REFERENCES
- United Nations Office on Drugs and Crime. Annual Report 2008. Available at: unodc.org/ documents/about-unodc/AR08_WEB.pdf. Accessed October 5, 2016.
- Radfar SR, Rawson RA. Current research on methamphetamine: Epidemiology, medical and psychiatric effects, treatment, and harm reduction efforts. Addict Health. 2014;6:146–154.
- Shrem MT, Halkitis PN. Methamphetamine abuse in the United States: contextual, psychological and sociological considerations. J Health Psych. 2008;13:669–679.
- Gonzales R, Mooney L, Rawson RA. The methamphetamine problem in the United States. Annu Rev Public Health. 2010:31:385–398.
- Shetty V, Mooney LJ, Zigler CM, Belin TR, Murphy D, Rawson R. The relationship between methamphetamine use and increased dental disease. J Am Dent Assoc. 2010;141:307–318.
- Shetty V, Harrell L, Murphy DA, et al. Dental disease patterns in methamphetamine users: Findings in a large urban sample. J Am Dent Assoc. 2015;146:875–885.
- Murphy DA, Harrell L, Fintzy R, Vitero S, Gutierrez A, Shetty V. Soda consumption among methamphetamine users in the USA: impact on oral health. Oral Health Prev Dent. 2016;14:227–234.
- Rusyniak DE. Neurologic manifestations of chronic methamphetamine abuse. Psychiatr Clin North Am. 2013;36:261–275.
- Rommel N, Rohleder NH, Wagenpfeil S, et al. The impact of the new scene drug “crystal meth” on oral health: a case-control study. Clin Oral Invest. 2016;20:469–475.
- Tatlock C, Wagner S, Williams N. Meth & Oral Health. A Guide for Dental Professionals. Available at: securiandental.com/cm_files/pdf/050208_ SecurianMethBookletFinal.pdf. Accessed October 5, 2016.
- Featherstone JD, Horst JA. Fresh approach to caries arrest in adults. Decisions in Dentistry. 2015;1(1):36–44.
- Horst JA, Ellenikiotis H, Milgrom PM. UCSF protocol for caries arrest using silver diamine fluoride: rationale, indications, and consent. J Calif Dent Assoc. 2016;44:16–28.
- Fa BA, Horst JA, Hirsch JP, Duffin S, Wong A, Young DA. Caries arrest with silver diamine fluoride. Decisions in Dentistry. 2016;2(10):48–51.
- Duffin S. Back to the future: the medical management of caries introduction. J Calif Dent Assoc. 2012;40:852–858.
- Featherstone JD. The caries balance: contributing factors and early detection. J Calif Dent Assoc. 2003;31:129–133.
- Jenner L, Lee N. Treatment Approaches for Users of Methamphetamine: A Practical Guide for Frontline Workers. Available at: nationaldrugstrategy.gov.au/internet/drugstrategy/publishing.nsf/Content/8D2E281FAC2346BBCA25764D007D2D3A/$File/tremeth.pdf. Accessed October 5, 2016.
- Cruickshank CC, Dyer KR. A review of the clinical pharmacology of methamphetamine. Addiction. 2009;104:1085–1099.
From Dimensions of Dental Hygiene. November 2016;14(11):36–39.




[…] https://dimensionsofdentalhygiene.com/article/Caring-for-Patients-Who-Abuse-Methamphetamine/ […]
[…] https://dimensionsofdentalhygiene.com/article/Caring-for-Patients-Who-Abuse-Methamphetamine/ […]
[…] https://dimensionsofdentalhygiene.com/article/Caring-for-Patients-Who-Abuse-Methamphetamine/ […]
[…] https://dimensionsofdentalhygiene.com/article/Caring-for-Patients-Who-Abuse-Methamphetamine/ […]
[…] https://dimensionsofdentalhygiene.com/article/Caring-for-Patients-Who-Abuse-Methamphetamine/ […]