 SPOTMATIK/ISTOCK/GETTY IMAGES PLUS
SPOTMATIK/ISTOCK/GETTY IMAGES PLUS
Caries Risk Assessment in Children With Special Needs
Understanding condition-specific caries risk factors can help clinicians identify successful prevention strategies.
By Elise Sarvas, DDS, MSD, MPH
This course was published in the June 2017 issue and expires June 2020. The author has no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Discuss factors and conditions that affect caries risk in children with special health care needs.
- Explain the role that ear, nose, and throat, as well as gastrointestinal conditions play in caries risk in this patient population.
- Identify preventive measures that can help mitigate caries risk in children with special health care needs.
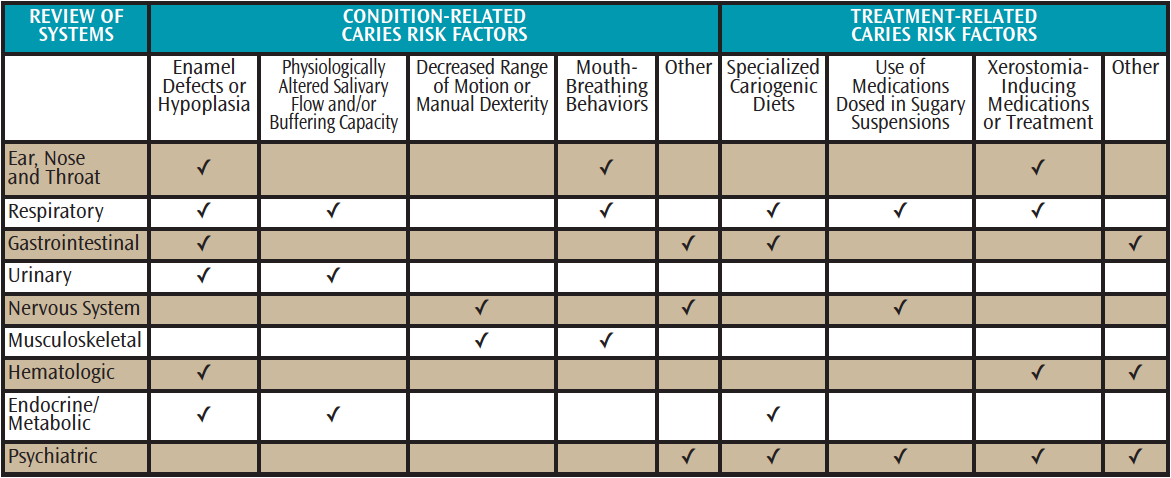
Risk assessment is a key component in the prevention and treatment of dental caries.1Multiple risk assessment tools are available for practitioners, and all place children with special health care needs into moderate- or high-risk categories.2–4 The term “special health care needs” encompasses a broad range of diagnoses and is comprehensively defined by the American Academy of Pediatric Dentistry as individuals with “any physical, developmental, mental, sensory, behavioral, cognitive, or emotional impairment or limiting condition that requires medical management, health care intervention, and/or use of specialized services or programs.”5 Intake forms and histories help clinicians identify patients with special health care needs. A “review of systems” is a structured way of cataloging a patient’s medical history by organ system, and may be helpful in examining conditions associated with increased caries risk (click here for an example).
Not all children with special health care needs are at high risk of caries, and prevention programs should be tailored to individual risk factors. Attention to the patient’s need for a cariogenic diet, use of medications that may be dosed in sugary suspensions or cause xerostomia, inability to maintain optimal oral hygiene, decreased salivary buffering capacity, and other condition-specific caries risk factors can help providers identify successful prevention strategies (Table 1)
EAR, NOSE, AND THROAT CONDITIONS
Given their physical proximity to the oral cavity, conditions affecting the ear, nose, and throat may significantly affect caries risk. Physical challenges for children with special health care needs, such as decreased nasal passages in individuals with craniosynostosis, may increase the chance of typical childhood infections, including sinusitis or otitis media. They can also exacerbate conditions such as obstructive sleep apnea.6
Sinusitis, transient or chronic obstruction of the nasal passages, and conditions such as cerebral palsy can produce mouth-breathing behaviors, which increase the risk of xerostomia. A study of children ages 3 to 5 found that those with parent-reported mouth-breathing behaviors had significantly more white spot lesions than children who were predominantly nasal breathers.7 Otitis media is common in young children and has been linked to enamel defects.8 It is believed these defects increase caries risk and are a result of early insult to sensitive ameloblasts during enamel formation. Population studies confirm that children with at least one treated episode of otitis media in the first year of life were at 29% greater risk of developing early childhood caries.6
Children who experience problems with muscle tone, such as those with muscular dystrophy or cerebral palsy, or with genetic conditions, such as Down syndrome, are at increased risk of obstructive sleep apnea.9 While obstructive sleep apnea can impact quality of life and is a contraindication for having dental work performed with sedation, a 2015 study showed these patients had fewer caries, less plaque deposition, and better gingival health than healthy controls.10
RESPIRATORY DISEASES
Children frequently experience acute respiratory diseases, but chronic respiratory conditions, such as asthma, cystic fibrosis, and allergic rhinitis, may alter the caries risk profile.11 Xerostomia is a common side effect of respiratory drug therapies that reduce buffering capacity and remineralization ability, leading to elevated caries risk.12 Many individuals with chronic respiratory conditions also experience severe gastroesophageal reflux disease (GERD), and the resulting acid regurgitation can erode tooth enamel.13
Characterized by airway inflammation and bronchioconstriction, asthma affects 9.3% of children in the United States.14 Treatment includes long-term use of medications, such as albuterol or inhaled corticosteroids, which can reduce salivary flow.15 Increased severity of the disease and a high number of therapeutic medications are associated with increased caries prevalence.16 In comparison to healthy controls, children with asthma are reported to have higher caries prevalence, especially in the primary dentition.17–19
Cystic fibrosis is the most common lethal genetic disease in Caucasians.20 It is caused by a defect in the cystic fibrosis transmembrane regulator gene that results in thick mucus obstructions throughout the body, most notably in the lungs.21 Children with cystic fibrosis have a number of caries risk factors, including a high prevalence of enamel defects, increased GERD, frequent use of inhaled xerostomia-inducing medications, and frequent need for nutrient intake due to the thickened mucosal lining.19,22,23Clinicians should monitor salivary flow for children with chronic respiratory conditions, and offer recommendations as appropriate. Most children with chronic respiratory conditions are poor candidates for dental treatment using in-office sedation, making caries prevention critical.19
GASTROINTESTINAL CONCERNS
Common pediatric chronic gastrointestinal conditions that impact caries risk include GERD and celiac disease. These are associated with high levels of acid erosion and enamel defects, which weaken the outer tooth layer, leaving it susceptible to caries lesions.13 In one study, children with celiac disease demonstrated a higher prevalence of enamel defects compared with matched healthy controls, and presence of enamel defects in both groups was positively associated with caries experience.24
The caries risk associated with other gastrointestinal conditions appears to be determined by individual dietary habits instead of the underlying illness. In a study of children with end-stage liver disease, only frequency of nighttime feeding—not disease severity—was associated with caries risk.25
Children with impairments that make feeding by mouth difficult are often fed by tube. Although significant calculus accumulation is common in these patients, caries activity is usually low.26 Practitioners should inquire if tube-fed children take any “social tastes” by mouth, as frequent introductions of sugar-sweetened foods may increase the otherwise low caries risk of this population.
RENAL DISORDERS
Advances in pediatric nephrology have resulted in increased survival for children with acquired or congenital renal disorders.27Despite the need for a carbohydrate-rich, protein-sparing diet, children with chronic kidney disease have dramatically lower reported caries prevalence when compared with healthy controls.28 This is almost certainly due to an increased total body pH that results in increased salivary buffering capacity, which, in turn, promotes enamel remineralization.29
Metabolic and physiologic changes associated with chronic renal failure affect mineralization of developing teeth. Kidney disease results in hypocalcemia, low serum levels of vitamin D3, and raised levels of inorganic phosphate and parathyroid hormone that cause enamel hypoplasia.30 While previously protected by high salivary pH, these individuals’ caries risk may change as a result of treatment. After a kidney transplant, the salivary composition returns to normal. The existing hypoplastic and pitted enamel easily harbors plaque that may increase caries risk in these now susceptible patients.29
NERVOUS SYSTEM DISORDERS
Epilepsy, the most common nervous system disorder in children, affects 1% to 3% of the US pediatric population. Reported caries prevalence in this cohort is high, and may be due to poor oral hygiene and frequent use of antiepileptic medications.31,32 Children with epilepsy often have concomitant intellectual and motor disabilities that make oral hygiene difficult. In one study, when compared to healthy controls children with epilepsy had significantly higher caries experience, brushed their teeth less often, and had worse self-rating of their oral health.32 In addition, common antiepileptic mediations, such as phenytoin, can cause gingival overgrowth that also makes oral hygiene challenging;33 although its etiology is poorly understood and likely multifactorial, optimal oral hygiene can reduce side effects.
MUSCULOSKELETAL CONCERNS
Chronic musculoskeletal conditions, such as cerebral palsy, Duchene’s muscular dystrophy, and scoliosis, may result in lifelong impaired mobility. This decrease in range of motion and dexterity often makes oral hygiene difficult, which can increase caries risk. Studies of individuals with Duchene’s muscular dystrophy and myotonic dystrophy type 1 corroborate that decreasing muscle function is associated with increased plaque and calculus accumulation. Caries experience, however, has been variable in these populations.34,35 In addition to receiving assistance with self-care, the use of specialized toothbrushes that are easy to grasp or power toothbrushes is important because dental care under general anesthesia with inhaled anesthetics is contraindicated due to the increased risk for developing malignant hyperthermia.36
Studies show that caries experience is high in individuals with bone conditions, such as osteogenesis imperfecta, fibrous dysplasia, and McCune-Albright syndrome.37 The underlying genetic cause of such conditions may also weaken tooth structure, predisposing the individual to high caries risk.
HEMATOLOGIC DISORDERS
Disorders of the blood in children are characterized as: red cell (anemia, thalassemia, sickle cell disease); white cell (chronic granulomatous disease, leukocyte adhesion deficiency, cyclic neutropenia, severe congenital neutropenia/Kostmann syndrome); failures of the bone marrow (leukemia, aplastic anemia, Fanconi anemia, congenital amegakaryocytic thrombocytopenia); bleeding disorders (hemophilia, von Willebrand disease, thrombocytopenia); and autoimmune conditions (immune thrombocytopenia, Evans syndrome). Acquired hematologic conditions, such as iron deficient anemia, have been associated with severe early childhood caries, but this is thought to result from malnutrition stemming from the poor dentition.38 Studies on genetic hematologic conditions, such as sickle cell disease or thalassemia, show that other known caries risk factors (diet or oral hygiene) have more influence than the underlying disease condition.39-41 Moreover, a study of children with hemophilia showed they had lower caries experience than healthy controls, and multivariate modeling confirmed that hemophilia status was not a risk factor beyond other known caries risk determinants.42
Leukemia is the most common childhood cancer, and treatment can cause oral complications, such as xerostomia, mucositis or dysgeusia, that increase caries risk.43 Additionally, treatment causes immunosuppression, which can exacerbate otherwise benign oral infections into life-threatening complications. This resulting neutropenia contraindicates most nonpalliative dental procedures.44Although survival rates for childhood cancers are increasing, an elevated risk for caries remains into adulthood. A study of childhood cancer survivors, for example, demonstrates higher caries experience than healthy controls years after remission.45 These subjects had a higher prevalence of dental anomalies associated with increased caries risk.45 Aggressive preventive measures, including optimal oral hygiene (when not in a neutropenia state), use of chemotherapeutics, and coordination between medical and dental teams, are essential to mitigate caries risk in these patients.
METABOLIC CONDITIONS
Endocrine and resulting metabolic conditions in children include diabetes mellitus, metabolic disorders, and thyroid gland problems. Although periodontitis as a complication of type 1 diabetes has been studied extensively, the reported caries experience of these patients has been inconsistent.46–48 Increased risk factors include decreased salivary flow, poor oral hygiene, and poor metabolic control, resulting in increased salivary glucose levels.49
“Inborn errors of metabolism” is a term that encompasses conditions resulting from a missing or deficient enzyme needed for metabolic processes, such as the breakdown of proteins. These include phenylketonuria, maple syrup urine disease, glycogen storage diseases, homocystinuria, galactosaemia, and medium chain acyl CoA dehydrogenase deficiency, among others. Management of these conditions often requires adherence to a low-protein, high-carbohydrate (and thus cariogenic) diet.50 Reported caries experience in these populations has been variable, and most studies include a small number of children due to the rarity of these conditions.51 Other endocrine-related conditions, such as hypothyroidism, include enlarged tongue, malocclusion, and delayed eruption of teeth, but do not appear to influence caries risk.52
PSYCHIATRIC ISSUES
Children can experience mental health issues, including attention deficit hyperactivity disorder, autism spectrum disorder, anxiety, depression, bipolar disorder, schizophrenia, or eating disorders. Some may have a dual diagnosis of an intellectual disability. In this diverse population, use of xerostomia-inducing medications, restrictive diets, and challenging behaviors can increase caries risk. Pharmacologic treatment may includes antipsychotic and antidepressant medications, whose anticholinergic effects cause xerostomia.53 While this does not appear to be solely responsible for the increased caries experience of this population, it may synergistically influence other risk factors.54
Challenging behaviors can affect an individual’s caries risk. Oral aversions are common in children with autism, and are associated with increased plaque levels and caries experience.55 Intellectual disabilities may also limit comprehension of the importance of self-care, resulting in poor oral hygiene.
CONCLUSION
Children with special health care needs may be at increased risk for caries. Therapy can prove challenging, and aggressive preventive measures—such as frequent recare intervals and regular fluoride treatment—should be implemented. In addition to encouraging optimal oral hygiene, clinicians should address risk factors resulting from characteristics of the individual’s underlying condition/treatment. Understanding these factors will help oral health professionals formulate and implement effective caries prevention strategies.
REFERENCES
- Tsang P, Qi F, Shi W. Medical approach to dental caries: fight the disease, not the lesion. Pediatr Dent. 2006;28:188–198.
- American Academy of Pediatric Dentistry. Guideline on caries-risk assessment and management for infants, children, and adolescents. Pediatr Dent. 2016;38:142–149.
- American Dental Association Caries Risk Assessment Form. Available at: ada.org/~/media/ADA/Science and Research/Files/topic_caries_over6.ashx. Accessed February 10, 2017.
- Jenson L, Budenz AW, Featherstone JD, Ramos-Gomez FJ, Spolsky VW, Young DA. Clinical protocols for caries management by risk assessment. J Calif Dent Assoc. 2007;35:714–723.
- American Academy of Pediatric Dentistry. Definition of Special Health Care Needs. Pediatr Dent. 2016;38:16.
- Alaki SM, Burt BA, Garetz SL. Middle ear and respiratory infections in early childhood and their association with early childhood caries. Pediatr Dent. 2008;30:105–110.
- Nascimento Filho E, Mayer MP, Pontes P, Pignatari AC, Weckx LL. Caries prevalence, levels of mutans streptococci, and gingival and plaque indices in 3.0- to 5.0-year-old mouth breathing children. Caries Res. 2004;38:572–575.
- Tapias MA, Gil A, Jiménez R, Lamas F. [Factors associated with dental enamel defects in the first molar in a population of children]. Aten Primaria. 2001;27:166–171.
- American Thoracic Society. Patient Information Series: Obstructive Sleep Apnea in Children. Am J Respir Crit Care Med. 2012;180:5–6.
- Al-Hammad NS, Hakeem LA, Salama FS. Oral health status of children with obstructive sleep apnea and snoring. Pediatr Dent. 2015;37:35–39.
- Widmer RP. Oral health of children with respiratory diseases. Paediatr Respir Rev. 2010;11:226–232.
- Flanagan D. Inspissated oral secretions and a review of their clinical, biological, and physiological significance. Spec Care Dentist. 2012;32:99–104.
- Ersin NK, Onçağ O, Tümgör G, Aydoğdu S, Hilmioğlu S. Oral and dental manifestations of gastroesophageal reflux disease in children: a preliminary study. Pediatr Dent. 2006;28:279–284.
- Akinbami LJ, Simon AE, Rossen LM. Changing trends in asthma prevalence among children. Pediatrics. 2016;137.
- Santos NC, Jamelli S, Costa L, et al. Assessing caries, dental plaque and salivary flow in asthmatic adolescents using inhaled corticosteroids. Allergol Immunopathol (Madr). 2012;40:220–224.
- Ersin NK, Gülen F, Eronat N, et al. Oral and dental manifestations of young asthmatics related to medication, severity and duration of condition. Pediatr Int. 2006;48:549–554.
- Paganini M, Dezan CC, Bichaco TR, de Andrade FB, Neto AC, Fernandes KB. Dental caries status and salivary properties of asthmatic children and adolescents. Int J Paediatr Dent. 2011;21:185–191.
- Boskabady M, Nematollahi H, Boskabady MH. Effect of inhaled medication and inhalation technique on dental caries in asthmatic patients. Iran Red Crescent Med J. 2012;14:816–821.
- Milano M, Lee JY, Donovan K, Chen JW. A cross-sectional study of medication-related factors and caries experience in asthmatic children. Pediatr Dent. 2006;28:415–419.
- Lipuma JJ. The changing microbial epidemiology in cystic fibrosis. Clin Microbiol Rev. 2010;23:299–323.
- Rowe SM, Miller S, Sorscher EJ. Cystic fibrosis. N Engl J Med. 2005;352:1992–2001.
- Ferrazzano GF, Sangianantoni G, Cantile T, Amato I, Orlando S, Ingenito A. Dental enamel defects in Italian children with cystic fibrosis: an observational study. Community Dent Health. 2012;29:106–109.
- Blondeau K, Pauwels A, Dupont LJ, et al. Characteristics of gastroesophageal reflux and potential risk of gastric content aspiration in children with cystic fibrosis. J Pediatr Gastroenterol Nutr. 2010;50:161–166.
- Avşar A, Kalayci AG. The presence and distribution of dental enamel defects and caries in children with celiac disease. Turk J Pediatr. 2008;50:45–50.
- Lin YT, Lin YT, Chen CL. A survey of the oral status of children undergoing liver transplantation. Chang Gung Med J. 2003;26:184–188.
- Dyment HA, Casas MJ. Dental care for children fed by tube: a critical review. Spec Care Dent. 1999;19:220-224.
- Subramaniam P, Gupta M, Mehta A. Oral health status in children with renal disorders. J Clin Pediatr Dent. 2012;37:89–93.
- Al Nowaiser A, Lucas VS, Wilson M, Roberts GJ, Trompeter RS. Oral health and caries related microflora in children during the first three months following renal transplantation. Int J Paediatr Dent. 2004;14:118–126.
- Lucas VS, Roberts GJ. Oro-dental health in children with chronic renal failure and after renal transplantation: a clinical review. Pediatr Nephrol. 2005;20:1388–1394.
- Nunn JH, Sharp J, Lambert HJ, Plant ND, Coulthard MG. Oral health in children with renal disease. Pediatr Nephrol. 2000;14:997–1001.
- Goyal A, Bhadravathi MC, Kumar A, Narang R, Gupta A, Singh H. Comparison of dental caries experience in children suffering from epilepsy with and without administration of long term liquid oral medication. J Clin Diagnostic Res. 2016;10:ZC78–ZC82.
- Gurbuz T, Tan H. Oral health status in epileptic children. Pediatr Int. 2010;52:279–283.
- Mohan RP, Rastogi K, Bhushan R, Verma S. Phenytoin-induced gingival enlargement: a dental awakening for patients with epilepsy. BMJ Case Rep. 2013:1–3.
- Symons AL, Townsend GC, Hughes TE. Dental characteristics of patients with Duchenne muscular dystrophy. ASDC J Dent Child. 2002;69:277–283, 234.
- Engvall M, Sjögreen L, Kjellberg H, Robertson A, Sundell S, Kiliaridis S. Oral health status in a group of children and adolescents with myotonic dystrophy type 1 over a 4-year period. Int J Paediatr Dent. 2009;19:412–422.
- Kawaai H, Tanaka K, Yamazaki S. Continuous infusion propofol general anesthesia for dental treatment in patients with progressive muscular dystrophy. Anesth Prog. 2005;52:12–16.
- Akintoye SO, Boyce AM, Collins MT. Dental perspectives in fibrous dysplasia and McCune-Albright syndrome. Oral Surg Oral Med Oral Pathol Oral Radiol. 2013;116:e149–155.
- Schroth RJ, Levi J, Kliewer E, Friel J, Moffatt ME. Association between iron status, iron deficiency anaemia, and severe early childhood caries: a case-control study. BMC Pediatr. 2013;13:22.
- Passos CP, Santos PR, Aguiar MC, et al. Sickle cell disease does not predispose to caries or periodontal disease. Spec Care Dentist. 2012;32:55–60.
- Lugliè PF, Campus G, Deiola C, Mela MG, Gallisai D. Oral condition, chemistry of saliva, and salivary levels of Streptococcus mutans in thalassemic patients. Clin Oral Investig. 2002;6:223–226.
- Hattab FN, Hazza’a AM, Yassin OM, al-Rimawi HS. Caries risk in patients with thalassaemia major. Int Dent J. 2001;51:35–38.
- Žaliu¯niene R, Alekseju¯niene˙ J, Brukiene˙ V, Pecˇiuliene˙ V. Do hemophiliacs have a higher risk for dental caries than the general population? Medicina (Kaunas). 2015;51:46–56.
- Sonis ST, Elting LS, Keefe D, et al. Perspectives on cancer therapy-induced mucosal injury: pathogenesis, measurement, epidemiology, and consequences for patients. Cancer. 2004;100:1995–2025.
- American Academy of Pediatric Dentistry. Guideline on dental management of pediatric patients receiving chemotherapy, hematopoietic cell transplantation, and/or radiation therapy. Pediatr Dent. 2016;38:334–342.
- Lauritano D, Petruzzi M. Decayed, missing and filled teeth index and dental anomalies in long-term survivors leukaemic children: a prospective controlled study. Med Oral Patol Oral Cir Bucal. 2012;17:e977–980.
- Arheiam A, Omar S. Dental caries experience and periodontal treatment needs of 10- to 15-year old children with type 1 diabetes mellitus. Int Dent J. 2014;64:150–154.
- Siudikiene J, Machiulskiene V, Nyvad B, Tenovuo J, Nedzelskiene I. Dental caries and salivary status in children with type 1 diabetes mellitus, related to the metabolic control of the disease. Eur J Oral Sci. 2006;114:8–14.
- Lamster IB, Lalla E, Borgnakke WS, Taylor GW. The relationship between oral health and diabetes mellitus. J Am Dent Assoc. 2008;139:19S–24S.
- Karjalainen KM, Knuuttila ML, Käär ML. Relationship between caries and level of metabolic balance in children and adolescents with insulin-dependent diabetes mellitus. Caries Res. 1997;31:13–18.
- Cleary MA, Francis DE, Kilpatrick NM. Oral health implications in children with inborn errors of intermediary metabolism: a review. Int J Paediatr Dent. 1997;7:133–141.
- Lucas VS, Contreras A, Loukissa M, Roberts GJ. Dental disease indices and caries related oral microflora in children with phenylketonuria. ASDC J Dent Child. 2001;68:263–267.
- Little JW. Thyroid disorders. Part II: hypothyroidism and thyroiditis. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2006;102:148–153.
- Vinayak V, Annigeri R, Patel H, Mittal S. Adverse affects of drugs on saliva and salivary glands. J Orofac Sci. 2013;5:15–20.
- Rosenberg SS, Kumar S, Williams NJ. Attention deficit/hyperactivity disorder medication and dental caries in children. J Dent Hyg. 2014;88:342–347.
- Marshall J, Sheller B, Mancl L. Caries–risk assessment and caries status of children with autism. Pediatr Dent. 2010;32:69–75.
From Dimensions of Dental Hygiene. June 2017;15(6):42-44, 47.



