
Improve the View
The dental videoscope provides direct visualization of nearly undetectable subgingival calculus deposits.
This course was published in the November 2016 issue and expires November 30, 2019. The authors have no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Discuss the role of calculus in periodontal inflammation.
- Compare and contrast the features of the dental videoscope with those of the dental endoscope.
- List the clinical applications for the dental videoscope.
Inflammatory periodontal diseases affect millions of patients in the United States and result in the vast majority of tooth loss in adults. Inflammatory periodontal diseases have also been associated with an increased incidence and prevalence of coronary artery disease, poor glycemic control, greater risk of stroke, and increased incidence of preterm birth in expectant mothers.1
Oral health professionals are well aware that biofilm is the primary extrinsic etiologic factor for inflammatory periodontal diseases. As the bacterial front progresses in the deepening sulcus, calculus forms on the root surface. It has been clearly shown that calculus deposits concentrate the toxins produced by the bacteria in the biofilm.
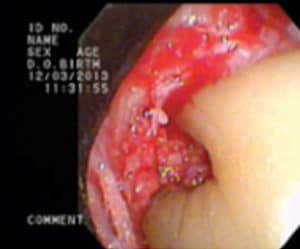
The complete removal of subgingival calculus deposits remains a significant challenge for oral health professionals. One of the most difficult aspects of deposit removal is the inability to see inside the pocket. The introduction of the dental videoscope (Figure 1)—a component-based intraoral video camera—is positioned to greatly improve clinicians’ visibility during nonsurgical periodontal therapy with the goal of boosting long-term outcomes.
BATTLE OVER BIOFILM
Articles written by Listgarten et al2–4 showed that it was possible to grow an epithelial attachment over previously root-planed subgingival calculus. This led to the theory that subgingival calculus was relatively innocuous and not directly involved in the inflammatory process of periodontal breakdown. A significant base of evidence, however, has demonstrated a direct connection between these hardened subgingival deposits and the inflammation seen in the pocket wall.5–12 This connection was made possible with the advent of new technology. A study by Wilson et al5 showed that nearly 70% of the inflammation found in the wall of an active periodontal pocket was associated with a combination of calculus and biofilm. Less than 10% of the inflammation noted in periodontal diseases was associated with bacteria plaque alone. The high level of association between calculus and clinically identifiable inflammation indicates that calculus plays a major role in the inflammatory element of periodontal diseases. This suggests that complete calculus removal is imperative to the treatment of periodontal diseases.

A NEW VIEW
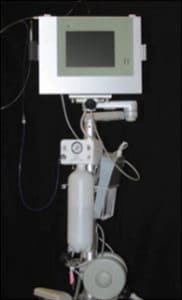
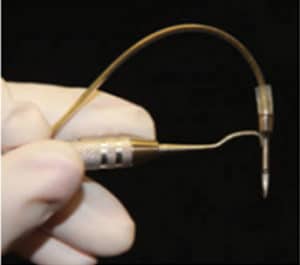
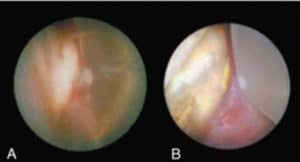
In the late 1990s, the ability to see the subgingival environment in real time was exponentially increased with the introduction of the dental endoscope (Figure 2).13 This device consists of a 0.9-mm flexible, fiber-optic cord with an externally mounted camera (Figure 3). This allows the operator to see an image on a monitor that represents a 3-mm-diameter subgingival space magnified by up to 48 times (Figure 4A and Figure 4B). The dental endoscope has revolutionized the treatment of probing depths in the range of 4 mm to 6 mm.
Through the use of the dental endoscope, the direct relationship between deposits of subgingival calculus and inflammation seen in the pocket wall has been demonstrated.5 As inflammation in these areas is a contributory factor to the progression of the disease process, as much of the subgingival calculus as possible must be removed. In the past, clinicians relied on tactile sense and clinical knowledge of root anatomy to determine the effectiveness of nonsurgical periodontal therapy. With the endoscope, the clinician is able to more effectively remove the calculus through better visualization and magnification of the subgingival space. Additionally, the improvement in visualization enables the clinician to provide a more thorough diagnosis and definitive nonsurgical therapy.
A series of studies by Stambaugh et al13,14 showed the efficacy of calculus detection and removal using the endoscope. In the first study, four dental hygienists evaluated 42 teeth with pockets greater than 5 mm.14 The teeth were subsequently extracted and evaluated with direct vision and scanning electron microscopy. The results showed that more than 95% of all root deposits were detected correctly with the endoscope. In a follow-up study, 31 patients with refractory periodontitis were subjected to scaling and root planing in 73 quadrants using the endoscope.13 The authors noted improvement in 71 out of the 73 quadrants tested following calculus removal. The authors saw improvements in probing depths and clinical attachment gain and reductions in bleeding on probing. Additionally, the authors noted that the pocket depth reduction continued to improve and was maintained over time.
The dental videoscope represents a significant advance in viewing technology. This device enables the clinician to view the subgingival environment much more accurately than the images seen with the dental endoscope, surgical telescopes (loupes), or surgical microscope. Images on the dental videoscope are in natural color and magnified from 20 times to 60 times, depending on the distance from the root surface. Unlike the endoscope, which uses a fiber optic cord to transmit the image to an external camera, the videoscope has a camera at the end of the scope that transmits the digital image to a monitor (Figure 5). A gas shield (air) is used to keep the field of view clean and prevents the videoscope from fogging. The videoscope makes it possible to see calculus deposits that would be not be visible by any other approach. It also allows the clinician to remove the calculus in real time and under direct visualization.15
The routine presence of very small lines or microgrooves on the root surfaces within periodontal defects was an unexpected finding uncovered by videoscope use. These lines or microgrooves are noted in the vast majority of periodontal lesions. These microgrooves, termed “Harrel’s lines” after their discoverer, appear to be routinely filled with small deposits of subgingival calculus. These calculus-filled microgrooves are not visible by any other device currently available and certainly not by the naked eye.
The fact that these microgrooves have not been previously detected seems to indicate that the calculus within the grooves has routinely been left following periodontal treatment. As such, their presence may be a factor in the unpredictability of periodontal therapy outcomes.
CLINICAL APPLICATION
The dental videoscope is routinely used for a regenerative periodontal surgical procedure know as videoscope-assisted minimally invasive periodontal surgery (VMIS).15 Briefly, VMIS consists of very small incisions of 3 mm to 5 mm in length that are usually placed only on the lingual side. This avoids surgery on the esthetically sensitive facial aspect of the teeth. Split thickness incisions are recommended to help maintain the blood supply to the flap. Full thickness flap reflection or reflection with a periosteal elevator often compromises the blood supply to the delicate papillary tissues and is not recommended for a minimally invasive approach. The lingual flap is carefully reflected just enough to allow for the placement of the videoscope so that the periodontal defect can be visualized on a monitor.
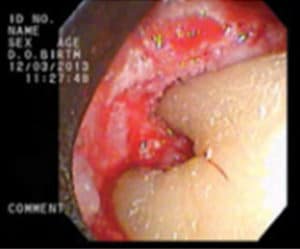
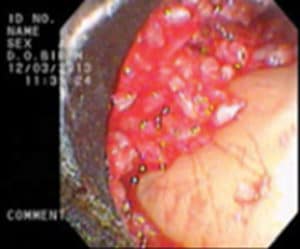
The periodontal lesion is debrided using a combination of hand curets and ultrasonic instrumentation to remove the infected granulation tissue, thus resulting in a root surface that is smooth with all calculus removed (Figure 6). Following calculus removal, the root surface is often treated with a root conditioning agent, such as ethylenediaminetetraacetic acid (EDTA), to remove any residual thin sheets of calculus (Figure 7). These sheets of calculus are not visible with the endoscope or any other type of magnification. The removal of the sheets of calculus is the critical last step in complete debridement of the defect. In most instances, a bone graft and enamel matrix derivatives are placed in the site (Figure 8), after which a single vertical mattress suture is used to close the surgical site.
The calculus-filled microgrooves are detectable during the debriding and smoothing of the root surface. Sheets of calculus are present in the periodontal defect, even after closed root planing has been performed. After the gross calculus is removed, the underlying root surface containing microgrooves can be seen with the videoscope. Traditional root planing motions will not remove the calculus from these grooves because the depths of the microgrooves are not accessible with curets or ultrasonic inserts/tips. Usually the microgrooves need to be cleaned by carefully using the tip of a curet, followed by the use of EDTA to dissolve the final smear layer of microscopic calculus. Calculus removal must be performed under direct visualization. Currently, the videoscope is the only method available that provides the direct vision needed for this procedure. Occasionally, the grooves and sheets of calculus need to be removed with a surgical bur to allow for complete debridement of the defect.
In addition to providing better visualization and complete debridement of the defect, the use of the dental videoscope in combination with a minimally invasive approach has additional post-surgical benefits for patients. Due to the minimally invasive nature of the incisions and the small amount of soft tissue reflection, patients typically experience less surgical trauma, post-operative pain, post-operative recession, and post-operative cold sensitivity. One of the biggest advantages to the minimally invasive approach is the lack of recession at the surgical site. In a 2010 study, Harrel et al16 noted 0.01 mm of recession at 1 year and 0.08 mm of recession 6 years post-surgery when using this minimally invasive approach.
In 2016, Harrel et al17 conducted a study on 30 patients with 110 sites of residual pocket probing depths of at least 5 mm, 2 mm loss of clinical attachment level, and radiographic evidence of bone loss. The patients received VMIS. At 6 months post-surgery, the patients presented with statistically significant improvement in pocket probing depths and clinical attachment levels. The approach has enabled clinicians to promote the regeneration of bone, ligament, and cementum using very small incisions, minimal reflection of tissue, maintenance of the blood supply, and restriction of the surgery only to those areas of localized, deep pocketing.

CALCULUS REMOVAL IS KEY
The role of subgingival calculus in the etiology and continuation of inflammatory periodontal diseases should be reassessed. The knowledge base regarding periodontal diseases and their treatment is changing. The relevant questions now become to what degree and with what approaches should these accretions be removed.
Ample data exist in the classic literature indicating that individuals with inflammatory periodontal diseases who have effective personal oral hygiene and who comply with routine maintenance visits tend to keep their teeth longer than individuals without these habits. Routine efforts on the part of patients and oral health professionals often result in stability of the periodontal lesion over the long-term. The question is how many individuals exhibit exemplary oral hygiene and maintain the number of suggested visits for periodontal maintenance? Unfortunately, the answer is a small percentage of those with periodontal diseases. As a result, progression of bone loss often occurs in noncompliant individuals.
These findings suggest a more thorough removal of subgingival calculus is necessary, in addition to encouraging better oral hygiene and maintenance compliance. So the next question is, what is the most effective way to achieve this goal and thereby increase the probability of stability for these lesions over the long term? Traditionally, closed subgingival scaling and root planing has been the answer. Multiple studies indicate the lack of effectiveness in removing subgingival accretions especially in probing depths deeper than 5 mm.18–20 In 1978, Waerhaug21 noted that in periodontal pockets greater than 5 mm, the chance of failure to remove all the calculus during closed subgingival scaling and root planing was high. This led the author to conclude that “one of the human causes of failure in subgingival scaling is that the operator cannot see what he or she is doing. But it may be equally bad that the operator cannot see the results of what he or she did not do. That is so because inflammatory foci caused by plaque remnants do not give clinical symptoms, which are visible at the gingival margin.”21
CONCLUSION
So how does the clinician working in private practice deal with this conundrum? For patients with probing depths in the range of 4 mm to 6 mm, the dental endoscope is a potential answer. For patients whose probing depths are deeper or possible regeneration of lost periodontal attachment is the goal, the use of the dental videoscope presents the most attractive alternative available at this time.
REFERENCES
- Friedewald VE, Kornman KS, Beck JD, Genco R,et al. The American Journal of Cardiology andJournal of Periodontology editor’s consensus:periodontitis and atherosclerotic cardiovasculardisease. J Periodontol. 2009;80:1021–1032.
- Listgarten MA, Ellegaard B. Electronmicroscopic evidence of a cellular attachmentbetween junctional epithelium and dentalcalculus. J Periodontal Res. 1973;8:143–50.
- Listgarten MA, Mayo H, Amsterdam M.Ultrastructure of the attachment device betweencoccal and filamentous microorganisms in “corncob” formations of dental plaque. Arch Oral Biol.1973;18:651–656.
- Listgarten MA. Structure of surface coatings onteeth. A review. J Periodontol. 1976;47:139–147.
- Wilson TG, Harrel SK, Nunn ME, Francis B, Webb K.The relationship between the presence of toothbornesubgingival deposits and inflammationfound with a dental endoscope. J Periodontol.2008;79:2029–2035.
- Wilson TG Jr, Carnio J, Schenk R, Myers G.Absence of histologic signs of chronic inflammation following closed subgingival scaling and root planing using the dental endoscope:human biopsies—a pilot study. J Periodontol. 2008;79:2036–2041.
- Canis MF, Kramer GM, Pameijer CM. Calculusattachment: review of the literature and findings.J Periodontol. 1979;50:406.
- Kepic TJ, O’Leary TJ, Kafrawy AH. Total calculusremoval: an attainable objective? J Periodontol.1990;61:16–20.
- Fleischer HC, Mellonig JT, Brayer WK, Gray JL,Barnett JD. Scaling and root planing efficacy in multi-rooted teeth. J Periodontol.1989;60:402–409.
- Brayer WK, Mellonig JT, Dunlap RM, MarinakKW, Carson RE. Scaling and root planing effectiveness: the effect of root surface access and operator experience. J Periodontol.1989:60:67–72.
- Stambaugh RV, Myers GC, Watenabe J, Lass C,Stambaugh KA. Clinical response to scaling androot planing aided by the dental endoscope[abstract]. J Dent Res. 2000;79:2762.
- Tan B, Gillam DG, Mordan NJ, Galgut PN. Apreliminary investigation into the ultrastructure ofdental calculus and associated bacteria. J ClinPeriodontol. 2004;31:364.
- Stambaugh RV, Myers GC, Ebling WV, Beckman B,Stambaugh KA. Endoscopic visualization of thesubmarginal gingiva dental sulcus and tooth rootsurfaces. J Periodontol. 2002;73:374–382.
- Stambaugh RV, Myers GC, Ebling WV, Beckman B,Stambaugh KA. Endoscopic visualization ofsubmarginal gingival root surfaces. J Dent Res.2000;79(Spec Issue):600.
- Harrel SK, Abraham CM, Rivera-Hidalgo F,Sbulman J, Nunn M. Videoscope-assisted minimallyinvasive periodontal surgery (V-MIS). J ClinPeriodontol. 2014;41:900–907.
- Harrel SK, Wilson TG, Nunn ME. Prospectiveassessment of the use of enamel matrix proteinswith minimally invasive surgery: Six year results.J Periodontol. 2010;81:435–441.
- Harrel SK, Abraham CM, Rivera-Hidalgo F,Shulman JD, Nunn ME. Videoscope-assistedminimally invasive periodontal surgery: one-yearoutcome and patient morbidity. Int JPeriodontics Restorative Dent. 2016;36:363–371.
- Deas DE, Moritz AJ, Sagun RS Jr, Gruwell SF,Powell CA. Scaling and root planing vs. conservativesurgery in the treatment of chronic periodontitis.Periodontol 2000. 2016;71:128–139.
- Pihlstrom BL, McHugh RB, Oliphant TH, Ortiz-Campos C. Comparison of surgical and nonsurgicaltreatment of periodontal disease. A review ofcurrent studies and additional results after 61?2 years.J Clin Periodontol. 1983;10:524–541.
- Heitz-Mayfield LJ, Trombelli L, Heitz F,Needleman I, Moles D. A systematic review of theeffect of surgical debridement vs non-surgicaldebridement for the treatment of chronicperiodontitis. J Clin Periodontol. 2002;29(Suppl 3):92–102.
- Waerhaug J. Healing of the dento-epithelialjunction following subgingival plaque control II: Asobserved on extracted teeth. J Periodontol.1978;49:119–134.
From Dimensions of Dental Hygiene. November 2016;14(11):53–56.



